Human Carcinoma-Associated and Salivary Mucins Detected by Anti-bovine Submaxillary Mucin Antibodies
S. J. Golubović* and Ž. T. Bojić-Trbojević
Institute for the Application of Nuclear Energy-INEP, Banatska 31b, Zemun-Belgrade, Serbia and Montenegro; fax: +381-11-618-724; E-mail: sneza@inep.co.yu; zana@inep.co.yu* To whom correspondence should be addressed.
Received September 3, 2004; Revision received November 22, 2004
Polyclonal rabbit anti-bovine submaxillary mucin antibodies, anti-BSM IgG, were analyzed by autoradiography and densitometry (after SDS electrophoresis and blotting), ELISA, and IRMA assays for reactivity against native antigen BSM, deglycosylated and desialylated BSM, and human salivary and carcinoma-associated mucins. Known human tumor marker CA19-9 antigen reacted with rabbit anti-BSM IgG under different conditions, either soluble or immobilized. As soluble antigen, in IRMA it reacted with anti-BSM antibodies used both as a catcher and a tracer, as well as in combination with monoclonal anti-CA19-9 antibodies as a catcher. MUC1 mucin is a carrier of this carbohydrate antigen, CA19-9, or sialyl-Lewisa, as well as of CA15-3 antigen, a known breast tumor marker. Autoradiography and densitometry demonstrated binding of anti-BSM IgG to intact MUC1 in a sample of commercial standard preparation of CA19-9 antigen. The same method and analysis demonstrated binding of anti-BSM IgG to MUC1 and to smaller antigens of 85-120 kD in samples containing CA15-3 antigen: commercial standard preparation, human breast tissue, and human milk. In a sample of whole human saliva, reactions of both isoforms of MUC7 were detected by autoradiography, as bands of 85 and 115 kD, and densitogram analysis also demonstrated reaction with MUC5B. Chemical modifications performed as periodate oxidation and desialylation of the BSM demonstrated carbohydrate (i.e., sialic acid) epitope sensitivity for anti-BSM IgG. The results presented in this work indicate that polyclonal anti-BSM antibodies are specific for sialylated carbohydrate structure on mucins and could serve as a tool for investigation of human carcinoma-associated and salivary mucins.
KEY WORDS: mucin, antibodies, carbohydrate epitopeDOI: 10.1134/S0006297906130025
Abbreviations: AP) alkaline phosphatase; BSM) bovine submaxillary mucin; HRP) horseradish peroxidase; IRMA) immunoradiometric assay; ISM) isolated salivary mucin; WHS) whole human saliva.
Mucins are high molecular mass glycoproteins produced by epithelial
cells to protect and lubricate the lumen of the gastrointestinal,
respiratory, and urogenital tract [1]. The roles of
various mucin molecules in homeostasis and pathological processes are
still being uncovered. Nineteen human apomucin genes have been
identified to date (MUC1-MUC19) and the corresponding fully
glycosylated mucins (up to 80% carbohydrate) are designed according to
them [2]. Different tissues normally express unique
combinations of these mucin genes, and this pattern is frequently
altered in neoplasia. More often, altered carbohydrate composition of
mucins and quantitative changes in their expression are exploited as
clinical tumor markers [3, 4].
Investigation of mucins is impeded due to their large size and abundant glycosylation. Much knowledge of structure and functions of various mucin molecules is obtained by means of immunochemical techniques using mucin-specific antibodies. Many of these antibodies are specific for carbohydrate chains or their reactivity depends on peptide glycosylation [5, 6]. Because of this, one antibody could react with mucins encoded by various genes, or antibody reactivity with the same apomucin differently glycosylated is completely altered. For instance, monoclonal antibody 1116NS 19-9 specific for sialyl-Lewisa carbohydrate epitope (CA19-9 antigen) reacts with MUC1 mucin produced by colon or pancreas tumor tissues, but not with the MUC1 synthesized by normal colon or pancreas cells [7].
The International Society for Oncodevelopmental Biology and Medicine has organized several Workshops for MUC1 [8]. Characterization of 56 monoclonal antibodies against MUC1 was a subject of the last TD-4 Workshop [5, 6, 9-11]. A panel of 16 research groups from different academic, hospital, and industrial organizations participated in it. Several aspects of analysis were included regarding antibody reactivity with mucin preparations, and with synthetic peptides, pepscan epitope mapping, carbohydrate epitope mapping, and clinical/diagnostic applications of antibodies. For the majority of the MUC1 antibodies (34 from 56) epitopes were located within a 20-amino acid tandem repeat sequence and there was evidence for the involvement of carbohydrate residues in the epitopes for 16 of the remaining 22 investigated antibodies. Six of these 16 antibodies were further analyzed. Six research groups participated in this second round of the MUC1 Workshop but, unfortunately, the specificities of antibodies remained ill-defined [8]. One of conclusions of the TD-4 Workshop was that there was no obvious relationship between the type of immunogen and the specificity of each antibody. Also, it was recognized that carbohydrate residues are involved in many epitopes, by regulating epitope accessibility or masking determinants, or by stabilizing preferred conformations of peptide epitopes within the MUC1 polypeptide [9-11].
The importance of mucin glycosylation for its conformation in tumor tissue was also shown in an investigation of the monoclonal antibodies VU-2-G7 [12]. The antibody is obtained by immunization with 60-mer MUC1 triple tandem repeat peptide with N-acetyl-galactosamine (GalNAc) located at the T in the PDTR region of each repeat. This demonstrated strong reactivity with tested tumor cell lines and seemed to show that the three-dimensional configuration of the glycosylated peptide might be closer to the natural antigen expressed on tumor cells than that of just core MUC1 peptides [12].
In this work, we report immunoreactivity of polyclonal anti-BSM antibodies towards human mucins. The suggestion is that human MUC5B could be a homolog of bovine submaxillary mucin (BSM) [13]. The MUC5B mucin is one of two mucins identified in human whole saliva, a large one MG1 [14]. The smaller, monomeric MG2, which exists in two isoforms, is identified to be encoded by the MUC7 apomucin gene [15]. Our previous findings suggested reactivity of obtained antibodies to human CA19-9 antigen [16]. To investigate whether rabbit anti-BSM antibodies could serve as a reagent for human mucins and to characterize their specificity, we used BSM and human mucins from different sources as antigens and methods such as autoradiography and immunometric and enzyme-linked immunosorbent assays. Accordingly, anti-BSM antibodies were probed with native and chemically modified BSM and with structurally related human salivary mucins as well as with differently glycosylated human MUC1 mucin expressed in neoplastic tissue, CA19-9 and CA15-3 antigens.
MATERIALS AND METHODS
Materials. Mucin from bovine submaxillary glands type I (BSM), molecular mass marker proteins, Freund's adjuvant, acrylamide, N,N´-methylene-bis-acrylamide, N,N,N´,N´-tetramethylenediamine, glycine, and Tris were purchased from Sigma (USA). Albumin bovine fraction V (BSA), horseradish peroxidase type VI (HRP), agarose, 2-mercaptoethanol, Coomassie Brilliant Blue R-250, and 3,3´,5,5´-tetramethyl-benzidine (TMB) were from ICN Biomedicals, Inc. (USA). 3,3´-Diaminobenzidine tetrahydrochloride (DAB) was from Merck (Germany) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP) was from Enzymatix Ltd. (Great Britain). Alkaline phosphatase (AP) was from Biozyme Laboratories Ltd. (Great Britain). Human antigens CA19-9 and CA15-3 and monoclonal antibody, clone M602207, specific for CA19-9 were from Fitzgerald Industrial International, Inc. (USA). CA19-9 and CA15-3 antigen standards were from CIS Biointernational (France). Immobilon-P PVDF membrane was from Millipore Corporation (USA). Radioactive isotope of iodine 125I was from Institute for Isotopes Co., Ltd. (Hungary). X-Ray film 13 × 18 cm, CURIX, XP was from AGFA (Belgium). Polystyrene microtiter plates were obtained from Dynatech Laboratories Inc. (USA). Polystyrene star-tubes were from Spektar (Serbia and Montenegro). Sephadex G-75 was from Pharmacia (Sweden). Human thyroglobulin and sheep anti-rabbit IgG antibodies were prepared in our laboratory. Whole human saliva (WHS) was collected by spitting from healthy donors. Salivary mucin was isolated as previously described [17]. Human milk was taken from healthy women postpartum (2 months). Breast tumor tissue was obtained from the Institute of Oncology, Sremska Kamenica (Serbia and Montenegro) and stored at -80°C until processed. All other chemicals were “pure for analysis” grade.
Preparation of the anti-mucin antibodies [16]. Production and purification of polyclonal anti-BSM antibodies were previously described, as well as results obtained by Ouchterlony double immunodiffusion technique and by solid phase binding assay. The obtained antibodies reacted with BSM and CA19-9 antigen and not with human saliva and CA15-3 antigen in both assays [16].
Briefly, polyclonal antibodies were produced according to the method described by Hudson and Hay. Bovine submaxillary mucin (1 mg) in Freund's complete adjuvant was injected intramuscularly into a rabbit. Two booster injections (1 mg) in Freund's incomplete adjuvant were given at 2-week intervals. The rabbit was bled one week later. Reactivity of obtained antiserum with the BSM, mucin antigens CA19-9 and CA15-3, and with whole human saliva was examined by Ouchterlony double immunodiffusion technique in 1% agarose and with solid phase binding assay.
Polyclonal anti-BSM IgG was purified from antiserum using caprylic acid. The obtained antiserum was diluted with two volumes of 0.06 M acetate buffer, pH 4.0, and then 0.08 ml of caprylic acid per ml of initial antiserum volume was added. After 30 min of stirring, the solution was centrifuged and the supernatant was filtered, concentrated, and dialyzed against 0.05 M PBS (phosphate-buffered saline), pH 7.2. The concentrated IgG solution was stored (after addition of 0.1% of sodium azide) at 4°C.
Solid phase binding assay [16]. Microtiter plates were coated with 0.1 ml of BSM, CA19-9, and CA15-3 (1 and 10 µg/well) in 0.05 M carbonate/bicarbonate buffer, pH 9.6, overnight at 4°C. After blocking with 0.2 ml of 3% BSA for 1 h at 37°C, plates were washed three times with 0.2 ml of distilled water. Serially diluted anti-BSM antibodies were added to each well (0.1 ml) and incubated for 90 min at room temperature. After washing, 0.1 ml of peroxidase-labeled sheep anti-rabbit IgG (1 : 1000 dilution) was used for the detection of immunoreaction by incubating for 90 min at room temperature. Bound conjugate was detected by adding 0.1 ml of substrate, 0.003% H2O2, and 0.1 ml of chromogen, and 0.05% TMB. The reaction was terminated after 15 min by adding 0.05 ml of 0.2 M H2SO4. The optical density was measured at 450 nm using a micro plate reader (Eurogenetics, Belgium).
Labeling procedures. a) Iodination of antibodies. Polyclonal anti-BSM IgG (0.05 g) and normal rabbit IgG (0.05 g) were labeled with 1 mCi of 125I using chloramine T as oxidizing agent [18]. After incubation in 0.3 M PBS, pH 7.4, with constant mixing for 1 min, the reaction was terminated by adding an excess of L-cysteine. The derivatized antibodies were separated from unreacted 125I by gel filtration on a Sephadex G-75 column (11 × 1 cm). Elution was performed with 0.05 M PBS, pH 7.2, containing 0.05% BSA.
b) Conjugation of antibodies and BSM with enzymes. Antibodies (the obtained rabbit anti-BSM, sheep anti-rabbit IgG) and the BSM were conjugated with HRP in molecular ratio 1 : 1 according to the method of Kurstak [19]. HRP was dissolved in 0.1 M sodium bicarbonate and oxidized with 8 mM of sodium periodate. The conjugation was performed in 0.02 M carbonate buffer, pH 9.5, at room temperature for 12 h. After adding NaBH4, the conjugates were dialyzed against 0.05 M PBS, pH 7.2. The conjugates were stored (after addition of an equal volume of glycerol) at -20°C.
Antibodies (sheep anti-rabbit IgG) were conjugated with AP using glutaraldehyde as coupling agent [19]. The conjugation was performed in 0.01 M PBS, pH 7.4, containing 0.5 M MgCl2 and 10-6 M ZnCl2 (reaction buffer) with mixing at room temperature for 3 h. The reactive sites were blocked by adding an excess of L-lysine, and the solution was dialyzed extensively against the reaction buffer. The conjugate was stored (after addition of an equal volume of glycerol) at -20°C.
SDS-polyacrylamide gel electrophoresis. SDS-PAGE was performed on a 5-20% polyacrylamide gradient gel with 4% stacking gel [20, 21] using a 2001 vertical electrophoresis unit (LKB, Sweden) at 120 V at room temperature for 6 h. Samples were prepared by boiling in 0.125 M Tris-HCl buffer containing 4% SDS, 20% glycerol, and 0.1% bromophenol blue with 10% 2-mercaptoethanol (mucin antigens) or without 2-mercaptoethanol (anti-BSM antibodies) for 5 min.
Western-blot. The proteins separated by electrophoresis were transferred onto PVDF membranes using a Multiphor II Nova Blot Unit (LKB). The transfer was performed at constant power (1.2 mA/cm2) for 1.5 h. After blotting, membranes were blocked with 3% BSA overnight at 4°C. Membranes with blotted anti-BSM antibodies were incubated with: a) BSM-HRP conjugate (dilution 1 : 500), and b) sheep anti-rabbit IgG-AP conjugate (dilution 1 : 500) or sheep anti-rabbit IgG-HRP conjugate (dilution 1 : 1000). The reactions proceeded for 15 h with constant shaking at room temperature. After intensive washing, bound conjugates were visualized using H2O2 as substrate and DAB as chromogen for HRP [22]. Bound AP-conjugate was visualized using BCIP as substrate and chromogen [22].
Autoradiography. Membranes with immobilized antigens were incubated with rabbit anti-BSM IgG or non-immune rabbit IgG (control) labeled with radioactive isotope of iodine 125I. Radiolabeled antibodies were diluted with 0.05 M PBS, pH 7.2, supplemented with 2% BSA (106 cpm/ml solution) and used for autoradiography. The incubation was performed at room temperature with constant shaking overnight. After extensive washing, membranes were exposed to CURIX, XP X-ray film for 6 weeks [23].
Molecular masses of the resulting bands were calculated in relation to standard proteins stained on the blot with Ponceau S.
Densitometry. Densitograms for each sample/lane on X-ray films obtained by autoradiography (autoradiograms) were produced using the ImageMaster TotalLab v. 2.01 program. Autoradiograms were scanned by ImageScanner Amersham (Biosciences). Densitograms were produced for specific and for nonspecific binding. The obtained peaks were compared according to computed Rf values. Rf (Retardation Factor) is a measurement of position along the lane, relative to its length. By default, the first position in each lane has an Rf of 0 and the last has an Rf of 1. There is linear increase in Rf from start to finish (TotalLab User Guide). Molecular masses were not computed directly because the standard proteins were not radiolabeled.
Coating of tubes with mucin-specific antibodies. Antibodies (monoclonal anti-CA19-9, polyclonal anti-BSM) were coated on polystyrene star-tubes by physical adsorption [24]. Tubes were coated with 5 µg antibody in 0.1 ml of 0.05 M PBS, pH 7.2, overnight at 4°C. After blocking with 1% BSA at 37°C for 1 h, the tubes were washed, dried, and stored at 4°C until use.
Immunoradiometric assay (IRMA). The capture antibodies were incubated with 0.05 ml of the BSM or the CA19-9 (CIS Biointernational) standards and 0.05 ml of 0.05 M citrate buffer, pH 4.0, with constant shaking for 4 h at room temperature. After washing three times with 1 ml of distilled water, 0.1 ml of the tracer antibodies, anti-BSM IgG (5*105 cpm/tube), in 0.05 M citrate buffer, pH 4.0, was added. The incubation was performed overnight with shaking at room temperature. The excess of tracer was removed by another three wash cycles. The bound radioactivity was measured using a gamma-counter (ICN Biomedicals, Inc.). The assay conditions were determined by previous experiments varying buffer composition and incubation times. All samples were done in duplicate.
The examined concentrations of BSM standards were: 0, 2, 10, 50, 100, 500, and 1000 µg/liter.
The examined concentrations of CA19-9 standards were: 0, 15, 30, 60, 120, and 240 kIU/liter.
The examined concentrations of CA15-3 standards were: 0, 15, 40, 80, 140, and 240 kIU/liter.
Carbohydrate-sensitivity assay for anti-BSM antibodies. ELISA. Chemical modification of BSM. a) Desialylation. The BSM was desialylated according to the method previously described [25]. To the BSM solution (1 mg/ml), 0.05 ml of 2 M H2SO4 was added to obtain the final concentration of 0.2 M H2SO4. The tube was tightly closed and incubated at 80°C for 1 h. The solution was dialyzed against 5 liters of 0.05 M phosphate buffer, pH 7.4, at 4°C and used for testing.
b) Periodate oxidation. The BSM was oxidized with NaIO4 according to the procedure of Bergeron [26]. BSM (0.5 mg/ml) was oxidized with 50 mM NaIO4 in 50 mM sodium acetate buffer, pH 4.5, at 37°C in the dark for 30 min. The reaction mixture was dialyzed against 5 liters of 0.05 M phosphate buffer, pH 7.4, at 4°C and used for testing.
The same concentrations of treated and untreated BSM, in the range from 0.15 to 10 µg/well, in 0.05 M carbonate/bicarbonate buffer, pH 9.6, were coated on microtiter wells overnight at 4°C. The plates were blocked with 0.2 ml of 3% BSA for 1 h at 37°C and then washed three times with 0.2 ml of distilled water. After blocking and wash cycles, the anti-BSM IgG conjugated with HRP (1 : 1000 dilution) was added to the wells (0.1 ml) and incubated for 3 h at room temperature. The wells were washed and incubated with 0.1 ml of substrate, 0.003% H2O2, and 0.1 ml of chromogen, 0.05% TMB. After 15 min, 0.05 ml of 0.2 M H2SO4 was added and the optical density was measured at 450 nm using a micro plate reader. All samples were done in triplicate.
RESULTS
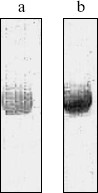
Antibody purity - SDS-polyacrylamide gel electrophoresis and Western-blot. Anti-BSM IgG purified using caprylic acid was used in this work. The purity and integrity of the isolated antibodies were judged by polyacrylamide gel electrophoresis under non-reducing conditions [21, 27]. The isolated IgG appeared as one clear band having the molecular mass of 160 kD with preserved binding capability and antigenic determinants, as shown in Fig. 1 (a and b). Anti-BSM IgG labeled with the radioactive isotope of iodine 125I was used as a tracer for autoradiography and IRMA. Anti-BSM IgG conjugated with HRP was used in ELISA.
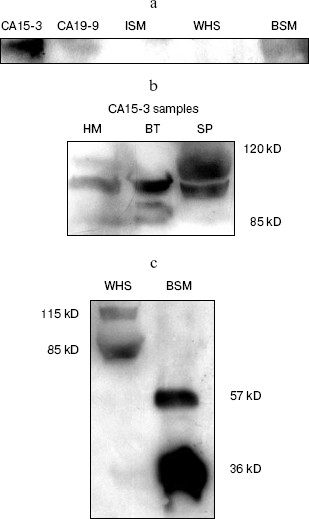
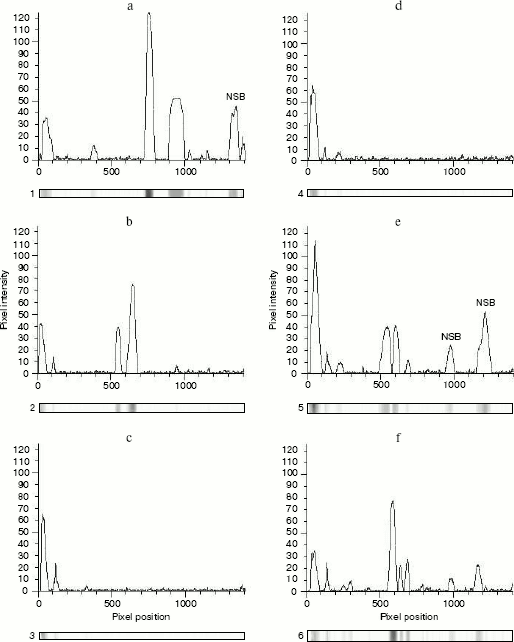
Antibody reactivity - SDS-polyacrylamide gel electrophoresis and Western-blot. Autoradiography and densitogram analysis. Our initial estimation of the anti-BSM antiserum reactivity obtained included solid phase binding assay and Ouchterlony double immunodiffusion technique. Both methods demonstrated anti-BSM antiserum reactivity toward the BSM as well as toward standard preparation of human CA19-9 antigen [16]. On the contrary, there was no reaction with standard preparation of human CA15-3 antigen or samples of whole human saliva (WHS). In this work the reactivity of MUC1 mucin from the CA19-9 antigen (Fitzgerald Industrial International, Inc.) with our rabbit anti-BSM IgG was confirmed by autoradiography after SDS electrophoresis and blotting (Fig. 2a). Besides CA19-9 antigen, samples containing CA15-3 antigen were used: commercial standard preparation (Fitzgerald Industrial International, Inc.), breast tumor tissue extract, and defatted human milk. The sample of WHS was used as a source of MG1 and MG2, as well as the sample of salivary mucin isolated by affinity chromatography. Isolated salivary mucin was obtained using monoclonal antibodies specific for sulfomucins and was identified to be MG1 (MUC5B) [17]. The results of specific binding obtained as clear bands on the autoradiogram are presented in Fig. 2 (a-c). The binding of non-immunized labeled rabbit IgG is not shown. Densitograms of specific bindings for each sample/lane are presented in Fig. 3 (a-g). The BSM and fully glycosylated MUC1 mucin molecules from standard preparations of both the CA19-9 and the CA15-3 antigen were retained in the stacking gel (Fig. 2a). Densitogram analysis demonstrated specific binding to retained mucins (in stacking gel) in all samples investigated: MUC1 in CA19-9 and CA15-3 samples, BSM, and MUC5B in salivary samples with computed Rf values from 0.023 to 0.037 (Fig. 3, a-g). The greater sensitivity of autoradiography (comparing with Ouchterlony technique and solid phase binding assay previously used) enabled demonstration of anti-BSM IgG reactivity with MUC1 mucin from standard preparation of the CA15-3 antigen, as well as with bands of smaller Mr in all samples that contained CA15-3 antigen. In each of them specific reactions of anti-BSM IgGs with three antigens of 85-120 kD were demonstrated (Fig. 2b). Densitogram analysis revealed identical peaks in all three examined samples with computed Rf of 0.486 (Fig. 3, e-g), which correspond to a band of 85 kD obtained on the autoradiogram (the band in the sample of standard preparation of the CA15-3 antigen is not visible). An additional, probably identical, peak was detected, with Rf of 0.410 (human milk), 0.417 (breast tumor tissue extract), and 0.422 (standard preparation of the CA15-3 antigen) corresponding to a band of 100 kD on the autoradiogram. Peaks with computed Rf values of 0.361 and 0.381 (i.e., bands of 120 kD) were detected in samples of human milk and standard preparation of the CA15-3. In a sample of breast tumor tissue a peak with Rf of 0.456, which corresponds to a band of 95 kD, was obtained. One additional peak, probably identical, was demonstrated in these three samples, with computed Rf of 0.692 (human breast tumor tissue), 0.702 (standard preparation of the CA15-3 antigen), and 0.705 (human milk). The Mr of this position was related to 30 kD on the autoradiogram. Also, densitogram analysis showed peaks with Rf of 0.562 kD (Mr of 45 kD) in samples of human breast tumor tissue and human milk and a peak with Rf of 0.821, only specific in a sample of human breast tumor tissue (Mr < 20 kD). It seems reasonable that antigens that specifically reacted with anti-BSM IgG could be the degradation parts of fully glycosylated mucin. In a sample of commercial BSM, apart from intact mucin, additional small bands of 57 and 36 kD (i.e., peaks with Rf of 0.534 and 0.671) were also detected (Figs. 2c and 3a). Low molecular mass components associated with mucins and detected only under reducing conditions were already observed during the purification of porcine gastric, canine tracheobronchial, as well as human intestinal and tracheobronchial mucins and were described as putative “link” glycoproteins by some investigators [26]. Bands of 85 and 115 kD were detected in a sample of WHS indicating reaction of anti-BSM IgG with both isoforms of the low molecular mass mucin, MG2 (MUC7), and not MG1 (MUC5B) (Fig. 2c). The computed Rf values on the densitogram (Fig. 3b) for these bands were 0.386 and 0.485. Knowing that MUC5B is a human analog of BSM, the result was rather unexpected, but reaction of anti-BSM IgG with MG1 (MUC5B) retained in a stacking gel was demonstrated by densitogram analysis in samples of WHS and of isolated human mucin (Figs. 3b and 3c).Fig. 1. Binding of BSM-HRP (a) and sheep anti-rabbit IgG-AP (b) to isolated anti-BSM IgG. After SDS-PAGE (5-20% gradient gel) and transfer, membranes with immobilized anti-BSM IgG were probed with labeled antigen (BSM-HRP) and antibody (sheep anti-rabbit IgG-AP).
Fig. 2. Binding pattern obtained with anti-BSM rabbit antibodies. Membranes with immobilized antigens were probed with antibodies labeled with iodine 125I and exposed to AGFA CURIX, XP X-ray film, for 6 weeks. Molecular masses of proteins in kD are indicated. a) MUC1 from commercial standard preparations of human CA15-3 antigen and human CA19-9 antigen (Fitzgerald Industrial International, Inc.) and BSM (Sigma) detected in the stacking gel. Mucin from sample of WHS (whole human saliva) and isolated salivary mucin (ISM) were not visible. b) Bands from 85-120 kD detected in samples containing CA15-3 antigen (HM, defatted human milk; BT, human breast tumor tissue; SP, commercial standard preparation of human CA15-3 antigen). c) Salivary antigens detected: bands of 85 and 115 kD in WHS and bands of 57 and 36 kD in BSM.
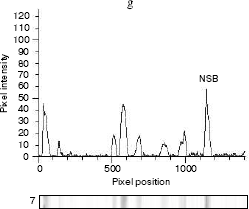
Fig. 3. Densitograms of autoradiogram of specific anti-BSM binding obtained with ImageMaster TotalLab v. 2.01: a) BSM (Sigma); b) WHS (whole human saliva); c) isolated salivary mucin (ISM); d) human CA19-9 antigen (Fitzgerald Industrial International, Inc.); e) human CA15-3 antigen (Fitzgerald Industrial International, Inc.); f) human breast tumor tissue. Nonspecific binding (NSB) was marked according to results and Rf values obtained for densitograms of nonspecific binding. The first peak in each densitogram corresponds to retained mucin in the stacking gel.
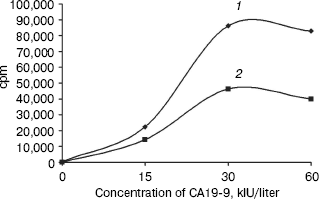
Antibody reactivity - IRMA. Monitoring of quantitative changes in mucin composition is of great value for studying their function as well as structure. The obtained anti-BSM antibodies were used as reagents for development of assays for quantitative mucin determination. The assays were immunoradiometric (IRMAs) with immobilized antibodies on polystyrene star-tubes as a solid phase. As described in methods, anti-BSM IgG was used either as both a catcher and a tracer or as a tracer in combination with immobilized monoclonal anti-CA19-9 antibody, clone M602207. In the first case, there was no reaction with soluble BSM or CA15-3 antigens. Using CA19-9 antigens in both IRMAs a narrow range of linear reactivities was obtained (Fig. 4, a and b). Reactivity of rabbit anti-BSM antibodies with the MUC1 mucin from standard preparation of the CA19-9 antigen was confirmed, apart from double diffusion technique and autoradiography. There are a few possible explanations for non-reactivity of the BSM as an antigen examined in the broad range of concentrations in IRMAs. It could be supposed that an antigenic structure on the BSM that provokes immunization is not abundant on the mucin. Knowing that reactivity of two antibodies in a sandwich assay, especially with mucin as an antigen, is not predictable [27], the more probably is that binding of one antibody with a mucin molecule could cause steric hindrance for binding of the second one.Fig. 3 (Contd.). Densitograms of autoradiogram of specific anti-BSM binding obtained with ImageMaster TotalLab v. 2.01: g) HM, defatted human milk.
Commercial immunometric assay ELSA CA19-9 (CIS Biointernational) was used for testing BSM for the presence of CA19-9 antigen. The result was negative (not shown).Fig. 4. The reaction of anti-BSM antibodies with human CA19-9 antigen in IRMA (cpm versus CA19-9 concentration). Anti-BSM IgG (1) or anti-CA19-9 monoclonal IgG (2) were coated on polystyrene star tubes and reacted with increased concentration of CA19-9. The tracer was anti-BSM IgG labeled with iodine 125I (5*105 cpm/tube) in both cases (1 and 2).
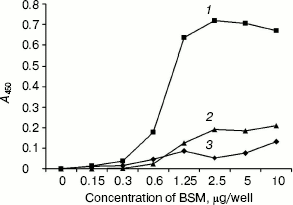
Carbohydrate-sensitivity assay for anti-BSM antibodies - ELISA. Investigation on a carbohydrate antigen that binds to the obtained antibodies was further done with chemically modified BSM in an ELISA, as described in “Materials and Methods”. We used acidic treatment, which was shown to result in the loss of sialic acid residues [25], and periodate oxidation for partial deglycosylation of the BSM. The same concentrations of treated and untreated BSM were coated on microtiter wells and incubated with anti-BSM IgG conjugated with HRP. As shown in Fig. 5, both treatments minimized the reactivity of the antigen with the antibodies. Reduction of reactivity after such chemical modifications that cause partial deglycosylation supported our previous finding. These results indicated not only a carbohydrate nature of the antigenic epitope, but also the fact that the anti-BSM antibodies are sialic acid dependent.
Fig. 5. Effect of acidic and periodate treatment of the BSM on recognition by anti-BSM antibodies: 1) BSM; 2) desialylated BSM; 3) oxidized BSM. The same concentrations of treated and untreated BSM (coated to microtiter wells in the range 0.15-10 µg/well) were reacted with anti-BSM IgG-HRP.
DISCUSSION
Binding of polyclonal rabbit anti-BSM IgG to human salivary mucins and to human mucins known as carcinoma-associated was demonstrated. Reaction of anti-BSM IgG with CA19-9 antigen was obtained under different conditions employing several methods--double immunodiffusion, solid phase binding assay [16], and here described autoradiography and IRMA. The CA19-9 antigen is carbohydrate antigen, sialyl-Lewisa, Neu5Ac(alpha2-3)Gal(beta1-3)(Fuc(alpha1-4))GlcNAc. It is a well known human tumor marker for colon and pancreas, defined by monoclonal antibody 1116NS 19-9 [28]. MUC1 mucin is a carrier of this antigen in human sera from tumor patients, but its presence was also confirmed on both human salivary mucins [14], MG1 and MG2, i.e., MUC5B [29] and MUC7 [15]. MUC1 mucin is also a carrier of the CA15-3 antigen, defined by monoclonal antibodies 115D8 and DF3 and employed as breast tumor marker [30]. Both commercial CA15-3 and CA19-9 antigens used in this investigation contain the MUC1 of tumor origin. The CA15-3 antigen is a less glycosylated form having 50% carbohydrate by weight, while pancreatic MUC1 has 80% carbohydrate [31]. As a source of CA15-3 antigen in this work, beside commercial standard preparation, we used samples of human breast tumor tissue extract and of defatted human milk. Three antigens of 85-120 kD were detected by autoradiography in each of these samples and they could be degradation parts of fully glycosylated mucin. Densitogram analysis further demonstrated that specific binding of anti-BSM IgG with retained MUC1 in three investigated samples was obtained and identified an identical peak with computed Rf of 0.486 in all of them. It correspond to a band of 85 kD obtained on the autoradiogram and it seems reasonable that it could be the same antigen. Another, probably identical, antigen in three examined samples was demonstrated as a clear band of 100 kD on the autoradiogram and as peaks on densitograms with computed Rf of 0.410 (human milk), 0.417 (human breast tumor tissue), and 0.422 (standard preparation of CA15-3 antigen). An antigen of 120 kD was present in samples of human milk and standard preparation of CA15-3 and of 95 kD only in a sample of human breast tumor tissue. Analysis of densitograms also demonstrated specific reaction of anti-BSM IgG with antigens of 45, 30, and <20 kD. Specific binding of anti-BSM IgG with bands of smaller Mr was not detected by autoradiography and densitometry in a sample of commercial standard preparation of CA19-9, the only antigen that reacted in all assays used, whether immobilized or solubilized (in IRMA). Autoradiography showed that intact MUC1 is recognized by polyclonal anti-BSM IgG.
Specific binding to MUC5B, a human analog of BSM, could only be demonstrated by densitogram analysis of samples of WHS and of isolated salivary mucin. Reactions with two isoforms of salivary MUC7 in WHS obtained by autoradiography and densitometry supported an assumption that the epitope for anti-BSM IgG is carbohydrate by nature. The obtained anti-BSM antibodies will be used in further investigation of human salivary mucins taking saliva from more individuals because inter- and intra-individual variations in the composition of salivary mucins has been documented [14].
Carbohydrate specificity of rabbit anti-BSM IgG was demonstrated by chemical modification of antigen (BSM) used for its obtaining. Reduction of reactivity after chemical modifications that cause partial deglycosylation supported our conclusion on a carbohydrate nature of the antigenic site, and it was shown that the anti-BSM antibodies are sialic acid dependent. Negatively charged sialic acid may participate directly in antibody binding, possibly via electrostatic interactions with basic and electropositive amino acids in the antibody binding domain. The other possibility is that terminal sialic acid residues could influence epitope availability. Charge repulsion of adjacent sialic acid residues could improve antibody access even to peptide determinants and removal of sialic acid could lead to steric hindrance of antibody binding [32]. Although our anti-BSM antibodies reacted with standard preparations of human CA19-9 antigen (CIS Biointernational) in several assays and using different methods, we cannot conclude that the binding epitope is sialyl-Lea antigen. First, according to Wu et al. [33], sialyl-Lea structure is not present in the carbohydrate moiety of BSM and the most abundant structures are sialyl Tn, NeuAcalpha2-6GalNAc (53%), and structure NeuAcalpha2-6(GlcNAcbeta1-3)GalNAc (22%), as well as T antigen, Galbeta1,3GalNAc and Tn antigen, GalNAc. Second, our testing of BSM used in the immunization for the presence of the CA19-9 antigen did not show any reactivity. On the contrary, as already mentioned, the presence of the sialyl-Lewisa antigen on both human salivary mucins, MG1 and MG2, was confirmed [14], as well as, for example, of another tumor marker, SPan-1 antigen [34]. The exact epitope of SPan-1 antigen has not yet been fully elucidated and it is supposed to be sialyl-lacto-N-tetraose, a precursor of the CA19-9 antigen. This antigen, i.e., the monoclonal antibody that defined it, could demonstrate the difficulties and obstacles in recognizing the exact nature of an antigen determinant on mucin molecules. As an explanation for the reactivity of standard preparations of CA19-9 antigen (CIS Biointernational) with anti-BSM IgG, we can assume that the MUC1 mucin from these preparations, beside sialyl-Lewisa, contains other sialylated carbohydrate antigen(s) recognized by the obtained antibodies. Such structure(s) was (were) also uncovered under reducing conditions on the MUC1 and antigens from CA15-3 samples. Whether it is the most abundant carbohydrate structure on the BSM, sialyl Tn, remains to be elucidated. According to the literature data, sialyl Tn could be a binding domain for the obtained antibodies because it is present on mucins of salivary as well as of tumor origin, including breast tumor [31, 35, 36].
Results obtained by the investigation on reactivity of polyclonal anti-BSM antibodies suggested that they recognize sialylated carbohydrate epitope, which remains to be defined. This determinant is expressed on mucin antigens of different origin (bovine, human), from different human tissue (saliva as a product of salivary glands secretion, milk from a healthy woman-postpartum, breast tumor), and on different human apomucins (MUC1, MUC7, MUC5B). The specificity as well as immunohistological reactivity of the obtained polyclonal anti-BSM antibodies will be further investigated. The most important is that they could serve as a tool for human mucin investigation, both in qualitative and quantitative methods.
This research was supported by the Ministry of Science and Environmental Protection of the Republic of Serbia, project numbers 1504 and 1500.
REFERENCES
1.Hilkens, J. (1988) Cancer Rev.,
11/12, 25-54.
2.Levi, E., Klimstra, D. S., Adsay, N. V., Andea, A.,
and Basturk, O. (2004) J. Clin. Pathol., 57, 456-462.
3.Golubovic, S., and Cuperlovic, M. (1998) Arch.
Oncol., 6, 131-133.
4.Kim, Y. J., and Varki, A. (1997) Glycoconj.
J., 14, 569-576.
5.Lloyd, K. O., and Kudryashov, V. (1998) Tumor
Biol., 19 (Suppl. 1), 118-121.
6.Galanina, O. E., Simeoni, L. A., Ter-Grigoryan, A.
T., Belyanchikov, I. M., and Bovin, N. V. (1998) Tumor Biol.,
19 (Suppl. 1), 79-87.
7.Magnani, J. L., Steplewski, Z., Koprowski, H., and
Ginsburg, W. (1983) Cancer Res., 43, 5489-5492.
8.www.ISOBM.org/MUC1-TD-4 htm.
9.Price, M. R., Rye, P. D., Finn, I. J., and Hilgers,
J. (1998) Tumor Biol., 19 (Suppl. 1), 1-20.
10.Cao, Y., Karsten, U., and Hilgers, J. (1998)
Tumor Biol., 19 (Suppl. 1), 88-99.
11.Sikut, R., Sikut, A., Zhang, K., Baeckstrom, D.,
and Hansson, G. C. (1998) Tumor Biol., 19 (Suppl. 1),
122-126.
12.Ryoko, K., Schol, D. J., Snijdewint, F. G. M.,
von Mensdorff-Pouilly, S., Poort-Keesom, R. J. J., Karuntu-Wanamarta,
Y. A., Verstraeten, R. A., Miayazaki, K., Kenemans, P., and Hilgers, J.
(2000) Tumor Biol., 21, 197-210.
13.Jiang, W., Gupta, D., Gallagher, D., Davis, S.,
and Bhavanandan, V. P. (2000) Eur. J. Biochem., 267,
2208-2217.
14.Veerman, E. C. I., Valentijn-Benz, M., van den
Keybus, P. A. M., Rahtman, W. M., Sheenan, J. K., and Nieuw Amerongen,
A. V. (1991) Arch. Oral Biol., 36, 923-932.
15.Bolscher, J. G. M., Groenink, J., van der Kwaak,
J. S., van den Keybus, P. A. M., vant Hof, W., Veerman, E. C. I., and
Nieuw Amerongen, A. V. (1999) J. Dent. Res., 78,
1362-1369.
16.Golubovic, S., and Bojic-Trbojevic, Z. (2003) in
Abst. Book, 4th Sci. Meet. of Immunology Society of Yugoslavia,
p. 91.
17.Bojic-Trbojevic, Z. (1998) MA Thesis,
University of Belgrade, Faculty of Chemistry.
18.Hunter, W. M., and Greenwood, F. C. (1962)
Nature, 194, 495-496.
19.Kurstak, E. (1985) Bulletin of WHO,
63, 793-811.
20.Laemmli, U. K. (1970) Nature, 227,
680-685.
21.Beeley, J. A., Sweeney, D., Lindsay, J. C. B.,
Buchanan, M. L., Sarka, L., and Khoo, K. S. (1991)
Electrophoresis, 12, 1032-1041.
22.Bollag, D. M., and Edelstein, S. J. (1991) in
Protein Methods, John Wiley & Sons, Inc., New
York-Chichester-Brisbane-Toronto-Singapore, pp. 181-208.
23.Circolo, A., and Gulati, S. (1996) in The
Protein Protocols Handbook (Walker, J. M., ed.) The Humana Press,
Totowa, NJ, pp. 235-242.
24.Golubovic, S. (1996) M A Thesis,
University of Belgrade, Faculty of Pharmacy.
25.Montreuil, J., Buoquelet, S., Debray, H.,
Fournet, B., Spik, G., and Strecker, G. (1986) in Carbohydrate
Analysis. A Practical Approach (Rickwood, D., and Hames, B.
D., eds.) IRL Press, Oxford-Washington, DC, pp. 143-204.
26.Bergeron, A., La Rue, H., and Fradet, Y. (1997)
Biochem. J., 321, 889-895.
27.Norum, L. F., Varaas, T., Kierulf, B., and
Nustad, K. (1998) Tumor Biol., 19 (Suppl. 1),
134-146.
28.Lamerz, R. (1992) in Serological Cancer
Marker (Sell, S., ed.) The Humana Press, Totowa, NJ, pp.
309-339.
29.Troxler, R. F., Iontcheva, I., Oppenheim, F. G.,
Nunes, D. P., and Offner, G. D. (1997) Glycobiology,
7, 965-973.
30.Hayes, D. F., Tondini, C., and Cufe, D. W. (1992)
in Serological Cancer Markers (Sell, S., ed.) The Humana Press,
Totowa, NJ, pp. 281-307.
31.Ho, J. L., and Kim, Y. S. (1994) Pancreas,
9, 674-691.
32.Dai, J., Allard, W. J., Davis, G., and
Yeung, K. K. (1998) Tumor Biol., 19 (Suppl. 1),
100-110.
33.Wu, A. M., Csako, G., and Herp, A. (1994) Mol.
Cel. Biochem., 137, 39-55.
34.Kawa, S., Tokoo, M., Oguchi, H., Furuta, S.,
Homma, T., Hasegawa, Y., Ogata, H., and Sakata, K. (1994)
Pancreas, 9, 692-697.
35.Therkildsen, M. H., Mandel, U., Thorn, J.,
Christensen, M., and Dabelsteen, E. (1994) J. Histochem.
Cytochem., 42, 1251-1259.
36.Irimura, T., Denda, K., Iida, S.,
Takeuchi, H., and Kato, K. (1999) J. Biochem., 126,
975-985.