Cytokine Profile in Children with Inflammatory Bowel Disease
A. P. Toptygina1,2*, E. L. Semikina3,4, G. V. Bobyleva1, L. V. Miroshkina3, and S. V. Petrichuk3
1Gabrichevsky Institute of Epidemiology and Microbiology, ul. Admirala Makarova 10, 125212 Moscow, Russia; fax: +7 (495) 452-1830; E-mail: toptyginaanna@rambler.ru2Lomonosov Moscow State University, Faculty of Biology, 119991 Moscow, Russia
3Scientific Center of Children Health, Russian Academy of Medical Sciences, 119991 Moscow, Russia
4Pirogov Russian National Research Medical University, 117997 Moscow, Russia
* To whom correspondence should be addressed.
Received July 2, 2014
Crohn’s disease (CD) and ulcerative colitis (UC) are inflammatory bowel diseases (IBD) characterized by immune reactivity against microbial and auto-antigens. This work was designed to study the cytokine profile in blood serum and coproextracts of children with CD and UC. The studied patients consisted of 17 children with CD (group I), 17 children with UC (group II), and 18 controls with intestinal dysbiosis (group III). The diagnosis of UC and CD was based on accepted clinical and endoscopic criteria. The levels of 13 cytokines (IL-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12p70, IL-17A, TNF-α, TGF-β, and IFN-γ) were determined in blood sera and coproextracts of the patients and controls using the BioPlex technology. The level of IL-17A was significantly increased and that of TGF-β was significantly decreased in the blood serum of the patients with IBDs. Changes in the cytokine profile in the coproextracts affected the wider spectrum of cytokines. The levels of proinflammatory cytokines (IL-2, IL-4, IL-6, IL-12p70, TNF-α, and IFN-γ) were increased 6-9-fold, whereas the level of the anti-inflammatory cytokine IL-10 was increased 3-fold. The cytokine balance was shifted to the proinflammatory cytokines. The TGF-β level was increased 9-fold and that of IL-17A was increased 3-fold. Thus, the cytokine profile in the coproextracts was more informative than that of the blood serum. The determination of cytokines in coproextracts is simple and noninvasive.
KEY WORDS: cytokines, interleukin, Crohn’s disease, ulcerative colitis, childrenDOI: 10.1134/S0006297914120116
Abbreviations: CD, Crohn’s disease; IBD, inflammatory bowel disease; Th, T-helpers; UC, ulcerative colitis.
Crohn’s disease (CD) and ulcerative colitis (UC) united as
inflammatory bowel diseases (IBD) are chronic relapsing noninfectious
diseases of the intestinal tract leading to irreversible damage to its
structure and functions [1]. At present, there is
no conventional opinion about the etiology and pathogenesis of IBD. The
intestinal microflora is known to play an important role in regulation
of the mucosal immunity. Constant interactions of the intestinal
epithelium with its normal microflora maintain homeostasis of the
epithelium and protects against pathogenic microorganisms, thus
preventing development of inflammation in the intestine [2]. This delicate balance is disturbed in patients
with IBD, and this is a triggering mechanism responsible for
development of inflammation. Some researchers believe that intestinal
dysbiosis can be an etiological precursor of IBD [3, 4]. In patients with IBD, the
amount of microorganisms of Firmicutes type was decreased [5, 6], as well as that of
Bacteroides [7, 8];
these microorganisms are mainly represented by Faecalibacterium
prausnitzii and Bacteroides fragilis, respectively.
Microorganisms of these two types represent about 90% of the normal
intestinal microflora. In patients with IBD, the total amount of
microorganisms is increased, including potentially pathogenic
Actinobacteria and Proteobacteria, in particular, different
representatives of Escherichia coli [9].
Insufficient functions of the intestinal epithelial barrier caused by genetic disorders in innate immunity mechanisms [10] lead to activation of antigen-presenting cells, which is accompanied by powerful secretion of proinflammatory cytokines and attraction of neutrophils and T- and B-lymphocytes into the inflammatory lesion [11, 12]. It is believed that CD is mediated by T-helper 1 (Th1) because in bioptates of the damaged intestine there is an increased level of Th1-type cytokines, such as interleukin-12 (IL-12), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α) [13, 14]. However, UC was considered as a Th2-type disease because it is often associated with increased levels of such Th2-type cytokines as IL-5 and IL-13, whereas the IL-4 concentration is virtually unchanged [13, 14]. However, recent data suggest that these two nosologies include different subtypes associated with different variants of IBD immunopathogenesis. Some years ago, a special role of Th17- and Treg-lymphocytes was established in pathogenesis of IBD [15]. The level of Th17-lymphocytes was significantly increased in the intestinal wall in patients with CD, which resulted in a hyperproduction of IL-17A, IL-17F, IL-22, IL-21, and IL-26. Cytokines of the IL-17 family have a proinflammatory action and are powerful chemoattractants for neutrophils and inducers of IL-6 and TNF-α synthesis [16]. Proinflammatory cytokines IL-1α, IL-1β, IL-2, -6, -8, -12, -17, -23, IFN-γ, and TNF-α are thought to play an important role in the initiation and progress of IBD. Anti-inflammatory cytokines such as IL-10 and transforming growth factor β (TGF-β) also contribute to pathogenesis of the disease. The domination of one or another type of cytokines is considered a variant of IBD immunopathogenesis [17].
Many authors have reported data on different cytokines in IBD, but they usually studied the cytokine spectra in blood serum and also the production of cytokines by peripheral blood lymphocytes or by lymphocytes from bioptates of the intestine of patients with IBD. However, cytokines in blood serum mainly characterize the systemic immune response of the body to all immune processes in the organism (not only IBD). The production of cytokines by peripheral blood lymphocytes characterizes the potential ability of a studied subject to produce cytokines, but it does not directly reflect the intestine condition. The production of cytokines by lymphocytes from intestine bioptates represents more accurately the inflammation picture in situ, but there are serious limitations for obtaining the material for this study. Difficulties associated with studies of biopsy material from children with IBD are caused by the pronounced invasiveness of colonoscopy and the necessity of general anesthesia. Therefore, such procedure can be performed only about two times per year. Moreover, the choice of the biopsy site is associated with a subjective assessment of the most damaged region of the intestine by the endoscopist, which can significantly influence the reliability of the investigation.
The purpose of this work was to study whether it is possible to determine cytokines in aqueous saline coproextracts for subsequent use of this approach for inflammatory activity assessment in patients with IBD.
MATERIALS AND METHODS
Fifty-two children (29 boys and 23 girls) were examined in ages from 7 to 17 years old. These children included 17 patients with CD (group I), 17 with UC (group II), and 18 were examined because of intestinal dysbiosis (control group III). The diagnosis of CD and UC in children of groups I and II was established based on case history, clinical, laboratory, endoscopic, and histological data. Duration of the disease on examination varied from 1 month to 13 years (4 years on average). In children of group III, the diagnosis was verified based on microbiological study of feces.
Blood samples for determination of cytokines in serum were taken using the BDVacutainer® system. To prepare a aqueous saline coproextract, one volume of feces was mixed with three volumes of phosphate buffered saline and precipitated by centrifugation at 3000 rpm for 15 min. The sample was diluted 1 : 4 for the control group and 1 : 40 for groups I and II, because in patients with IBD the cytokine level is known to be increased. The supernatant was used for determination of cytokine concentrations.
Contents of 13 cytokines (IL-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12p70, IL-17A, TNF-α, TGF-β and IFN-γ) were determined in blood serum and aqueous saline coproextracts with a double-beam laser automatic analyzer (BioPlex Protein Assay System; BioRad, USA) using commercial test systems (measurable dynamical range 0.2-3200 pg/ml) [18].
The results were treated statistically using nonparametric approaches (Mann–Whitney test) and methods of descriptive statistics. The difference was considered significant at p < 0.05. The calculations were performed using Microsoft Office Excel 2003 and Statistica 6.0 programs.
RESULTS
Microbiological study of feces revealed in group III a decrease in the normal flora: in 55.6% of the children, there was a decreased amount of lactobacteria; in 33.3% – of bifidobacteria; and in 27.8% – of E. coli. At the same time, in 44.4% of the children of this group hemolyzing E. coli was found, in 27.7% – Klebsiella, and in 33.3% – Staphylococcus aureus.
The cytokine distribution in blood sera is presented in Table 1. The level of IL-17A was significantly increased and of TGF-β significantly decreased (p < 0.05) in groups I and II as compared to the control group. Contents of other studied cytokines were similar in the children of the three groups.
Table 1. Contents of cytokines in blood
serum of studied children, pg/ml Me(LQ-HQ)
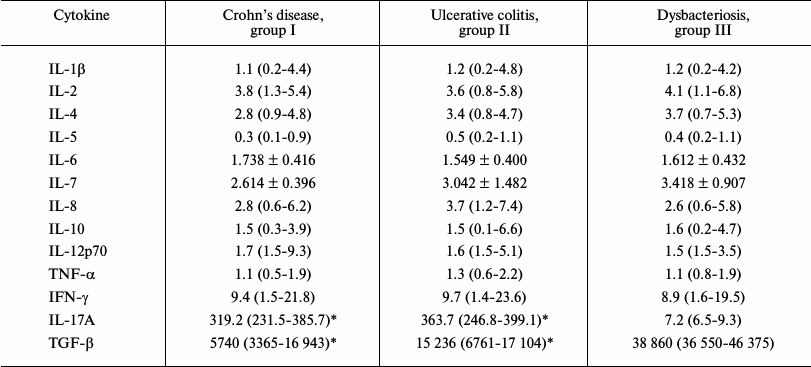
* p < 0.05 as compared to the group III.
No IL-7 was found in the aqueous saline coproextracts. IL-8 was found only in three children of group I and in three children of group II, but the level of this cytokine in these children was higher than 800 pg/ml. The IL-5 concentration was at the lower limit of determination in the majority of the children, but in some the level of this cytokine was high (200-900 pg/ml) independently of diagnosis. This might be caused by a concomitant sensibilization to nutritional antigens in these children. All of the other studied cytokines were found in all coproextracts of the children as shown in Table 2. Table 2 shows that the levels of cytokines in the coproextracts of the children of groups I and II were significantly higher (p < 0.05) than in the coproextracts of the control group children. Moreover, the levels of such cytokines as IL-2, IL-4, IL-6, IL-12p70, TNF-α, and IFN-γ were increased 6-9-fold. However, the level of the anti-inflammatory cytokine IL-10 was 2-3-fold higher than in the control group, and the level of TGF-β, which is very important for regulation of intestinal homeostasis, was increased 9-fold. The level of IL-17A was increased 3-fold in groups I and II as compared to group III (p < 0.05).
Table 2. Contents of cytokines in
coproextracts of the studied children, pg/ml Me(LQ-HQ)
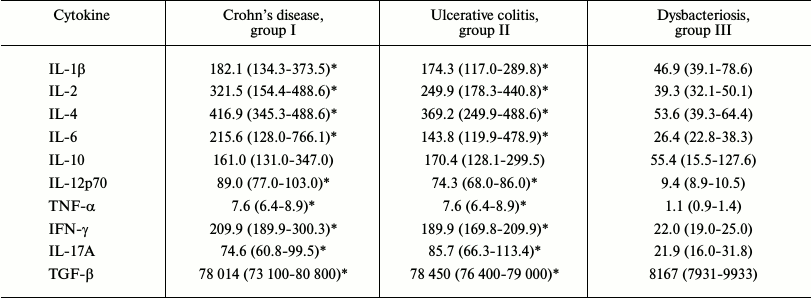
* p < 0.05 as compared to group III.
In the blood serum of the children with IBD, only the levels of IL-17A and TGF-β were significantly different from their values in group III; therefore, correlation coefficients were calculated for concentrations of these cytokines in blood serum and coproextracts. A direct moderate correlation was found for IL-17A (R = 0.465) and an inverse moderate correlation was found for TGF-β (R = –0.389).
DISCUSSION
The levels of 11 cytokines in blood serum of three groups of studied children were similar and virtually did not differ from the cytokine levels in blood serum of healthy children [19]. However, the IL-17A level in blood serum of the children with IBD was 40-50 times higher than in the control group. This cytokine is known for its ability to attract neutrophils and monocytes into the zone of chronic inflammation [20]. Experimental colitis could not be obtained in mice with IL-17A–/– gene knockout, which suggested an important role of this cytokine in development of intestinal inflammation [21]. Moreover, in blood serum of the children with IBD the level of such important immunoregulatory cytokine as TGF-β was significantly decreased. It was shown on mouse models that the absence of this cytokine signaling led to inflammation in the intestine [22]. No significant difference was found in the cytokine profiles from the children with different nosology of IBD. This could be associated with heterogeneity of the groups in the phenotype of disease and in the inflammation localization that could correspond to different pathways of IBD immunopathogenesis. Earlier we showed that children with IBD were very different in levels of proinflammatory cytokines in blood serum, from very high to virtually undeterminable. Moreover, the result of biological therapy of a patient strongly depended on the initial level of these cytokines [23].
Changes in the cytokine profiles of the coproextracts were more pronounced than in those of blood serum. The levels of proinflammatory cytokines, such as IL-1β, IL-6, and TNF-α were 9- and more times higher in children with IBD than in the control group. Earlier we showed that no proinflammatory cytokines were found in coproextracts of healthy children [24]. In total, these results seem to indicate that, although intestinal dysbiosis had symptoms similar to beginning of IBD (pains in the intestine, meteorism, stool disturbance, and even appearance of blood in feces), the inflammatory process in dysbiosis is significantly lower. It may be that intestinal dysbiosis can provoke the beginning of IBD, but it seems that for realization of IBD a genetic predisposition and some environmental factors are needed for its triggering.
The levels of Th1 cytokines (IL-2, IL-12p70, and IFN-γ) were 8-9-fold increased in the patients with IBD. The levels of these cytokines are known to correlate with the severity of inflammation in IBD [25]. The level of IL-4 was increased 7-fold, whereas the level of IL-17A was only 3-fold higher than in the control group. It is known that just the Th1/Th17 type of immune response is most frequent in CD, whereas for UC the Th2 type is more specific [26]. Nevertheless, we did not find differences in the levels of the corresponding cytokine markers of the main types of immune response in coproextracts of the children with CD and UC. The levels of both pro- and anti-inflammatory cytokines were increased in coproextracts of the children from groups I and II. However, the levels of proinflammatory cytokines were 8-9-fold higher than in the control group, whereas the levels of anti-inflammatory cytokines were only 3-fold higher, thus, we can conclude that the cytokine balance was shifted to proinflammatory cytokines.
Ratios of changes in the concentrations of IL-17A and TGF-β in blood serum and coproextracts were opposite. In blood serum of the patients with IBD, the IL-17A levels were increased 40-50-fold and the levels of TGF-β were decreased 3.5-4-fold, whereas in the coproextracts of these patients the level of IL-17A was increased 3-4-fold and the level of TGF-β was increased more than 9-fold. Note that the correlation was direct for IL-17A and inverse for TGF-β. The discordance of changes in the general and local concentrations of cytokines is probably associated with the functional activity of these mediators. These findings seem to indicate that for IL-17A a parallelism can exist between local inflammation and reaction on the organism’s level. In the case of TGF-β, this seems to mean that a local decrease in the regulatory functions of this cytokine can be associated with triggering of a central regulatory mechanism as an attempt of the organism to suppress the autoimmune process.
No IL-7 was found in the aqueous saline coproextracts. It was shown recently that this cytokine, in addition to supporting homeostasis of the T-lymphocyte pool, also stimulated intestinal epithelium cells. In mice, this cytokine was shown to prevent damage of the intestinal epithelium in experimental colitis, but the affinity of receptors for IL-7 on the epithelial cells was significantly lower than on T-lymphocytes. Protective properties of IL-7 manifest themselves only under conditions of severe lymphopenia [27]. However, in CD and UC, T-lymphocytes are actively attracted into the inflammation zone and seem to capture residual IL-7, which prevents the repair of the epithelium.
Thus, the present work has shown that levels of cytokines can be measured in coproextracts of children with IBD using a multiplex technology. Changes in the cytokine profile in coproextracts of children with IBD as compared to intestinal dysbiosis were even more pronounced than in blood serum. Moreover, the determination of cytokines in coproextracts is noninvasive and easily reproducible. We have not found significant difference between children with different nosological forms of IBD. This finding correlates with the concept of the existence of numerous subtypes of IBD that are different in the leading immunopathogenetic mechanism and correspond to different pathologies [14, 17, 28]. According to this concept, IBD are to be differentiated by the depth and degree of the intestinal damage and by the presence of extra-intestinal manifestations. The absence of differences in the cytokine profiles of the patients with CD and UC can also be explained by heterogeneity of the studied groups in other features: the duration and activity of the disease, therapy, surgical history, etc. It seems that an increase in the number of observations can result in more uniform groups and reveal differences between IBD types with different immunopathogenesis.
REFERENCES
1.Yablokova, E. A., Gorelov, A. V., Ratnikova, M. A.,
Sichinava, I. V., Grammatopulo, M. I., Polotnyanko, E. Yu., and
Borisova, E. V. (2006) Inflammatory bowel disease in children,
Pediatriya, 5, 99-102.
2.Abraham, C., and Medzhitov, R. (2011) Interactions
between the host innate immune system and microbes in inflammatory
bowel disease, Gastroenterology, 140, 1729-1737.
3.Hansen, R., Thomson, J. M., El-Omar, E. M., and
Hold, G. L. (2010) The role of infection in the aetiology of
inflammatory bowel disease, J. Gastroenterol., 45,
266-276.
4.Kaur, N., Chen, C. C., Luther, J., and Kao, J. Y.
(2011) Intestinal dysbiosis in inflammatory bowel disease, Gut
Microbes, 2, 211-216.
5.Frank, D. N., St. Amand, A. L., Feldman, R. A.,
Boedeker, E. C., Harpaz, N., and Pace, N. R. (2007)
Molecular-phylogenetic characterization of microbial community
imbalances in human inflammatory bowel diseases, Proc. Natl. Acad.
Sci. USA, 104, 13780-13785.
6.Sokol, H., Seksik, P., Rigottier-Gois, L., Lay, C.,
Lepage, P., Podglajen, I., Marteau, P., and Dore, J. (2006)
Specificities of the fecal microbiota in inflammatory bowel disease,
Inflamm. Bowel Dis., 12, 106-111.
7.Neut, C., Bulois, P., Desreumaux, P., Membre, J.
M., Lederman, E., Gambiez, L., Cortot, A., Quandalle, P., van
Kruiningen, H., and Colombel, J. F. (2002) Changes in the bacterial
flora of the neoterminal ileum after ileocolonic resection for
Crohn’s disease, Am. J. Gastroenterol., 97,
939-946.
8.Andoh, A., Kuzuoka, H., Tsujikawa, T., Nakamura,
S., Hirai, F., Suzuki, Y., Matsui, T., Fujiyama, Y., and Matsumoto, T.
(2012) Multicenter analysis of fecal microbiota profiles in Japanese
patients with Crohn’s disease, J. Gastroenterol.,
47, 1298-1307.
9.Mukhopadhya, I., Hansen, R., El-Omar, E. M., and
Hold, G. L. (2012) IBD-what role do Proteobacteria play? Nat. Rev.
Gastroenterol. Hepatol., 9, 219-230.
10.Simms, L. A., Doecke, J. D., Walsh, M. D., Huang,
N., Fowler, E. V., and Radford-Smith, G. L. (2008) Reduced
alpha-defensin expression is associated with inflammation and not NOD2
mutation status in ileal Crohn’s disease, Gut, 57,
903-910.
11.Abraham, C., and Cho, J. H. (2009) Inflammatory
bowel disease, N. Engl. J. Med., 361, 2066-2078.
12.Sanchez-Munoz, F., Dominguez-Lopez, A., and
Yamamoto-Furusho, J. K. (2008) Role of cytokines in inflammatory bowel
disease, World J. Gastroenterol., 14, 4280-4288.
13.Fuss, I. J., Neurath, M., Boirivant, M., Klein,
J. S., de la Motte, C., Strong, S. A., Fiocchi, C., and Strober, W.
(1996) Disparate CD4+ lamina propria (LP) lymphokine secretion profiles
in inflammatory bowel disease. Crohn’s disease LP cells manifest
increased secretion of IFN-gamma, whereas ulcerative colitis LP cells
manifest increased secretion of IL-5, J. Immunol., 157,
1261-1270.
14.Bamias, G., Nyce, M. R., De La Rue, S. A., and
Cominelli, F. (2005) New concepts in the pathophysiology of
inflammatory bowel disease, Ann. Intern. Med., 143,
895-904.
15.Elgbratt, K., Kurlberg, G., Hahn-Zohric, M., and
Hultgren Hornquist, E. (2010) Rapid migration of thymic emigrants to
the colonic mucosa in ulcerative colitis patients, Clin. Exp.
Immunol., 162, 325-336.
16.Fina, D., and Pallone, F. (2008) What is the role
of cytokines and chemokines in IBD? Inflamm. Bowel Dis.,
14, 117-118.
17.Muzes, G., Molnar, B., Tulassay, Z., and Sipos,
F. (2012) Changes of the cytokine profile in inflammatory bowel
diseases, World J. Gastroenterol., 18, 5848-5861.
18.Ostanin, A. A., and Chernykh, E. P. (2005)
Comparative assessment of the level of 17 cytokines in serum and blood
of healthy donors using flow fluorimetry, Tsitokiny Vospalenie,
4, 25-32.
19.Toptygina, A. P., and Aleshkin, V. A. (2011)
Production of cytokines in intact children and children vaccinated with
the Priorix vaccine in response to stimulation by measles and rubella
virus antigens, Immunologiya, 32, 200-205.
20.Shen, W., and Durum, S. K. (2010) Synergy of
IL-23 and Th17 cytokines: new light on inflammatory bowel disease,
Neurochem. Res., 35, 940-946.
21.Ito, R., Kita M., Shin-Ya, M., Kishida, T.,
Urano, A., Takada, R., Sakagami, J., Imanishi, J., Iwakura, Y.,
Okanoue, T., Yoshikawa, T., Kataoka, K., and Mazda, O. (2008)
Involvement of IL-17A in the pathogenesis of DSS-induced colitis in
mice, Biochem. Biophys. Res. Commun., 377, 12-16.
22.Fahlen, L., Read, S., Gorelik, L., Hurst, S. D.,
Coffman, R. L., Flavell, R. A., and Powrie, F. (2005) T cells that
cannot respond to TGF-beta escape control by CD4(+)CD25(+) regulatory T
cells, J. Exp. Med., 201, 737-746.
23.Miroshkina, L. V., Petrichuk, S. V., Tsimbalova,
E. G., Potapov, A. S., Venediktova, M. M., Toptygina, A. P., Kurbatova,
O. V., Izmailova, T. D., Semikina, E. L., and Varichkina, M. A. (2013)
Cytokine profile in children with inflammatory bowel diseases as a
predictor of efficiency of therapy with infliximab, Vopr. Diagnost.
Pediatr., 5, 33-38.
24.Toptygina, A. P., Bobyleva, G. V., and Aleshkin,
V. A. (2008) Correlation between mucosal and systemic immune responses
in infants, Ros. Immunol. Zh., 2, 449-453.
25.Nielsen, O. H., Kirman, I., Rudiger, N., Hendel,
J., and Vainer, B. (2003) Upregulation of interleukin-12 and -17 in
active inflammatory bowel disease, Scand. J. Gastroenterol.,
38, 180-185.
26.Sakuraba, A., Sato, T., Kamada, N., Kitazume, M.,
Sugita, A., and Hibi, T. (2009) Th1/Th17 immune response is induced by
mesenteric lymph node dendritic cells in Crohn’s disease,
Gastroenterology, 137, 1736-1745.
27.Shalapour, S., Deiser, K., Kuhl, A. A., Glauben,
R., Krug, S. M., Fischer, A., Sercan, O., Chappaz, S., Bereswill, S.,
Heimesaat, M. M., Loddenkemper, C., Fromm, M., Finke, D., Hammerling,
G. J., Arnold, B., Siegmund, B., and Schuler, T. (2012) Interleukin-7
links T lymphocyte and intestinal epithelial cell homeostasis, PLoS
One, 7, e31939.
28.Xavier, R. J., and Podolsky, D. K. (2007)
Unravelling the pathogenesis of inflammatory bowel disease,
Nature, 448, 427-434.