Novel Mitochondrial Cationic Uncoupler C4R1 Is an Effective Treatment for Combating Obesity in Mice
A. V. Kalinovich1,2* and I. G. Shabalina1,2
1Department of Molecular Biosciences, Wenner-Gren Institute, Stockholm, Sweden; fax: +46 (8) 164209; E-mail: anastasia.kalinovich@su.se; irina.shabalina@su.se2Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received January 19, 2015; Revision received January 30, 2015
Obesity is associated with premature mortality, impaired quality of life, and large healthcare costs. However, treatment options remain quite limited. Here we studied potential anti-obesity effects of a novel cationic mitochondrial uncoupler, C4R1 (derivative of rhodamine 19) in C57Bl/6 mice. Obesity was induced by long-term (eight weeks) high fat diet feeding at thermoneutrality. The treated group of mice received consecutively two doses of C4R1 in drinking water (30 and 12-14 µmol/kg daily) during 30 days. Effects of C4R1 were dose-dependent. After six days of C4R1 treatment at dose 30 µmol/kg daily, food intake was reduced by 68%, body weight by 19%, and fat mass by 21%. Body weight decrease was explained partly by reduced food intake and partly by increased metabolism, likely resulting from uncoupling. Body fat reduction upon C4R1 treatment was associated with improved lipid utilization estimated from decrease in respiratory quotient to the minimal level (0.7). Interestingly, the classical uncoupler 2,4-dinitrophenol at similar dose (27 µmol/kg daily) did not have any effect. Our results are relevant to the search for substances causing mild uncoupling of mitochondria that could be a promising therapeutic strategy to treat obesity.
KEY WORDS: cationic mitochondrial uncoupler, 2,4-dinitrophenol, obesity, resting metabolic rate, energy expenditureDOI: 10.1134/S0006297915050156
Abbreviations: C4R1, a short-chain alkyl derivative of rhodamine 19; DNP, 2,4-dinitrophenol; MRI, magnetic resonance imaging; RMR, resting metabolic rate; RQ, respiratory quotient; UCP1, uncoupling protein 1.
The number of obese people has been increasing over recent decades, and
now more than one billion adults and children are overweight. Obesity
has a crucial role in developing many diseases such as diabetes
mellitus, hyperlipidemia, cardiovascular diseases, and cancer [1, 2]. Obesity can be overcome by
increase in energy expenditure in the form of increased physical
activity or heat production (thermogenesis) or by reduction of energy
intake from diet. Weight loss induced by dieting has been shown to be
successful in reducing body weight and the health consequences of
obesity, but unfortunately >90% of individuals who lose weight
through dietary control eventually return to their original weight [2, 3]. Pharmacological treatment
may therefore be desirable for those patients with associated comorbid
conditions who have been unable to control their obesity through diet
and exercise.
Mitochondrial uncoupling agents were proposed to be a potential treatment for obesity [1, 4, 5]. The artificial uncoupler 2,4-dinitrophenol (DNP) was used for this purpose for many years in the 1930s [6, 7], but then was discontinued due to high toxicity. DNP is a weak acid, which acts as a protonophore: it crosses the inner mitochondrial membrane in neutral protonated state, loses its proton, and returns as the anion driven by membrane potential. In this way, it increases the proton conductance of mitochondria and energy expenditure of the whole organism [8-10].
DNP is now extensively studied in mice as a model compound to elucidate the effects of mitochondrial uncoupling on animal energy homeostasis, mitochondrial adaptations, mechanisms of oxidative stress, and aging [9-13]. Long-term treatment with a very low dose of DNP reduced body weight, improved serological glucose, triglyceride, and insulin levels, as well as extended the life span of mice [11]. Another study confirmed the positive effects of DNP on body weight and metabolic parameters of mice but reported a negative effect on exercise capacity [9]. In experiments with isolated heart, a low concentration of DNP caused profound QT shortening on the electrocardiogram and triggered ventricular fibrillation [14]. Such inappropriate activities in critical tissues in combination with a narrow therapeutic range (small difference between the effective and the fatal doses) of DNP [6, 15] excluded its therapeutic application against obesity [16].
Alternative cationic uncoupling agents [17, 18], including derivatives of rhodamine 19 [19, 20], have been recently designed. A positive charge on these compounds moves them into mitochondria. Moreover, their uncoupling activity linearly depends on membrane potential, thus, to be self-regulating [21]. Due to the self-regulation as well as an ability to accumulate specifically in mitochondria and probable low toxicity, novel cationic uncouplers could be considered as promising anti-obesity agents.
A long-chain alkyl derivative of rhodamine, C12R1, increases proton conductance of artificial bilayer lipid membranes as well as possessing uncoupling activity in mitochondria and intact cells [19, 20]. A short-chain alkyl derivative of rhodamine 19, C4R1, exhibits low protonophoric activity in artificial bilayer lipid membranes; however, it uncouples mitochondria even more effectively than C12R1 did [20].
The aim of the present investigation was to study effects of the novel cationic uncoupler C4R1 in vivo in mice. To humanize our research, it was performed in C57Bl/6 mice prone to obesity, the animals being kept on high fat diet and at thermoneutrality (30°C). It is well known that mice living at low ambient temperatures produce a lot of heat to maintain their body temperature [22, 23], and this facultative thermogenesis masks uncoupling effect of studied compounds [10, 24]. Quite often anti-obesity drugs decrease food intake. Ignoring this fact leads to overestimation of the role of increased energy expenditure in loss of weight [25, 26]. To distinguish food intake-dependent and independent effects of C4R1, a pair-fed group of mice was included in the study. Another task of our study was to compare effects of C4R1 with effects of the classical uncoupler DNP. DNP is known to uncouple mitochondria at 10 times higher concentration than C4R1 ([20] and [27] for comparison). However, data about DNP doses effective in vivo are quite contradictory (effective doses differ 1000 times in [11] and [9]). In our work, we studied effects of DNP at a dose similar to the C4R1 dose.
MATERIALS AND METHODS
Animals housing. Thirty male C57Bl/6J mice, 7-8-week-old in the beginning of the experiment, were used. The mice were kept at thermoneutrality (30°C) and on high fat diet (45% fat content; D12451; Research Diets Inc., USA) eight weeks prior to the experiment to induce obesity and during the experiment. The mice were housed under 12 h light/12 h dark regime. The light period started at 8 a.m., and the dark period started at 8 p.m. The mice were single caged. Cages were enriched with tissue paper, tissue tube as a shelter, and sawdust for welfare. The entire experiment was approved by the ethical committee of North Stockholm.
C4R1 and DNP treatments and monitoring of physiological parameters. Prior to treatment, the mice were divided into three groups according to their body weight and fat mass: 5 control, 5 pair-fed, and 5 treated mice. Treated mice received food ad libitum and C4R1 in drinking water. C4R1 was initially dissolved in 95% ethanol to concentration 0.25 M and then added in estimated amount (according to body weight of mice) to 30 ml tap water in small drinking bottles. The concentration of the drug in the drinking water varied during the experiment: 0.39-0.41 mM (depending on body weight) for the first six days, 0.19-0.21 mM for the next 16 days. On the 22nd day, the experiment was discontinued for six days and then started again with 0.19-0.21 mM C4R1 for 8 days. The control group received food ad libitum and drinking water with corresponding concentration of sodium bromide and ethanol (0.08-0.16%), since C4R1 was a bromide initially dissolved in ethanol. Pair-fed mice received drinking water with the same supplementations as the control group and the same amount of food as treated mice ate in the previous 1-2 days.
The water was changed every 4-5 days. Water and food intakes were measured every 1-2 days. Actual dose of C4R1 (in µmol/kg daily) was calculated based on water intake. The pair-fed group received a daily meal every day at 8 p.m., when active night period starts, to avoid disturbing circadian rhythms of the animals. For this reason, water and food intakes as well as body weight were measured also at 8 p.m. Body composition (fat and lean masses) was measured twice before the treatment as well as directly prior to the treatment (day 0) and on the 2nd, 6th, and 15th days of the treatment using in vivo magnetic resonance imaging (MRI) with an EchoMRI-100 TM instrument (EchoMRI, USA). Note that fat mass measured by MRI is a much broader concept than mass of adipose tissues. It is sum of all lipids in all tissues of the body. Similar to this, lean mass is a broader concept than mass of muscles and includes proteins and carbohydrates of all tissues in the body, cytoplasm of all cells and extracellular fluid, blood plasma (except for lipids of plasma), etc. In other words, lean mass is the body mass with the exception of all lipids.
The experiment with DNP was performed separately during eight days. Groups of mice were formed similarly to the C4R1 experiment. DNP was initially dissolved in 95% ethanol to concentration 0.5 M and then diluted in 30 ml tap water to concentration 0.39-0.41 mM (according to body weight of the mice). Bottles with water containing DNP were protected from light. Control and pair-fed mice received ethanol (0.07-0.1%) in tap water. Other conditions were like in the C4R1 experiment.
Indirect calorimetry. In the C4R1 experiment during the first two days and on the 7th and the 16th days of treatment, the mice were placed in metabolic chambers (Somedic INCA, Sweden) for measurement of gas exchange by means of indirect calorimetry. Mice from the DNP experiment were placed in metabolic chambers on the 7th day of the treatment. Before measurements, an oxygen analyzer (zirconium oxide detector) was calibrated using two reference gases containing 18 and 25% oxygen in nitrogen. Carbon dioxide was measured using a sensor based on the dual-channel infrared absorption principle (model 0633-1240; Testo, Germany) connected to a reference instrument (model 650; Testo). The carbon dioxide detector was calibrated using two reference gases containing carbon dioxide 0.02 and 0.2% in nitrogen. The metabolic chamber (volume 4 liters) was supplied with air at a flow rate of 1 liter/min. The air leaving the chamber was dried with silica gel (2 mm molecular sieves with pores size 0.3 nm; Merck, Germany). The oxygen and carbon dioxide contents were measured every 2 min in the air supplying the chamber and in the air leaving it. Oxygen consumption and carbon dioxide production were calculated continuously as the difference in the concentrations of the gases in these two samples. Resting metabolic rate (RMR) was estimated as the lowest oxygen consumption rate for a stable 10-15 min period during the day. The respiratory quotient (RQ) value was calculated as ratio of rate of CO2 production to the rate of O2 consumption. Energy expenditure was calculated using the Weir equation and normalized to lean mass:
Energy expenditure (mW/g lean) = (16.3 × VO2 + 4.57 × VCO2) × 1000/(60 × lean),
where VO2 is rate of oxygen consumption, ml/min; VCO2 is rate of carbon dioxide production, ml/min.
Statistics. All data are presented as means ± standard errors of means. The results were analyzed in the KaleidaGraph program (version 4.5.2). Unpaired Student’s t-test with unequal variances was applied to calculate significance with p < 0.05 or p < 0.01.
RESULTS
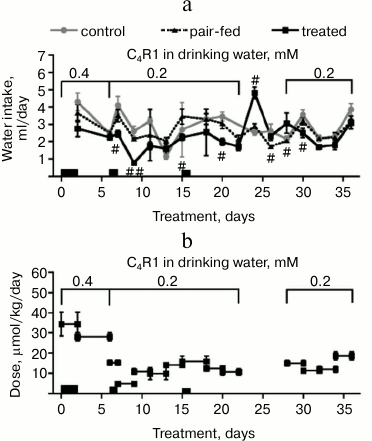
Time course of food intake and body weight. C4R1-treated mice drank somewhat less water during the treatment than control and pair-fed mice (Fig. 1a). Reduction of water intake under C4R1 treatment cannot be explained by the influence of C4R1 on food intake as water intake of pair-fed mice was not different from the control mice. The possibility that C4R1 has an adverse effect on the mice’s sense of taste should be checked by means of preference test. Actual dose of C4R1 received by the mice was estimated from water intake (Fig. 1b).
Fig. 1. Water intake (a) and actual C4R1 dose obtained by mice (b). a) Water intake was measured every 2-3 days. Every data point on the graph represents a water intake (ml/day) during the previous period of measurement; b) actual dose of C4R1 obtained by mice (µmol/kg daily) was estimated based on water intake (see Fig. 1a). Three groups of mice are designated as control, pair-fed, and treated. The x-axis is time relative to start of the first treatment. Periods in metabolic chambers are indicated by black rectangles above the x-axis, n = 5 in each group. Data points are means ± S.E.; #, ## indicate statistically significant differences between treated and pair-fed groups with p < 0.05 and p < 0.01, respectively. For convenience, statistical differences only between treated and pair-fed groups are presented.
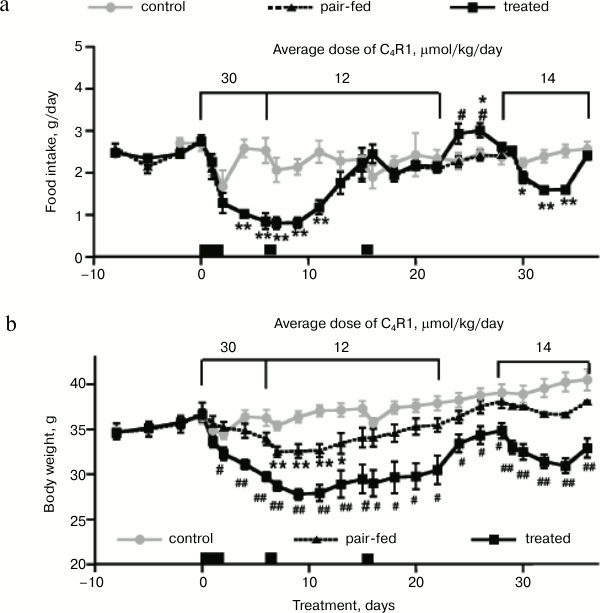
Food intake and body weight are shown in Fig. 2. It should be noted that study of the C4R1 effect on appetite was not a task of the present work. During the first six days, average actual dose of C4R1 was 30 µmol/kg daily (Fig. 1b). Treatment started at 8 p.m. of the first day (time 0 on the figures), when mice started receiving drinking water supplemented with C4R1 or sodium bromide and ethanol. At the same time, mice were placed in metabolic chambers for two days. The new environment caused stress for mice and led to a small reduction in food intake and body weight in all groups. Food intake was not significantly different between the groups; however, treated mice lost significantly more body weight than pair-fed and control mice.
Fig. 2. Effects of C4R1 on food intake (a) and body weight of mice (b). a) Food intake was measured every 2-3 days, as in Fig. 1a; b) body weight. Conditions are as in Fig. 1. *, ** indicate statistically significant differences with control group; #, ## statistically significant differences with pair-fed group with p < 0.05 (one symbol) and p < 0.01 (two symbols).
On days 2-6, mice were removed from the metabolic chambers. Food intake and body weight of control mice were restored. Food intake of treated mice (and correspondingly pair-fed mice) continued decreasing, as well as body weight. During the four days, treated mice lost twice more weight than pair-fed mice (2.54 and 1.24 g, respectively).
On day 6, food intake of most of the treated mice went down to 0.5-0.8 g/day (which is 20-30% of initial food intake) and was not stabilized. According to the ethical committee, this food intake reduction was considered to be critical, and to prevent excessive impact on the well-being of the mice, the concentration of the drug in their drinking water was decreased twice. On days 7-22, average actual dose of C4R1 was 12 µmol/kg daily. At 8 p.m. of the 7th day simultaneously with reduction of C4R1 concentration, mice were placed in metabolic chambers for 24 h.
Again, similarly to the first two days in metabolic chambers, food intake and body weight of control mice were slightly reduced. The treated mice continued to reduce food intake and body weight without visible acceleration caused by the metabolic chambers. Pair-fed mice lost a little body weight due to influence of the metabolic chamber environment, similarly to control mice.
On days 6-9 of C4R1 treatment, food intake of treated mice was stabilized at the minimal level. Body weight of pair-fed mice was stabilized in parallel to food intake stabilization. In contrast to pair-fed mice, treated mice continued loosing body weight during these three days.
On days 9-15, food intake of treated mice started to spontaneously restore in spite of continuous treatment. Parallel to food intake, body weight of treated and pair-fed mice was first stabilized at minimal levels and then started to increase. At this point of the experiment, pair-fed mice lost 11% of body weight, whereas treated mice lost 24% of their body weight.
On day 15 of C4R1 treatment, when food intake of treated mice was fully restored, mice were placed in the metabolic chambers for 24 h.
On days 15-22 of C4R1 treatment, food intake of treated mice was stabilized at the level of control mice. However, body weight of treated mice was still significantly lower than body weight of control and pair-fed mice.
On day 22, C4R1 treatment was discontinued for six days and mice received regular drinking water. Immediately after the treatment cancellation, food intake of treated mice increased and was 35% higher in comparison to control mice eating ad libitum. After four days, food intake of treated mice was back to normal values. During these days body weight of treated mice increased by 14%. Such a quick recovery of appetite and body weight gain of treated mice points to an absence of irreversible toxic effects of C4R1 on the digestive tract and other organs. Interestingly, pair-fed mice, that continued to receive the same amount of food as treated mice ate, did not increase food intake and ate the same amount of food as control mice.
On the 28th day of the experiment, after 6 days of the “recovery” period, the new round of treatment started (average actual dose of C4R1 was 14 µmol/kg daily). The second treatment was performed to check whether the same effect of the drug could be achieved repeatedly after recover. As shown in Fig. 2, food intake and body weight were repeatedly decreased and then spontaneously restored. However, effects were twice smaller than before – the first six days of the first treatment (average actual dose 30 µmol/kg daily) resulted in 68% decrease of food intake and 19% (6.8 g) of body weight loss, whereas the first six days of the second treatment (average actual dose 14 µmol/kg daily) resulted in 38% decrease of food intake and 11% (3.8 g) decrease in body weight. Thus, first, C4R1 influences appetite and body weight repeatedly even after spontaneous recovery of both parameters and, second, it acts in dose-dependent manner.
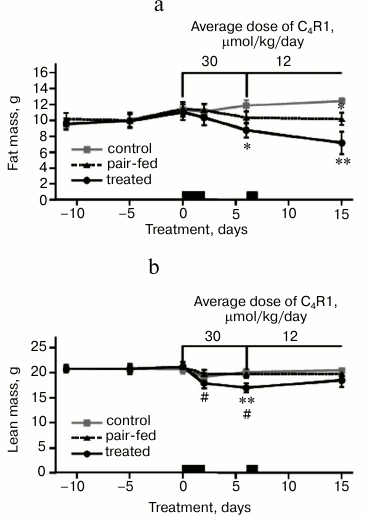
Time course of body composition. Body composition was measured before treatment and during treatment (Fig. 3). Body weight loss by treated and pair-fed mice was accompanied by fat loss (Fig. 3a). During 15 days of the treatment, pair-fed mice lost three times less body fat than treated mice.
Fig. 3. Effect of C4R1 on body composition: body fat mass (a) and body lean mass (b). Body composition was measured by means of MRI. Mice were in metabolic chambers during the 1st-2nd and the 7th day of treatment as indicated by black rectangles above the x-axes, n = 5 in each group. Data points are means ± S.E.; *, ** indicate statistically significant differences with control group; # statistically significant differences with pair-fed group with p < 0.05.
C4R1 caused loss of lean mass as well (Fig. 3b), more pronounced in comparison to the pair-fed group. Note that the difference might result from higher loss of body water, which accompanied higher loss of body weight, and not necessarily from actual loss of proteins and carbohydrates. At day 15, lean mass of treated mice was restored.
Time course of metabolic parameters. Oxygen consumption is a main metabolic parameter reflecting rate of metabolism and energy expenditure. During nighttime, mice are active with the maximal oxygen consumption rate. During daytime, mice mostly sleep and consume much less oxygen. Resting metabolic rate (RMR) was calculated as the minimal stable (during 10-15 min) oxygen consumption rate during the daytime. RMR was the same in three groups of mice during the first two days of the treatment (not shown).
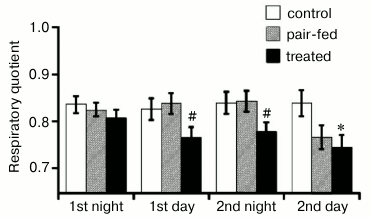
Respiratory quotient (RQ) is another important metabolic parameter, which reflects the source of fuel utilized (proteins, lipids, or carbohydrates). RQ is calculated as a ratio of carbon dioxide production rate to oxygen consumption rate. By definition, RQ could vary between 1 (utilization of pure carbohydrates) and 0.7 (utilization of pure lipids). Mean values of RQ during the first two days of the treatment (separately for night and day periods) are shown in Fig. 4. The RQ of the control mice was typical for high fat diet (around 0.80-0.85 independently on time of day). RQ of pair-fed mice did not differ from RQ of control group during the first day of the treatment. Only in the end of the second day, when food intake of pair-fed mice was somewhat decreased, RQ values dropped. Interestingly, RQ values of treated mice started to decrease already after a few hours of the treatment. The difference was significant already at the first day of the treatment, when food intake did not differ between groups. These data are consistent with larger body weight loss by treated mice during this period of time (Fig. 2b) and are probably important for understanding of food-independent effects of C4R1.
Fig. 4. Respiratory quotient (RQ) during two first days of the treatment. Mean RQ values are presented (n = 5 for each group of mice); * indicates statistically significant difference with control group with p < 0.05; # statistically significant difference with pair-fed group with p < 0.05.
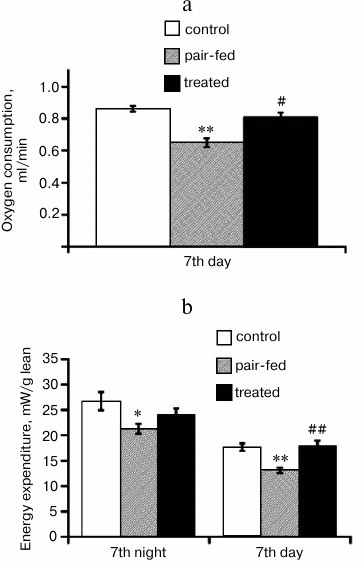
On the 7th day of the treatment, when food intake of treated mice was stabilized at the minimal level and body weight continued decreasing, mice were again placed in the metabolic chambers for 24 h. Oxygen consumption rate of treated mice was almost all the time higher than oxygen consumption rate of pair-fed mice (not shown). RMR was significantly lower in pair-fed group than in control group (Fig. 5a), which is probably associated with low food intake. However, RMR was higher by 23% in treated group than in pair-fed group in spite of the same food intake, and did not differ from RMR of the control group.
Fig. 5. Resting metabolic rate (a) and energy expenditure (b) during the 7th day of the treatment. a) Resting metabolic rate calculated for each mouse as minimal stable for 10-15 min oxygen consumption rate. The values are means ± S.E. (n = 5 for each group). b) Energy expenditure calculated by means of Weir equation and normalized to lean mass. The values are means ± S.E. during day and night period (n = 5 for each group); *, ** indicate statistically significant differences with control group; #, ## statistically significant differences with pair-fed group with p < 0.05 (one symbol) and p < 0.01 (two symbols).
Energy expenditure can be estimated from respiratory exchanges. Energy expenditure is proportional to volume of oxygen consumed. But since the amount of oxygen needed to burn different fuels – fat, proteins, carbohydrates – is not strictly proportional to energy released from this burn, or in other words, since energy equivalent of oxygen depends on the substrate oxidized, oxygen consumption should be corrected by respiratory quotient or carbon dioxide produced. In our study, we used the Weir equation to calculate energy expenditure and normalized it to lean mass (metabolically more active). Energy expenditure was higher in the treated group than in pair-fed group (Fig. 5b).
On the 15th day of the treatment, when food intake of treated and pair-fed mice was fully restored and body weight started to increase, the mice were placed in the metabolic chambers for 24 h. At this stage, RMR was the same in all groups of mice (not shown). RQ of pair-fed and treated mice was back to normal values (not shown).
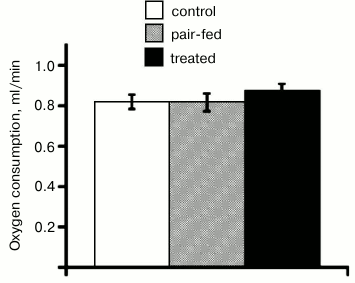
Effects of DNP on metabolic parameters of mice. The second aim of the present study was to compare anti-obesity effects of C4R1 with the effect of the classical uncoupler DNP. DNP was administrated in drinking water as for C4R1. The actual dose of DNP calculated based on water intake was 27 µmol/kg daily. DNP in this dose did not affect water intake, food intake, body weight (not shown), or resting metabolic rate (Fig. 6).
Fig. 6. Effect of DNP on resting metabolic rate. RMR was calculated on the 7th day of treatment, as in Fig. 5a. The values are means ± S.E. (n = 5 for each group). Dose of DNP was 27 µmol/kg daily.
DISCUSSION
In the present study, we found that the novel cationic mitochondrial uncoupling agent C4R1 effectively reduced body weight of obese mice. An attempt to extrapolate results of the study from mice to humans is impeded by substantial differences in thermoregulation and metabolism between these two mammals. The major problem of diet studies performed with mice is a significant elevation of thermogenesis in response to relatively small decrease in environmental temperature. Such extra heat production may mask the stimulating effects of potential anti-obesity drugs on metabolism [22, 23]. The problem has been clearly demonstrated in studies [8, 10, 24] where effects of DNP and uncoupling protein 1 (UCP1) were investigated in mice living at thermoneutrality (30°C) or at standard animal house temperature (22°C). Anti-obesity effects of DNP and UCP1 were evident only at thermoneutrality [8, 10, 24]. Thus, environmental temperature should be taken into account when assessing an anti-obesity drug in mice, particularly agents acting on energy expenditure. Our C4R1 study was performed under humanized conditions, when human metabolism is maximally reproduced in mice, namely at thermoneutrality (30°C) and with high fat and high sugar diet resembling the human obesogenic western diet. It suggests that anti-obesity effects of C4R1 observed here in mice could be developed in humans as well.
Two anti-obesity effects of C4R1 were observed. First, it reduced food intake, and second, increased resting metabolic rate. Such combination of two modes of action appears to be a desirable feature of C4R1 as an anti-obesity agent. It is known that poly-therapeutic strategies against obesity (targeting different pathways) often yield better results than strategies that modify one pathway alone [2]. Similarly to C4R1, two anti-obesity mechanisms (decreasing energy intake and increasing energy expenditure) were attributed to the anti-obesity agent that has been most popular in last decade, sibutramine, a dual monoamine (noradrenaline and serotonin)-reuptake inhibitor [28, 29].
A 25-50% loss of body weight and body fat mass at different stages of C4R1 treatment was explained by reduced food intake (as calculated from comparison with pair-fed group). The possible mechanism of food intake reduction could be either direct action at satiety centers in the central nervous system or influence on production (or functioning) of hormone-like peptides affecting appetite. Basic research (for review, see [30]) has identified a wide range of signal molecules produced in the gut (ghrelin, glucagon-like peptide-1, glucose-dependent insulinotropic polypeptide, oxyntomodulin, peptide YY), pancreas (insulin, amylin) and adipose tissue (leptin, adiponectin) and acting on satiety centers in the hypothalamus.
C4R1 increased RMR as compared to the pair-fed group. The mechanism behind elevated RMR is probably attributable to increased mitochondrial uncoupling. The studies of in vitro effects of C4R1 on isolated mitochondria revealed its uncoupling activity [20]. It has been suggested that C4R1 uncouples via an interaction with carrier protein(s) of the mitochondrial inner membrane [20]. Many carrier proteins have uncoupling activity under certain conditions [31-33]. Carrier protein UCP1 in brown adipose tissue mitochondria possesses the maximal uncoupling activity [34]. Previously, we showed that uncoupling by UCP1 is three times higher than uncoupling mediated by all carrier proteins together [35, 36]. An attractive idea is to explore whether C4R1 could interact with UCP1 in brown fat mitochondria. Brown adipose tissue as well as recently discovered “brite” adipocytes within white adipose tissue are the most powerful thermogenic and metabolically active (due to UCP1) systems [37-39]. The fact that C4R1 quickly started to stimulate lipid oxidation independently of food intake (decreased RQ already a few hours after the start of the treatment, Fig. 4) indicates the possible selective activation of UCP1 by C4R1. It is well known that brown adipose tissue was evolutionarily developed as the most effective system for burning lipids.
Remarkably, the classical anionic uncoupler DNP in dose 27 µmol/kg daily did not elevate the metabolism of mice. Our results are consistent with the study of dose-dependent effects of DNP [10]: only a dose 19 times higher than the dose used here decreased body weight, body fat mass, and increased energy expenditure [10]. The difference between in vivo effective doses of C4R1 and DNP is consistent with the difference between effective concentrations of the uncouplers in isolated mitochondria [20, 27].
Interestingly, in a study of Caldeira da Silva et al. [11] extremely low doses of DNP (50-170 times lower than dose used by us and 1000 times lower than dose used in [9, 10]) effectively decreased body weight and improved symptoms of metabolic syndrome. Caldeira da Silva et al. explained their results by uncoupling of oxidative phosphorylation [11], but did not prove it persuasively: neither RMR nor mitochondrial uncoupling (oligomycin insensitive respiration normalized to mg of mitochondrial protein) were measured. As an alternative explanation, effects of long-term treatment with DNP on gut microbiota could be proposed.
Our several step treatment with C4R1 (30 days in total) did not induce visible toxic effects in the mice. The behavior of mice was normal even with initial high dose, which substantially reduced food intake. Intriguingly, in nine days after the start of the treatment, food intake and body weight spontaneously started to restore during the treatment. This indicates the absence of toxic effects of C4R1 on the digestive tract. The fact that all in vivo effects of C4R1 observed by us (on body weight, food intake, RMR, and RQ) were temporal and started to spontaneously restore on 9-15th day of the treatment appears to be extremely interesting and needs to be studied further. Repeated treatment with C4R1 in six days after treatment cancellation had similar effects (reduced body weight and food intake). This opens the possibility to use the drug not constantly, but with short (few days) courses.
Thus, the novel cationic uncoupler C4R1 may be a stepping-stone to creation of an effective and safe anti-obesity drug.
The authors are deeply grateful to Prof. V. P. Skulachev, Prof. B. Cannon, and Prof. J. Nedergaard for discussion of the results and valuable comments, and to K. G. Lyamzaev and E. B. Menshikova for help with figures and text of the paper.
This study was sponsored by the Institute of Mitoengineering of Moscow State University and by Mitotech S.A.
REFERENCES
1.Tseng, Y. H., Cypess, A. M., and Kahn, C. R. (2010)
Cellular bioenergetics as a target for obesity therapy, Nature Rev.
Drug Discov., 9, 465-482.
2.Rodgers, R. J., Tschop, M. H., and Wilding, J. P.
(2012) Anti-obesity drugs: past, present and future, Dis. Model
Mech., 5, 621-626.
3.Wadden, T. A. (1993) Treatment of obesity by
moderate and severe caloric restriction. Results of clinical research
trials, Ann. Intern. Med., 119, 688-693.
4.Harper, J. A., Dickinson, K., and Brand, M. D.
(2001) Mitochondrial uncoupling as a target for drug development for
the treatment of obesity, Obes. Rev., 2, 255-265.
5.Nedergaard, J., and Cannon, B. (2010) The changed
metabolic world with human brown adipose tissue: therapeutic visions,
Cell Metab., 11, 268-272.
6.Tainter, M. L., Cutting, W. C., and Stockton, A. B.
(1934) Use of dinitrophenol in nutritional disorders: a critical survey
of clinical results, Am. J. Public Health Nations Health,
24, 1045-1053.
7.Parascandola, J. (1974) Dinitrophenol and
bioenergetics: an historical perspective, Mol. Cell Biochem.,
5, 69-77.
8.Shemano, I., and Nickerson, M. (1963) Mechanisms of
thermal responses to 2,4-dinitrophenol, J. Pharmacol. Exp.
Ther., 139, 88-93.
9.Schlagowski, A. I., Singh, F., Charles, A. L., Gali
Ramamoorthy, T., Favret, F., Piquard, F., Geny, B., and Zoll, J. (2014)
Mitochondrial uncoupling reduces exercise capacity despite several
skeletal muscle metabolic adaptations, J. Appl. Physiol. (1985),
116, 364-375.
10.Goldgof, M., Xiao, C., Chanturiya, T., Jou, W.,
Gavrilova, O., and Reitman, M. L. (2014) The chemical uncoupler
2,4-dinitrophenol (DNP) protects against diet-induced obesity and
improves energy homeostasis in mice at thermoneutrality, J. Biol.
Chem., 289, 19341-19350.
11.Caldeira da Silva, C. C., Cerqueira, F. M.,
Barbosa, L. F., Medeiros, M. H., and Kowaltowski, A. J. (2008) Mild
mitochondrial uncoupling in mice affects energy metabolism, redox
balance and longevity, Aging Cell, 7, 552-560.
12.Quin, C., Robertson, L., McQuaker, S. J., Price,
N. C., Brand, M. D., and Hartley, R. C. (2010) Caged mitochondrial
uncouplers that are released in response to hydrogen peroxide,
Tetrahedron, 66, 2384-2389.
13.Shabalina, I. G., and Nedergaard, J. (2011)
Mitochondrial (“mild”) uncoupling and ROS production:
physiologically relevant or not, Biochem. Soc. Trans.,
39, 1305-1309.
14.Hatcher, A. S., Alderson, J. M., and
Clements-Jewery, H. (2011) Cardiac mitochondrial uncoupling agents
trigger ventricular fibrillation in isolated rat hearts, J.
Cardiovasc. Pharmacol., 28, 28.
15.McFee, R. B., Caraccio, T. R., McGuigan, M. A.,
Reynolds, S. A., and Bellanger, P. (2004) Dying to be thin: a
dinitrophenol related fatality, Vet. Hum. Toxicol., 46,
251-254.
16.Grundlingh, J., Dargan, P. I., El-Zanfaly, M.,
and Wood, D. M. (2011) 2,4-dinitrophenol (DNP): a weight loss agent
with significant acute toxicity and risk of death, J. Med.
Toxicol., 7, 205-212.
17.Severin, F. F., Severina, I. I., Antonenko, Y.
N., Rokitskaya, T. I., Cherepanov, D. A., Mokhova, E. N., Vyssokikh, M.
Y., Pustovidko, A. V., Markova, O. V., Yaguzhinsky, L. S., Korshunova,
G. A., Sumbatyan, N. V., Skulachev, M. V., and Skulachev, V. P. (2010)
Penetrating cation/fatty acid anion pair as a mitochondria-targeted
protonophore, Proc. Natl. Acad. Sci. USA, 107,
663-668.
18.Trendeleva, T. A., Sukhanova, E. I., Rogov, A.
G., Zvyagilskaya, R. A., Seveina, I. I., Ilyasova, T. M., Cherepanov,
D. A., and Skulachev, V. P. (2013) Role of charge screening and
delocalization for lipophilic cation permeability of model and
mitochondrial membranes, Mitochondrion, 13, 500-506.
19.Antonenko, Y. N., Avetisyan, A. V., Cherepanov,
D. A., Knorre, D. A., Korshunova, G. A., Markova, O. V., Ojovan, S. M.,
Perevoshchikova, I. V., Pustovidko, A. V., Rokitskaya, T. I., Severina,
I. I., Simonyan, R. A., Smirnova, E. A., Sobko, A. A., Sumbatyan, N.
V., Severin, F. F., and Skulachev, V. P. (2011) Derivatives of
rhodamine 19 as mild mitochondria-targeted cationic uncouplers, J.
Biol. Chem., 286, 17831-17840.
20.Khailova, L. S., Silachev, D. N., Rokitskaya, T.
I., Avetisyan, A. V., Lyamsaev, K. G., Severina, I. I.,
Il’yasova, T. M., Gulyaev, M. V., Dedukhova, V. I., Trendeleva,
T. A., Plotnikov, E. Y., Zvyagilskaya, R. A., Chernyak, B. V., Zorov,
D. B., Antonenko, Y. N., and Skulachev, V. P. (2014) A short-chain
alkyl derivative of rhodamine 19 acts as a mild uncoupler of
mitochondria and a neuroprotector, Biochim. Biophys. Acta,
1837, 1739-1747.
21.Rokitskaya, T. I., Ilyasova, T. M., Severina, I.
I., Antonenko, Y. N., and Skulachev, V. P. (2013) Electrogenic proton
transport across lipid bilayer membranes mediated by cationic
derivatives of rhodamine 19: comparison with anionic protonophores,
Eur. Biophys. J., 42, 477-485.
22.Lodhi, I. J., and Semenkovich, C. F. (2009) Why
we should put clothes on mice, Cell Metab., 9,
111-112.
23.Nedergaard, J., and Cannon, B. (2014) The
browning of white adipose tissue: some burning issues, Cell
Metab., 20, 396-407.
24.Feldmann, H. M., Golozoubova, V., Cannon, B., and
Nedergaard, J. (2009) UCP1 ablation induces obesity and abolishes
diet-induced thermogenesis in mice exempt from thermal stress by living
at thermoneutrality, Cell Metab., 9, 203-209.
25.Teodoro, J. S., Zouhar, P., Flachs, P., Bardova,
K., Janovska, P., Gomes, A. P., Duarte, F. V., Varela, A. T., Rolo, A.
P., Palmeira, C. M., and Kopecky, J. (2014) Enhancement of brown fat
thermogenesis using chenodeoxycholic acid in mice, Int. J. Obes.
(Lond.), 38, 1027-1034.
26.Watanabe, M., Houten, S. M., Mataki, C.,
Christoffolete, M. A., Kim, B. W., Sato, H., Messaddeq, N., Harney, J.
W., Ezaki, O., Kodama, T., Schoonjans, K., Bianco, A. C., and Auwerx,
J. (2006) Bile acids induce energy expenditure by promoting
intracellular thyroid hormone activation, Nature, 439,
484-489.
27.Jacob, M., Bjarnason, I., Rafi, S.,
Wrigglesworth, J., and Simpson, R. J. (2001) A study of the effects of
indometacin on liver mitochondria from rats, mice and humans,
Aliment. Pharmacol. Ther., 15, 1837-1842.
28.Connoley, I. P., Liu, Y. L., Frost, I., Reckless,
I. P., Heal, D. J., and Stock, M. J. (1999) Thermogenic effects of
sibutramine and its metabolites, Br. J. Pharmacol., 126,
1487-1495.
29.Ryan, D. H. (2000) Use of sibutramine and other
noradrenergic and serotonergic drugs in the management of obesity,
Endocrine, 13, 193-199.
30.Halford, J. C., Boyland, E. J., Blundell, J. E.,
Kirkham, T. C., and Harrold, J. A. (2010) Pharmacological management of
appetite expression in obesity, Nature Rev. Endocrinol.,
6, 255-269.
31.Andreyev, A. Y., Bondareva, T. O., Dedukhova, V.
I., Mokhova, E. N., Skulachev, V. P., Tsofina, L. M., Volkov, N. I.,
and Vygodina, T. V. (1989) The ATP/ADP-antiporter is involved in the
uncoupling effect of fatty acids on mitochondria, Eur. J.
Biochem., 182, 585-592.
32.Samartsev, V. N., Smirnov, A. V., Zeldi, I. P.,
Markova, O. V., Mokhova, E. N., and Skulachev, V. P. (1997) Involvement
of aspartate/glutamate antiporter in fatty acid-induced uncoupling of
liver mitochondria, Biochim. Biophys. Acta, 1319,
251-257.
33.Skulachev, V. P. (1999) Anion carriers in fatty
acid-mediated physiological uncoupling, J. Bioenerg. Biomembr.,
31, 431-445.
34.Nedergaard, J., Golozoubova, V., Matthias, A.,
Asadi, A., Jacobsson, A., and Cannon, B. (2001) UCP1: the only protein
able to mediate adaptive non-shivering thermogenesis and metabolic
inefficiency, Biochim. Biophys. Acta, 1504, 82-106.
35.Shabalina, I. G., Jacobsson, A., Cannon, B., and
Nedergaard, J. (2004) Native UCP1 displays simple competitive kinetics
between the regulators purine nucleotides and fatty acids, J. Biol.
Chem., 279, 38236-38248.
36.Shabalina, I. G., Kramarova, T. V., Nedergaard,
J., and Cannon, B. (2006) Carboxyatractyloside effects on brown-fat
mitochondria imply that the adenine nucleotide translocator isoforms
ANT1 and ANT2 may be responsible for basal and fatty-acid-induced
uncoupling respectively, Biochem. J., 399, 405-414.
37.Petrovic, N., Walden, T. B., Shabalina, I. G.,
Timmons, J. A., Cannon, B., and Nedergaard, J. (2010) Chronic
peroxisome proliferator-activated receptor γ (PPAR γ)
activation of epididymally derived white adipocyte cultures reveals a
population of thermogenically competent, UCP1-containing adipocytes
molecularly distinct from classic brown adipocytes, J. Biol.
Chem., 285, 7153-7164.
38.Shabalina, I. G., Ost, M., Petrovic, N., Vrbacky,
M., Nedergaard, J., and Cannon, B. (2010) Uncoupling protein-1 is not
leaky, Biochim. Biophys. Acta, 1797, 773-784.
39.Shabalina, I. G., Petrovic, N., de Jong, J. M.,
Kalinovich, A. V., Cannon, B., and Nedergaard, J. (2013) UCP1 in
brite/beige adipose tissue mitochondria is functionally thermogenic,
Cell Rep., 5, 1196-1203.