REVIEW: Role of Histidine-Related Compounds as Intracellular Proton Buffering Constituents in Vertebrate Muscle
H. Abe
Laboratory of Marine Biochemistry, Graduate School of Agricultural and Life Sciences, The University of Tokyo, Bunkyo-ku, Tokyo 113-8657, Japan; fax: +81-3-5841-8166; E-mail: aabe@mail.ecc.u-tokyo.ac.jp
Received November 22, 1999
The intracellular non-bicarbonate buffering capacity of vertebrate muscle is mainly supported by the imidazole groups of histidine residues in proteins, free L-histidine in some fish species, and histidine-containing dipeptides such as carnosine, anserine, and balenine (ophidine). The proton buffering capacity markedly differs between muscle types and animal species depending on the ability for anaerobic exercise. The capacity is typically high in fast-twitch glycolytic muscles of vertebrates adapted for anaerobic performance such as burst swimming in fishes, prolonged anoxic diving in marine mammals, flight in birds, sprint running in mammalian sprinters, and hopping locomotion in some terrestrial mammals. A high correlation between buffering capacity, concentration of histidine-related compounds in muscle, and percentage of fast-twitch fibers in all vertebrates adapted for intense anaerobic performance clearly supports the idea that proton buffering is the main physiological function of histidine-related compounds.1
KEY WORDS: histidine, carnosine, anserine, balenine, buffering capacity, vertebrate, muscle, anaerobic exercise
1Editor's note: Carnosine is now well known to be an efficient intracellular pH buffer (V. Skulachev, H. Abe), hydrophilic antioxidant (A. Boldyrev, E. Decker), heavy metal chelator (P. Trombley, E. Baran), potent anti-glycating agent (A. Hipkiss), and regulator of many specific receptors (F. Margolis, A. Fasolo, D. Miller) and enzymes (I. Severina, S. Stvolinsky). These features make this compound useful to treat ischemic brain and heart (D. Dobrota, P. Roberts, G. Zaloga) and to decelerate some senescence processes (A. Wang, S. Gallant, R. Holliday, G. McFarland). Many of these properties of carnosine are discussed in this volume.
During high-intensity anaerobic exercise, a large number of protons accumulate in vertebrate muscle as ATP is hydrolyzed to ADP [1]. The proton accumulation causes a decrease in intracellular pH (pHi) which, in turn, causes the inactivation of glycolytic enzymes such as phosphofructokinase and hence a decrease in glycolytic flux [2]. The decrease in glycolytic flux due to low pHi is also found during prolonged anoxia of perfused rat heart [2]. The changes of pHi are known to affect the rate of other various metabolic functions [2]. As a defense mechanism against changes in pHi, proton buffering systems may evolve in the cell. Thus, a high buffering capacity in muscle can stabilize intramuscular pH and enhance the capability for anaerobic exercise performance or anoxia tolerance [2, 3].
The intracellular non-bicarbonate buffering of vertebrate muscle is dominated by the imidazole group which exists in histidine residues of proteins, in free L-histidine, and in histidine-containing dipeptides such as carnosine, anserine, and balenine (also known as ophidine) [1-4]. Because the pK values of these imidazole groups are close to pHi, one of the two nitrogens of the imidazole ring can be protonated in the physiological range of pH. Thus, imidazole groups are utilized as potent proton buffering constituents. The regulatory process keeping pHi close to the pK values of imidazole groups is called “alphastat regulation”. Its role is to maintain alpha-imidazole relatively constant (alpha-imidazole being defined as non-protonated imidazole/(non-protonated imidazole + protonated imidazole)). Typical alphaImid is conserved at a value of about 0.55 in intracellular fluid [3]. Inorganic orthophosphate also serves as a typical inorganic buffer component in addition to imidazole compounds (Table 1).
Table 1. Apparent pK values of
imidazole groups (according to [3-5])
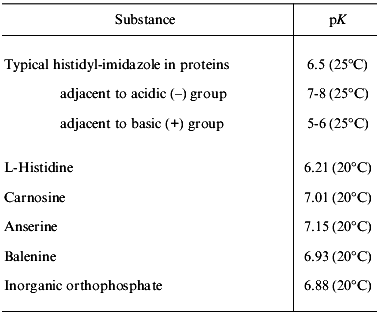
Note: pK measurements were performed at temperatures specified in
parentheses.
The proton buffering capacity markedly differs between muscle types and animal species depending on the ability for anaerobic exercise [3]. This review mainly focuses on the relationship between the buffering capacities of histidine-related compounds (HRC) and the anaerobic capabilities of vertebrate skeletal muscle.
TELEOST FISHES
Proton buffering capacity (termed beta value and measured as the “slyke” unit) is defined as the µmoles of sodium hydroxide or hydrogen chloride required to change the pH of one gram of tissue by one unit, i.e., from 6 to 7 or from 6.5 to 7.5 [4, 6-8]. Table 2 shows beta values, total HRC contents, and lactate dehydrogenase (LDH) activities in the white and red muscles of teleost fishes having a wide variety of anaerobic performance capabilities. Fish myotomal muscle comprises two functionally different muscle fiber types, red and white, as it is the case in other vertebrates. In fish, however, the two muscles are separated spatially. Fish red muscle exists as a thin triangular strip running longitudinally beneath the lateral line (superficial red muscle) or located more axially, extending to the vertebrae (deep-seated red muscle as in tuna fishes). Fish red muscle is an aerobic slow-twitch oxidative tissue and it is recruited during sustained, steady state swimming. In contrast, fish white muscle mainly consists of fast-twitch glycolytic fibers and is recruited during anaerobic burst locomotion only lasting for short times.
Table 2. Buffering capacity (beta),
concentration of histidine-related compounds (HRC), and lactate
dehydrogenase (LDH) activity in fish muscle (according to [4, 6, 8-13])
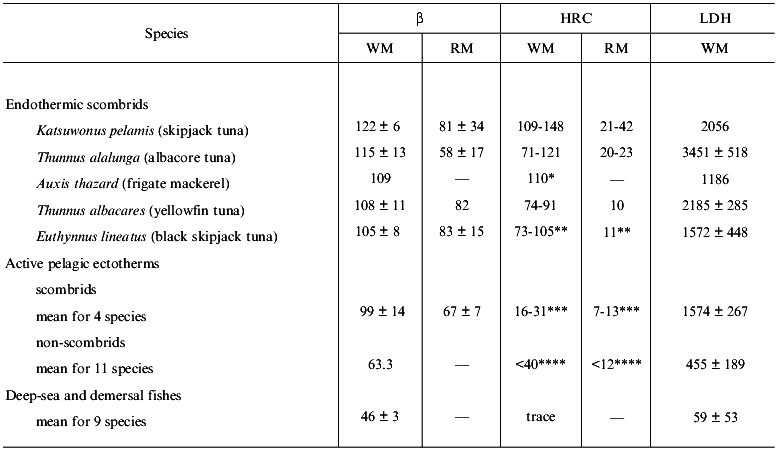
Note: Values (means ± SD) are expressed as µmole NaOH
per pH unit per gram of wet weight of muscle (pH 6-7) in case of
beta, as µmole per gram of wet weight of muscle in case of
HRC, and as activity units per gram of wet weight of muscle in case of
LDH. Abbreviations: WM, white muscle; RM, red muscle; -, not
determined.
*Values for Auxis tapeinocephalus.
**Values for Euthynnus affinis.
***Values for chub mackerel Scomber japonicus.
****Values for sardine Sardinops melanostictus.
The white muscle shows much higher beta values and HRC contents than red muscle in a given species. HRC contents in red muscle correspond generally to one-tenth to one-quarter of the contents in the white muscle of various fish species [4]. The beta values and HRC contents are typically high in endothermic scombrids such as tuna fishes. Tunas have a large mass of deep-seated red muscle surrounded by the “rete mirabile” (dense capillary network) used as a vascular countercurrent heat exchanger that maintains muscle temperature above ambient temperature by up to 10°C [3]. Tunas contain 60-100 µmol/g muscle wet weight of free L-histidine and 15-60 µmol/g muscle wet weight of anserine in their white muscle [4], and show over 100 slykes of proton buffering capacity (Table 2). In the white muscle of skipjack tuna, for instance, total HRC content reaches about 150 mM. A large amount of free L-histidine is also found in salmonids and cyprinids and is thought to have the same physiological function as histidine-containing dipeptides [4, 9]. Tunas also have the highest LDH activity (Table 2) found in any animals including mammals and they show very high capacities for anaerobic glycolysis. These observations clearly support the idea that high muscle buffering capacity is necessary to support the high speed, long duration, and increasing frequency of burst swimming typically seen in tuna fishes.
As seen in Table 2, ectothermic scombrids such as chub mackerel or Pacific bonito have lower beta values and HRC contents than warm-bodied tuna [11]. Of the actively foraging pelagic ectotherms, non-scombrids such as sardine, rainbow trout, and several bass species have much lower beta and HRC in their white muscle. Of these fish species, sardine Sardinops spp. contains 40 µmol/g of free L-histidine and rainbow trout contains about 20 µmol/g of anserine in white muscle, but other white-fleshed fishes have only trace amounts of HRC, even in their white muscle. Deep-sea and demersal fishes, with more sluggish locomotor activities, are found to have the lowest buffering capacities, HRC levels, and LDH activities [3, 8, 11].
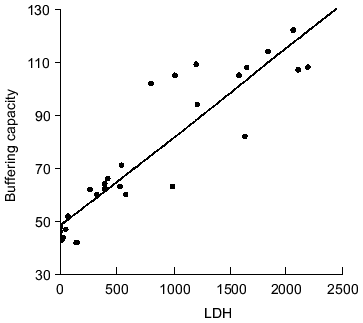
The correlation between beta values and LDH activities is shown in Fig. 1 for various fish species listed in Table 2. These two parameters are highly correlated (r = 0.83), indicating that buffering capacity is high in the white muscles having high glycolytic capacities and hence in species with high burst exercise capability [8, 11].
Fig. 1. Correlation between lactate dehydrogenase activity (units/g muscle) and buffering capacity (µmol NaOH per pH unit per g muscle, pH 6-7) in fish white muscle (data compiled from references [8, 11, 16]).
MARINE MAMMALS
Table 3 shows muscle beta values, HRC contents, and LDH activities in several diving mammals and birds, as well as terrestrial animals for comparison. The muscles of these animals contain less than 1 µmol/g of free L-histidine but a rather large amount of one of the dipeptides. Whale muscle is unique because it contains a large amount of balenine, ranging from 20 to 80 µmol/g [14]. Whale typically shows beta values as high as warm-bodied fishes. Marine diving mammals, which contain a rather large amount of carnosine in their skeletal muscle, have higher average beta_than terrestrial mammals [8]. LDH activity does not differ significantly between marine and terrestrial mammals, but correlates closely with beta [8]. The beta value also correlates strongly with muscle myoglobin concentration in these diving mammals [8]. Thus, HRC buffering may prevent a decrease in muscle pH during prolonged breath-hold diving, when oxygen stores are depleted and large amounts of lactate and protons produced.
Table 3. Buffering capacity (beta),
concentration of histidine-related compounds (HRC), and lactate
dehydrogenase (LDH) activity in the muscle of marine and terrestrial
animals (according to [8, 15,
16])
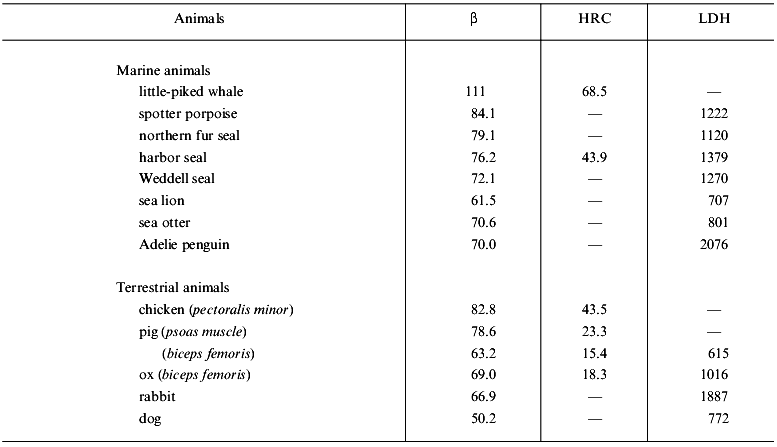
Note: See the note of Table 2 for units of
beta, HRC, and LDH activity; -, not determined.
TERRESTRIAL ANIMALS
Interspecies differences in beta values and HRC contents are very large among terrestrial animals. As shown in Table 4, terrestrial mammals and birds have large amounts of carnosine or anserine, or even a small amount of balenine. Birds typically have a larger amount of anserine than carnosine, and total HRC contents are higher in white breast muscle of chicken and turkey than in red leg muscle. This distribution pattern of HRC is also found in deer muscle, which contains the highest amount of balenine in the terrestrial mammals thus far examined. Mammals containing higher anserine than carnosine are rather limited to some special species such as kangaroo, goat and sheep, rabbit, and deer which show hopping locomotion [4]. In contrast, carnosine content is higher in ox, pig, horse, and many other mammalian muscles. As seen in Table 3, buffering capacities of these terrestrial mammals and birds are as high as those of the active pelagic fishes shown in Table 2.
Table 4. Concentration of histidine-related
compounds in the muscle of terrestrial mammals and birds (according to
[15])

Note: Values are expressed as µmol/g wet weight (means ±
SD).
*Average from the same muscle from different animals.
**Average from different muscles of one animal.
Rao and Gault [17] quantified the characteristics of bovine muscle for five white muscles abundant in fast fibers and seven red muscles abundant in slow fibers (Table 5). Carnosine content is significantly higher in white than in red muscle whereas no significant difference is seen in anserine content. In white muscle, inorganic phosphate is also significantly higher than in red muscle. After 48 h post-mortem, these muscles show an intracellular pH below 6, lower in white than in red. They exhibit no significant difference in buffering capacity from pHi to 5 (Table 5), but significantly differ from each other in beta from pHi to more acidic pH values [17]. Thus, a strong correlation exists between carnosine and inorganic phosphate contents, and buffering capacity in bovine muscle.
Table 5. Characteristics of bovine white and
red muscle (according to [17])

Note: Fiber types were based on myosin ATPase staining method.
pHi means intracellular pH. Value of beta
(pHi 5.0) means the buffering capacity (milliequivalents HCl
per 100 g muscle required to lower the muscle pH from pHi to
5.0). Values are means ± SD for five different white muscles and
for seven different red muscles.
*p < 0.01.
**p < 0.001.
Mammalian muscle is a mixture of three fiber types, slow-twitch oxidative red fiber (type I), fast-twitch oxidative-glycolytic white fiber (type IIA), and fast-twitch glycolytic white fiber (type IIB). Table 6 shows the HRC and taurine contents in the middle gluteal muscle of camel [18]. The carnosine and anserine contents of both type IIA and IIB fibers are significantly higher than those of type I fiber but there is no significant difference between type IIA and IIB. In contrast, taurine is much more concentrated in type I fiber in camel, as is the case in fish muscle; taurine content is much higher in fish red muscle than white and is thought to contribute to red muscle buffering [6].
Table 6. Concentration of histidine-related
compounds and taurine in the middle gluteal muscle of camel
(according to [18])
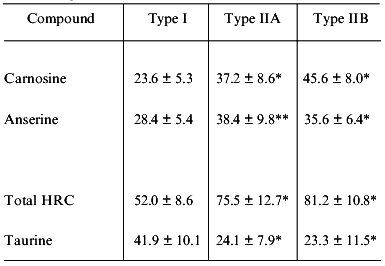
Note: Values are means ± SD for four post-mortem muscles as
mmol per kg dry weight. Type IIA and IIB are significantly different
compared with type I muscle.
*p < 0.05.
**p < 0.01.
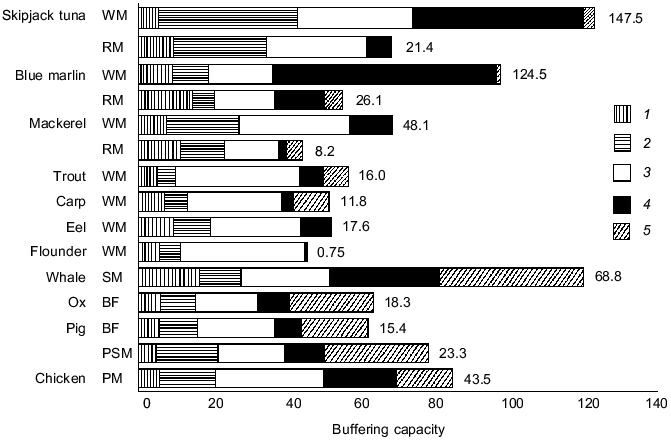
Figure 2 represents muscle homogenate buffering and the major compounds contributing to buffering in the pH range of 6.5-7.5 [4, 16]. The relative contribution of contractile proteins to total buffering is a few percents, while soluble proteins contribute from 9 to 38%. The contribution of soluble proteins is rather high for dark-fleshed fishes containing a lot of myoglobin such as tuna and mackerel. The contributions of HRC in whale skeletal muscle, skipjack tuna white muscle, and marlin white muscle are as high as 25, 40, and 60%, respectively. Blue marlin, Makaira nigricans, contains 120 µmol per g of anserine in white muscle on average [4, 6]. The contribution of HRC is also high for bovine, porcine, and chicken muscle, ranging from 12 to 23%, but it is only 1 to 6% for carp and flounder white muscle that contain only a small amount of HRC. In contrast, the contribution of inorganic phosphate to total muscle beta is high in the white muscle of trout, carp, and flounder, ranging from 50 to 80%. The concentration of inorganic phosphate is rather species independent and includes phosphate liberated from organic phosphates such as phosphocreatine and ATP. In the case of mammalian muscle, a rather high contribution is attributed to unknown compounds, which may include some nucleotides, organic acids, and taurine. These data indicate that the large variation in muscle beta is, therefore, mainly due to changing levels of HRC. Thus, the accumulation of HRC in muscle increases the muscle beta and, hence, the muscle anaerobic capability. This strategy is used by animals such as tunas, billfishes such as marlin, and marine mammals that show an elevated capacity for burst anaerobic swimming or for anoxia tolerance.
Fig. 2. Contribution of proteins (contractile (1) and soluble (2)), inorganic orthophosphate (3), histidine-related compounds (4), and unknown compounds (5) to the buffering capacity of muscle homogenate from several vertebrates. Buffering capacity is expressed as µmol NaOH per pH unit per g muscle over pH 6.5-7.5. Values indicated on the bars represent the concentration of total histidine-related compounds (µmol/g muscle). WM, white muscle; RM, red muscle; SM, skeletal muscle; BF, biceps femoris; PSM, psoas muscle; PM, pectoralis minor (from reference [4]).
MAMMALIAN SPRINTERS
The white muscle fibers (type IIA and IIB) are especially abundant in the muscle of outstanding mammalian sprinters such as thoroughbred horse (Table 7). The superficial portion of the middle gluteal muscle contains many type IIB fibers and the deep portion many type IIA and I fibers [19]. Estimated carnosine levels are over two times higher in type IIB than in IIA (Table 7). The contribution of carnosine to the total buffering capacity in muscle is estimated to be 46% in type IIB fiber.
Table 7. Fiber types, carnosine content, and
buffering capacity of the middle gluteal muscle of thoroughbred
horse (according to [19])

Note: Buffering capacity (beta) is microequivalent of
H+ per g muscle dry weight required to lower the pH from 7.1
to 6.5.
The muscle fiber type distribution and carnosine levels also differ between breeds of horses (Table 8). Of the middle gluteal muscle of Quarter Horses (sprinter), thoroughbreds, and standardbreds, type IIA fibers are lowest and type IIB are highest in Quarter Horses. Carnosine content is also the highest in Quarter Horses. From these results, we can conclude that horses selected for sprinting have a higher percentage of fast-twitch glycolytic fibers and require more muscle buffering capacity [20].
Table 8. Effects of breed of horse on muscle
fiber types and carnosine content (according to [20])
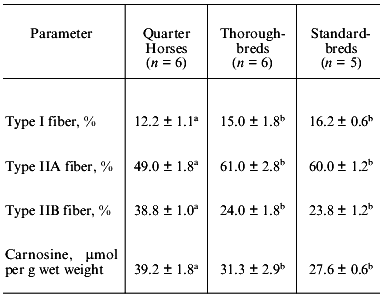
Note: Percentages of fibers are based on the staining negative for
succinate dehydrogenase for fast twitch glycolytic fiber and for myosin
ATPase for slow twitch oxidative fiber.
a, b Values in the same horizontal line with different
superscript letters are significantly different (p <
0.05).
As shown in Table 9, the beta value is also high in the muscles of another sprinter, the greyhound dog, compared with human muscle [21]. Significant differences in betatotal exist between man, horse, and dog (p < 0.001). Total HRC is also high in greyhound as well as thoroughbred horse. The contribution of HRC to betatotal reaches the high value of 25% in greyhound dogs.
Table 9. Dipeptide content and buffering
capacity in the muscle of thoroughbred horse, greyhound dog, and human
(according to [21])
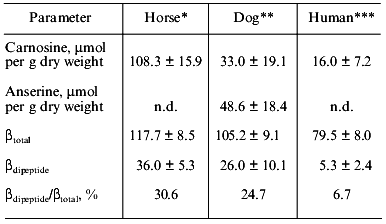
Note: See the legend of Table 7 for buffering
capacity; n.d., not detected.
*Middle gluteal muscle (n = 20).
**Mean ± SD for five different muscles in four
dogs.
***Vastus lateralis muscle (n = 20).
HUMANS
As seen in Table 9, human muscle contains a rather small amount of HRC, which contributes little to betatotal. The carnosine level in the quadriceps femoris muscle of human volunteers is significantly higher (p < 0.05) in male than in female subjects of similar age and training status (Table 10), although large inter-individual differences are found in both sexes [22]. As shown in Table 11, the carnosine level is twice as high in type II fibers of human vastus lateralis muscle compared with type I [23]. In contrast, the taurine level is four times higher in type I than type II muscle fibers as described earlier in camel and fish. This is also true in thoroughbred horses [24], where the contribution of carnosine to muscle betatotal is 9.4% in type II but only 4.5% in type I fibers.
Table 10. Carnosine content in the
quadriceps femoris muscle of male and female humans (according
to [22])
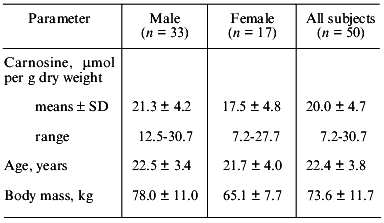
Note: Means ± SD of carnosine content are significantly different
between male and female (p < 0.05).
Table 11. Buffering capacity and carnosine
and taurine contents in the human vastus lateralis muscle (n
= 4) (according to [23])
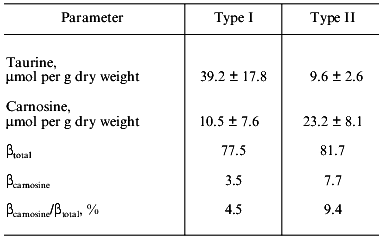
Note: See the legend of Table 7 for buffering
capacity.
Parkhouse et al. [25] characterized the effects of exercise training on muscle fiber types, HRC levels, and beta values in human vastus lateralis muscle (Table 12). Four groups of five subjects were compared: highly trained 800-m sprinters, rowers (varsity oarsmen), endurance trained marathon runners, and untrained controls. The marathon runners were significantly (p < 0.05) older than the other groups. Body weight and fat reserves were also significantly higher in untrained controls than in the other groups (data not shown). With respect to anaerobic speed test (high-intensity running performance), the sprinters performed significantly better than the rowers who also performed significantly better than the marathoners and untrained controls. Post-exercise blood lactate differed significantly between the sprinters, rowers, and the other two groups. Significant differences in the fast-twitch percentage were found between the groups (p < 0.05). Muscle buffering capacity was significantly higher in the sprinters and rowers than in the marathoners and untrained subjects. Results show no significant difference in histidine levels, but carnosine levels are significantly elevated in the sprinters and rowers. Significant (p < 0.05) correlations are found between beta and carnosine levels (r = 0.69), and beta and fast-twitch percentage (r = 0.51). Overall, these data suggest that repetitive high-intensity anaerobic exercise causes muscle adaptations that include an increase in percent fast-twitch fibers and muscle beta. The observed increase in carnosine levels may contribute, at least partly, to the increase of muscle beta values.
Table 12. Buffering capacity and
histidine-related compounds in human vastus lateralis muscle
with special reference to different exercise training (according to [25])

Note: Values are means ± SD. Buffering capacity (beta)
is µmol H+ per g muscle wet weight required to change
the pH by one unit over the pH range 7.0-6.0.
* Significantly higher (p < 0.01) than in marathoners and
untrained.
** Significantly higher (p < 0.01) than in all
other groups.
Since the early works of Bate-Smith [7] and Davey [26], the importance of non-bicarbonate intracellular buffering of vertebrate muscle has been demonstrated. In this review, I show that the buffering capacity is typically high in the fast-twitch glycolytic and anaerobic white muscle of vertebrates adapted for anaerobic performance such as burst swimming, prolonged breath-hold diving, flight, sprint running, and intense hopping locomotion. These activities are especially necessary for animals dwelling in the open oceans, grassy plains, or in the sky where they allow them to escape predators or to catch their prey. Imidazole buffer systems appear to have evolved in the muscles of vertebrates requiring high burst exercise capabilities or high anoxia tolerance for their survival. The fact that buffering capacity, HRC content, and percent fast-twitch fibers are highly correlated for a wide variety of vertebrate muscles supports the idea that the main physiological function of HRC is proton buffering. Inorganic phosphate, histidine residues in proteins, and organic phosphate compounds also contribute to proton buffering. However, the cellular concentration of these other buffering components may be constrained because their primary physiological roles have nothing to do with buffering. In contrast, HRC, and especially the metabolically inert dipeptides among them, can be stored in large amounts without harmful side effect to the cell. Finally, combining varying amounts of different HRC constituents, each with their own pK values (ranging from 6.21 to 7.15, see Table 1) may provide the necessary flexibility to cope with the particular physiological challenges of each vertebrate species.
REFERENCES
1.Hochachka, P. W., and Mommsen, T. P. (1983)
Science, 219, 1391-1397.
2.Somero, G. N. (1986) Am. J. Physiol.,
251, R197-R213.
3.Hochachka, P. W., and Somero, G. N. (1984)
Biochemical Adaptation, Princeton University Press, Princeton,
New Jersey, pp. 337-348.
4.Abe, H. (1995) in Biochemistry and Molecular
Biology of Fishes - Metabolic Biochemistry, Vol. 4 (Hochachka, P.
W., and Mommsen, T. P., eds.) Elsevier, Amsterdam, pp. 309-333.
5.Abe, H., and Okuma, E. (1991) Bull. Jap. Soc.
Sci. Fish., 57, 2101-2107.
6.Abe, H., Dobson, G. P., Hoeger, U., and Parkhouse,
W. S. (1985) Am. J. Physiol., 249, R449-R454.
7.Bate-Smith, E. C. (1938) J. Physiol.,
92, 336-343.
8.Castellini, M. A., and Somero, G. N. (1981) J.
Comp. Physiol., 143B, 191-198.
9.Abe, H., Brill, R. W., and Hochachka, P. W. (1986)
Physiol. Zool., 59, 439-450.
10.Abe, H. (1983) Bull. Jap. Soc. Sci. Fish.,
49, 1683-1687.
11.Dickson, K. A., and Somero, G. N. (1987)
Physiol. Zool., 60, 699-706.
12.Suzuki, T., Hirano, T., and Suyama, M. (1987)
Comp. Biochem. Physiol., 87B, 615-619.
13.Suyama, M., and Yoshizawa, Y. (1973) Bull.
Jap. Soc. Sci. Fish., 39, 1339-1343.
14.Suyama, M., Suzuki, T., and Yamamoto, A. (1977)
J. Tokyo Univ. Fish., 63, 189-196.
15.Abe, H., and Okuma, E. (1995) Nippon Shokuhin
Kagaku Kogaku Kaishi, 42, 827-834.
16.Okuma, E., and Abe, H. (1992) Comp. Biochem.
Physiol., 102A, 37-41.
17.Rao, M. V., and Gault, N. F. S. (1989) Meat
Sci., 26, 5-18.
18.Dunnett, M., and Harris, R. C. (1997) Res.
Veter. Sci., 62, 213-216.
19.Sewell, D. A., Harris, R. C., Marlin, D. J., and
Dunnett, M. (1992) J. Physiol., 455, 447-453.
20.Bump, K. D., Lawrence, L. M., Moser, L. R.,
Miller-Graber, P. A., and Kurcz, E. V. (1990) Comp. Biochem.
Physiol., 96, 195-198.
21.Harris, R. C., Marlin, D. J., Dunnett, M., Snow,
D. H., and Hultman, E. (1990) Comp. Biochem. Physiol.,
97A, 249-251.
22.Mannion, A. F., Jakeman, P. M., Dunnett, M.,
Harris, R. C., and Willan, P. L. T. (1992) Eur. J. Appl.
Physiol., 64, 47-50.
23.Harris, R. C., Dunnett, M., and Greenhaff, P. L.
(1998) J. Sports Sci., 16, 639-643.
24.Dunnett, M., and Harris, R. C. (1995) Equine
Veter. J., 18, 214-217.
25.Parkhouse, W. S., McKenzie, D. C., Hochachka, P.
W., and Ovalle, W. K. (1985) J. Appl. Physiol., 58,
14-17.
26.Davey, C. L. (1960) Arch. Biochem.
Biophys., 89, 303-308.