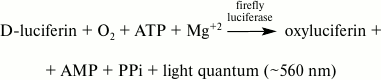REVIEW: Bioluminescence Assay for Cell Viability
G. Yu. Lomakina, Yu. A. Modestova, and N. N. Ugarova*
Faculty of Chemistry, Lomonosov Moscow State University, 119991 Moscow, Russia; fax: +7 (495) 939-2660; E-mail: nugarova@gmail.com* To whom correspondence should be addressed.
Received January 16, 2015
Theoretical aspects of the adenosine triphosphate bioluminescence assay based on the use of the firefly luciferin–luciferase system are considered, as well as its application for assessing cell viability in microbiology, sanitation, medicine, and ecology. Various approaches for the analysis of individual or mixed cultures of microorganisms are presented, and capabilities of the method for investigation of biological processes in live cells including necrosis, apoptosis, as well as for investigation of the dynamics of metabolism are described.
KEY WORDS: bioluminescence, firefly luciferase, ATP, intracellular ATP, cell lysis, extracellular ATP, lyophilized vaccinesDOI: 10.1134/S0006297915060061
GENERAL CHARACTERISTICS OF BIOLUMINESCENCE ASSAY OF INTRACELLULAR ATP
ATP as an indicator of cell viability. Adenosine triphosphate (systematic name 9-β-D-ribofuranosyl adenine-5′-triphosphate or 9-β-D-ribofuranosyl-6-amino-purine-5′-triphosphate) − a nucleotide, an adenosine triphosphate ester that is a derivative of adenine and ribose (ATP) − is the main energy carrier in cells of all living organisms (mammals, microorganisms, plants, etc.) [1]. Cleavage of one or two phosphate groups that occurs during ATP hydrolysis is accompanied by the release of energy. In cells, ATP transfers energy to other molecules upon hydrolysis to its low-energy analogs (ADP and/or AMP), which, in turn, acquire energy by adding phosphate groups and transforming into ATP. Intracellular ATP content is the main indicator of cell viability. Upon cell death, ATP synthesis is the first to be arrested, while its hydrolysis can continue for some time, hence, the intracellular ATP content drops sharply to zero value. The ATP content in viable cells of microorganisms is quite high – it ranges from 500 to 10,000 µg per g of dry biomass [2] or from 10–19 to 10–15 mol of ATP per cell. It can vary depending on the type and size of cells and their energy state. Cellular response to any stress involves change in the intracellular ATP content. The intracellular ATP concentration can also vary depending on cultivation conditions, but the physiological changes that do not lead to the cell death do not exceed 10-50-fold. On the example of individual strains of microorganisms, it was shown that the intracellular ATP content for a cell suspension under standard conditions is proportional to the concentration of cells in the suspension [3]. Hence, it is possible to estimate the number of viable cells in a sample by measuring the intracellular ATP concentration.
Determination of ATP concentration using bioluminescence assay. There are various methods to determine ATP concentration: enzymatic methods with spectrophotometric detection, radioactive and chromatographic methods, and others. The bioluminescence ATP assay is the most sensitive, rapid, and selective. As far back as in the 1940s, it was shown that ATP is a required component in the reaction catalyzed by the firefly luciferase enzyme [4]. The overall reaction that proceeds in the luciferin–luciferase system of fireflies is described by the following scheme:
According to this scheme, an organic substrate (luciferin) is rapidly oxidized in the presence of ATP and magnesium ions by air oxygen to oxyluciferin with simultaneous formation of pyrophosphate (PPi) and AMP. Initially the oxyluciferin is formed in an electronically excited state, and a light quantum is emitted during the transfer of the product to the ground state. The advantages of firefly luciferase include its absolute specificity towards ATP and high emission quantum yield (~0.5 – the highest among known bioluminescence systems) [5].
Luminometers – specialized instruments that were first introduced to the market in 1970s – are used for bioluminescence measurements. The first luminometers were based on the technology developed for detection of radioactive emission – liquid scintillation counters [6]. Eventually another approach was developed, a photon counter in combination with a high-efficiency photomultiplier tube (PMT) is used in modern instruments for detection of luminescence signal. It must be noted that the signal recorded by a luminometer is proportional but not equal to the number of photons emitted by the sample. The signal is presented in relative luminescence units (RLU). Its absolute value is determined by the parameters of the particular instrument and can differ even for luminometers of the same model [7]. The light intensity recorded in RLU is proportional to the ATP concentration in the ATP linear range from 10 fM to 1 µM. There are a great variety of modern luminometers that differ in a number of parameters: sensitivity, type (tube luminometers and plate readers), size and design complexity (stationary and portable), availability of injectors or lack of them (i.e. intended for recording stationary (glow-type) or pulsed (flash) luminescence), etc. The portable luminometer LUM-1 – high-sensitivity photon counter capable of recording luminescence intensity in the range from 10 to 800 thousand pulses/s and with built-in computer interface – is manufactured in Russia (Fig. 1). The LUM-1 luminometer demonstrates the same sensitivity and operation stability as instruments of the same class from Berthold Detection Systems GmbH (Germany).
Fig. 1. LUM-1 portable luminometer (Lumtek, Russia).
Universal instruments capable of operating in multiple modes such as recording luminescence, fluorescence, and absorption are also available commercially. Accuracy of results is determined primarily by the selection of a suitable instrument for conducting analysis.
To unify the methods of analysis, dozens of foreign companies offer reagent kits for bioluminescence ATP assay, the so-called ATP reagents. The ATP reagents comprise lyophilized mixtures containing all components required for the luciferase reaction to proceed (luciferase, luciferin, magnesium salt, buffer component, and stabilizers) except for ATP. The lyophilized ATP reagents are reconstituted prior to use by adding a special solution for the ATP reagent reconstitution. The ATP reagents based on Russian luciferase from Luciola mingrelica fireflies have been developed [8, 9]. The reagents include a mutant recombinant luciferase that is significantly more active and thermostable than native luciferase [10, 11]. The sensitivity of the modern bioluminescence test systems allows detecting attomolar quantities of ATP in a sample and revealing the presence of single cells of microorganisms [12]. For quantitative determination of intracellular ATP, the cells are lysed to release the ATP, and the extract is introduced into the measuring cell, then the ATP reagent is added and the luminescence intensity is measured. To calculate accurate ATP concentration in the sample, a control is usually used that employs a standard ATP solution (ATP control) containing a known amount of ATP. The lyophilized ATP control is usually supplied together with the ATP reagent. The ATP control is reconstituted with the same extractant used for the cell lysis in the sample, and then the luminescence intensity is measured. The ATP concentration in the analyzed sample can be calculated by comparison with the standard.
The construction of graduation dependence between concentration of intracellular ATP and concentration of cells in a sample is an important step in quantitative determination of viability of microbial cells. Data of the standard method used for evaluation of the viability of microbial cells – serial dilution – are used for this purpose. The number of viable cells (CFU/ml) is determined using the standard method, and the ATP content in 1 ml of the sample is determined using the bioluminescence method. Linear correlation dependencies (y = a + bx) are obtained as a result:
[ATP], mol/ml = a + b·(CFU/ml),
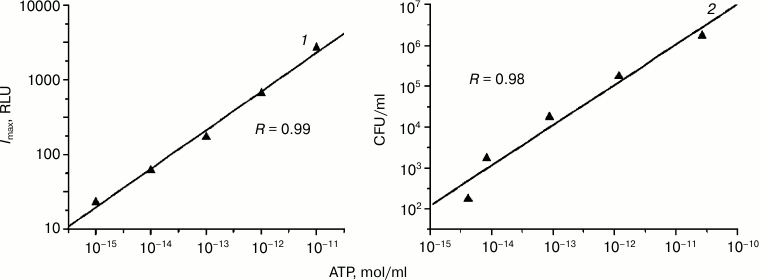
which are then used for determination of the (CFU/ml) value based on the value of [ATP], mol/ml. Good linear correlation between ATP concentration and CFU/ml is observed for individual cultures in the range from 103 to 108 CFU/ml (Fig. 2).
Fig. 2. Correlation between ATP concentration and bioluminescence intensity (1) and cell titer (2) in a sample.
The bioluminescence method allows rapid and quantitative determination of ATP amount. For this reason, it is used in various areas of molecular biology, sanitation, medicine, clinical diagnostics, food industry, ecology, and others. The possibilities and features of the use of the bioluminescence method for determination of the viability of microorganisms are presented below.
DETERMINATION OF INTRACELLULAR ATP CONTENT IN INDIVIDUAL CULTURES
OF MICROORGANISMS
Intracellular ATP is inaccessible to luciferase because the enzyme does not penetrate inside cells. Reagents that increase cell membrane permeability or disrupt it are used for the extraction of intracellular ATP. The type of the analyzed cells and the needs of the particular analysis primarily define the selection of the lysing agent. Extractants used for ATP assay must: (i) rapidly extract ATP; (ii) inactivate intracellular enzymes participating in interconversion of adenine nucleotides within the cell; (iii) maintain the initial ATP level of the intracellular ATP during extraction, storage, and analysis; (iv) not exhibit an inhibitory effect on the luciferase reaction during the ATP measurement.
Various types of compounds can be used for the ATP extraction: detergents and their mixtures, strong acids (i.e. perchloric and trichloroacetic), organic solvents (butanol, chloroform, dimethyl sulfoxide, etc.). Their application simplifies the experiment and allows conducting differential analysis based on different permeability and strength of the cell wall of different organisms in the presence of the extractants. Often a combination of chemical and physical methods for cell disruption is used: disintegration of cells by short-term boiling, sonication [13], or direct current [14].
Efficiency of ATP extraction from microorganisms depends mostly on the nature of the cell wall. Cytoplasmic membranes of microorganisms have similar structure and are easily disrupted with weak acid solutions or surfactants. The cell wall structures of Gram-positive and Gram-negative bacteria and of yeasts differ significantly, and these features must be considered upon selection of the lytic agent. For example, it was shown in [15], when the efficiency of different types of extractants was compared, that the maximum sensitivity of the bioluminescence analysis of intracellular ATP of Gram-positive bacteria and yeasts was observed using 0.1% solution of the detergent cetyltrimethylammonium bromide (CTAB). It was one order of magnitude higher in comparison with the 2.5% trichloroacetic acid (TCA) and 90% dimethyl sulfoxide (DMSO). This is mainly related to the fact that CTAB disrupts cells rapidly and effectively, while it inhibits luciferase significantly less in comparison with TCA and DMSO. However, CTAB was not applicable for the analysis of highly concentrated suspensions of Gram-negative microorganisms such as E. coli and Pseudomonas sp. It was not possible to use CTAB because noticeable deviations from linearity of the dependence of the ATP concentration on the cell concentration were observed when it was used. This is due to the fact that Gram-negative bacteria, in contrast to Gram-positive bacteria and yeasts, have significantly stronger cell wall, and, hence, stronger chemical agents are required to disrupt it. Unlike CTAB, DMSO was universal; it disrupted the cell walls of all investigated microorganisms instantly and inactivated all intracellular enzymes. The extraction time did not exceed 1 min. The resulting extracts were stable at room temperature for several hours, and it was possible to store them at 4°C for several days and even weeks, while the extracts obtained using CTAB were unstable during storage. Nevertheless, the use of detergents including CTAB for ATP extraction is the most popular method, and it is used in analysis very often. For example, cells were treated with 5 mM CTAB for 2 min for ATP extraction when a disposable sensor for assessing microbiological cleanness of a surface was developed [16].
The cationic detergent benzalkonium chloride (BAC), which compared well in efficiency with TCA, was used in [17] for analysis of intracellular ATP in 17 yeast strains (with suspension concentration of 105 CFU/ml) and 37 strains of Gram-positive and Gram-negative microorganisms (with concentration of 107 CFU/ml). It was shown that the average degree of ATP extraction by 0.2% BAC solution from Gram-positive bacteria was above 99.4%, and from yeast cells – above 97%. The extraction efficiency from Gram-negative bacteria was significantly less (~81%) in comparison with the TCA extraction. Especially low extraction was observed for Escherichia, Proteus, and Serratia microorganisms (74-80%). Increase in BAC concentration to 0.5% resulted in inhibition of the luciferase activity and significant increase in the bioluminescence signal decay rate from 5 to 50%/min. One hundred percent extraction of ATP from fungal spores was achieved upon treatment of the sample with 90% DMSO in TAE buffer, pH 7.75, for 1 min at 100°C [18]. For comparison, the efficiency of ATP extraction with boiling TAE buffer without DMSO (it was no more than 37.7%) and with 0.1-3% TCA solution (it was up to 23.3%) was assessed.
Special extraction conditions are required for analysis of mycobacteria, which differ from the other Gram-positive bacteria by unusual structure of their cell wall. In mycobacteria, the dense layer of peptidoglycan (the feature of Gram-positive bacteria) in the cells of this type of bacteria is strengthened by mycolic acid, mycolates, and arabinogalactan; their surface is hydrophobic due to the presence of lipids [19]. Dodecyltrimethylammonium bromide (2%) in Tris-EDTA buffer (pH 7.75) (100°C, 1 min) in the presence of α-cyclodextrin was among the optimal extractants [20]. An approach combining mechanical cell disruption (bead beating) and enzymatic action of lysozyme (Bactozyme preparation) [21] was also suggested. It was shown in [22] that the use of such extractants as TCA, perchloric acid (PCA), and ethylene glycol was undesirable for ATP extraction from cells with high protein content because their presence caused protein denaturation and partial ATP coprecipitation. Phenol saturated with Tris-EDTA is recommended for this case; this approach increases the ATP yield 14.5-fold in comparison with 5% TCA and 1000-fold in comparison with ethylene glycol.
Various procedures can be employed to account for the effect of anionic, cationic, and amphoteric detergents on the luciferase activity. One of the classic methods involves the use of various cyclodextrins [7]. For example, 7.5 mM β-cyclodextrin was used to decrease the inhibiting effect of 5 mM CTAB [16]. Liposomes were used to bind BAC following the ATP extraction from bacterial cells [23]. The addition of diethylaminoethyl dextran increased the luciferase activity in the presence of TCA and Triton X-100 [24]. Genetic engineering approaches are also used. For example, a double mutant of luciferase resistant to BAC was used in [17] to decrease the inhibiting effect of this extractant on the luciferin–luciferase reaction.
Intracellular ATP level is different in cells of different type and depends on many factors. The review [25] devoted to practical aspects of the bioluminescence assay application presents data from different sources on the ATP content in cells of bacteria and yeasts, in the spores of both bacteria and fungi, and in actinomycetes. It is stated that the ATP content in bacterial cells can differ by 4-5 orders of magnitude. For example, the ATP content of Gram-negative bacteria during the stationary growth phase was within the narrow range (~10–18 mol/CFU), while the ATP content of Gram-positive bacteria varied in a wide range from 0.4⋅10–18 to 16⋅10–18 mol/CFU, but on average it was one order of magnitude higher as compared with the Gram-negative bacteria [17]. The intracellular ATP content of different strains of yeast cells was in the range (0.7-54)⋅10–16 mol/CFU. The ATP content in the cell can be related with its size [26] and volume [27]. The intracellular ATP concentration is generally rather high; on average, it is about 1-10 mM. According to [28], the ATP concentration in the E. coli cell is no less than 3 mM and independent of the cell propagation rate in the mid-log growth phase. In 2014, a group of scientists was able to determine an absolute content of ATP in individual E. coli cells using the genetically encoded fluorescent ATP indicator QUEEN [29]. It was found that the ATP content within a cell population has a positively asymmetric distribution; average ATP concentration within an individual cell was 1.54 ± 1.22 mM. The averaged data obtained using fluorescence (QUEEN) and bioluminescence analysis were in full agreement with each other. Moreover, using five different species of marine bacteria, it was shown that there is a good correlation between the ATP and carbon content in the cell [30].
Evaluation of metabolic activity of cells based on intracellular ATP content. An increasing number of researchers use the bioluminescence method as a powerful tool for evaluation of the metabolic activity of cells. When using the bioluminescence assay, the detection limit of bacterial cells may essentially depend on the phase of their life cycle. On the example of E. coli and S. aureus cells [31], it was shown that the level of intracellular ATP changes depending on the growth phase of the microorganism (lag-phase, log-phase, stationary phase, and cell death). Metabolic activity changes in S. cerevisiae during growth and at different temperatures were studied in [32]. Three sub-populations of the yeast that respond differently to elevated temperature were distinguished: viable cells, dead cells, and viable but nonculturable cells (hence undetectable by the plating method). The ATP level in the cell can increase with increasing temperature because of activation of metabolic paths essential for the cell to withstand this stress.
Bioluminescence analysis was used in [33] to study the effect of cell adhesion to a glass surface on metabolic activity and, respectively, on the level of intracellular ATP. It was established that the intracellular ATP content increased 2-5-fold during adhesion of E. coli and B. brevis cells to the glass surface in comparison with the level characteristic of the cells in suspension. Data obtained in [34] indicate that the level of metabolic activity in the cell depends on the properties of the adhering surface: for a number of cells, adhesion to a hydrophobic surface resulted in two-fold increase in the intracellular ATP content in comparison with the same cells adhered on a hydrophilic surface.
Kinetic analysis of change in intracellular ATP content as well as analysis of ATP/ADP ratio allows investigating various cell pathologies including mechanisms of cell death – necrosis and apoptosis. As long as the cells are alive, they tend to maintain normal ATP level, whereas the level of intracellular ATP decreases to zero upon cell death. And yet the rate of ATP hydrolysis in cells is different. Mechanical damage of cytoplasmic and intracellular membranes occurs under the action of biological, physical, or chemical factors during necrosis, which results in disruption of organelles, release of lysosomal enzymes, and release of the cytoplasmic content into the extracellular space. Mechanisms of necrosis do not require energy, unlike apoptosis, during which the cell attempts not only to maintain but to increase ATP level for several hours to supply energy for the process via glycolysis or synthesis of mitochondrial ATP. The type of cell death – via apoptosis or necrosis – is determined mainly by the intracellular concentrations of NADH and ATP. The bioluminescence method was used for determination of intracellular ATP when investigating mechanisms of cell death of macrophages under the action of different strains of M. tuberculosis and showed that ATP synthesis increased sharply in the cells infected by the virulent strain H37Rv, unlike in cells in the presence of nonvirulent H37Ra strain [35].
Bioluminescence methods for evaluation of cytotoxicity of various preparations against bacterial cells. Good agreement was obtained between the bioluminescence method and laser nephelometry when antibacterial properties of a series of cyclodextrin complexes were evaluated [36]. To assess the efficiency of new chemical disinfectants, standard techniques developed by the European Committee for Standardization, EN 1276 and EN 13727 in particular, are used. These standards describe the analysis of bacterial suspensions. They prescribe the following steps in the measurement: a biocide solution is mixed with the bacterial suspension in the presence of organic matter (bovine serum albumin, for example) that mimics conditions under which the test solution is used in practice. Following a prescribed time period, the activity of the effector is inhibited and the viability of the microorganisms is determined using the colony counting technique on nutrient medium. It was shown in [37] that these techniques could be significantly simplified and optimized if the colony counting technique was replaced by semiautomatic bioluminescence assay for bacteria viability. The bioluminescence method was also used for cytotoxicity screening of pharmaceutical products against Leishmania promastigotes (a type of parasitic protozoans) [38]. Bioluminescence was also used in [39] for quantitative assessment of proliferation of another protozoan – Perkinsus marinus, which is an oyster parasite. The efficiency of oral antiseptics was assessed in [40] using bioluminescence analysis. The bioluminescence method for assessing the effectiveness of mouthwashes against biofilms grown in vitro using mouth microflora was validated in [41].
Biological fluids can be analyzed for ATP content without pretreatment of the sample because the turbidity and coloring of the liquid do not significantly interfere with the analysis. For example, the effect of radiation and xenobiotics on intracellular ATP content was investigated in [42] not only for blood cells (erythrocytes and neutrophils), but also for the whole blood, which was diluted 10-fold with physiological solution prior to analysis; DMSO was used as the extractant. The dynamics of ATP content and its absolute values indicated metabolic changes. The observed increase in the ATP level in whole blood and in neutrophils was statistically significant. An interesting approach for application of bioluminescence assay was suggested in [43]. The authors used a plating method for detection of bacterial cells and spores, which was modified as follows: the sample was plated onto a filter surface, which was placed on the surface of solidified nutrient medium and incubated for a certain time that depended on the growth rate of the cells of this type. Next the filter was transferred into a special device where the filter surface was treated to remove extracellular ATP, and the intracellular ATP was extracted from the colonies grown on the filter that were still invisible to the naked eye. Then the filter surface was treated with the ATP reagent, and luminescence was recorded with a sensitive CCD camera. This shortened the time required for the cell growth, and it was possible to count the colonies before they became visible to the naked eye.
DETERMINATION OF INTRACELLULAR ATP IN A MIXED CULTURE OF
MICROORGANISMS
The method for determination of intracellular ATP can be applied not only for analysis of individual cultures, but also of their mixtures, and it can be successfully used instead of time-consuming microbiological analysis in cases when knowledge of total microbial counts is required. In work [26], bioluminescence ATP assay was used for analysis of sterility of indoor facilities. Plating onto solid TSA medium followed by colony counting was used for detection of culturable microorganisms, and also the average ATP content in a live cell – ATP/CFU ratio – was determined. In the case of low ATP/CFU ratio, it can be assumed that predominately Gram-negative microorganisms and spores are present in the sample, while if the ratio is high – Gram-positive bacteria and yeasts. It was found that a large number of samples did not form any colonies on the nutrient medium, and yet a high level of intracellular ATP was recorded. This suggested the presence of viable but nonculturable microorganisms, which was later confirmed by DNA analysis. Hence, bioluminescence ATP assay can be used for express detection of nonculturable or slowly growing microorganisms. It must be mentioned that despite a wide variety of microorganisms in nature, only a limited number can be cultivated under laboratory conditions. Data on the efficiency of the cultivation characteristic of bacteria in different habitats are presented in review [44]. The least effective cultivation is usually observed for bacteria living in environments with low nutrient levels (below 1%), and the highest for bacteria living in nutrient-rich habitats (up to 58%).
Determination of total bacterial count in air (TBC, CFU/m3), 1 m3 of which can contain up to 106 cells of aerobic pathogenic and nonpathogenic microorganisms (fungi, bacteria, spores, and others), is very important for analysis of sanitary quality of air in facilities with different air quality requirements (according to ISO 14644-1-99), including sterile and clean rooms (0-10 CFU/m3) in health care facilities, industrial premises, and offices in food and microbiological industries. The commonly used microbiological method requires no less than 24 h. A semiquantitative method for evaluation of TBC in air in rooms with low concentrations of microorganisms was suggested in [45]. Air was pumped (optimal air flow 200 liters/min) through a wet sterile adsorbing surface to capture aerobic microorganisms, and the samples were incubated in nutrient medium for 3-6 h to the beginning of the logarithmic growth phase. Both Tryptic Soy Broth (Difco, USA) and nutrient broth (State Research Center for Applied Microbiology and Biotechnology, Russia) were found to be suitable. Good agreement between the bioluminescence assay and the microbiological plating method was obtained: 3-h incubation of the sample at 37°C revealed 50-100 CFU/m3 (cleanness grade 6-7), and 5-h – 5-50 CFU/m3 (cleanness grade 5, especially clean facilities). If the total count of microorganisms was ≥500 CFU/m3 (grade 9 and above), the result of analysis was obtained within 5 min without prior incubation.
A system for automatic investigation of bioaerosols was described in [46]; it comprised an atomizer for preparation of bioaerosols from suspension of bacterial cells, an air ionizer, and a measuring unit of complex design in which cell lysis occurred under the action of ionized air followed by bioluminescence ATP assay. The authors automated the experiment, decreased the analysis time to 5 min, and detected bacterial cells (E. coli and S. epidermidis) at concentrations from 173 to 541 CFU/m3 without enrichment.
Adhesion of bacterial cells to surfaces of various nature (glass, metal, plastic, etc.) results in formation of biofilms, which create significant problems in both medicine and industry. Bioluminescence analysis allows the investigation of the formation of communities of microorganism comprising biofilms, their existence, survival, and distraction under the action of different effectors [41, 47-50].
Determination of cells of individual microorganism in the presence of accompanying microflora is a rather difficult task. Different approaches are used for this purpose. The traditional way to selectively isolate cells of a target strain is cultivation of the sample on selective nutrient medium, which supports preferable growth of the strain of interest. For example, milk samples were incubated in selective nutrient medium for 6 h for determination of coli-form organisms in food (raw milk and ice cream) and rinses, which allowed the authors to detect 10 CFU/ml in a sample [51]. The authors in [52] used an immunomagnetic separation technique for specific capture of E. coli O157:H7 by antibodies attached to magnetic particles. The cell content was then determined from the level of intracellular ATP. The reached limit of detection was 102 CFU/ml.
The use of lytic bacteriophage for rapid and specific lysis of target cells allows reliable detection of 104 CFU/ml in the presence of nonspecific microflora. Concentration of the initial cell suspension prior to detection improves the analytical characteristics of the method by one order of magnitude. In such manner, positively charged nanoaluminum fiber-based filters with pore size of 2 µm with adsorbed T4 bacteriophage allowed quantitative determination of E. coli cells in the presence of 60-fold excess of S. typhimurium [53]. A photodynamic method for selective disruption of Gram-negative and Gram-positive bacteria, using strains of E. coli O157:H7 and L. monocytogenes as an example, as well as for specific detection of yeast cells in mixture with E. coli was used in [54, 55].
Total ATP content in a sample represents a convenient indicator for estimation of hygienic conditions by characterizing residual content of live and disrupted bacterial or somatic cells. A large number of studies have been devoted to express analysis of rinses from industrial surfaces, dishes, equipment, food products, etc. All cells provide ATP, both microbial and non-microbial (somatic), as well as cellular matter of organic nature, for example, food residues. Treatment of cells with disinfecting agents results in their destruction and release of ATP. Hence, this method can be employed for evaluation of the efficiency of disinfecting agents and quality of sanitation of an object. Special instrumentation has been designed and is widely used for this purpose that allows primary screening of hygienic condition of facilities on-site and in real time. Numerous companies (Biotrace from New Zealand; HY-LiTETM, Merck, Hygiena International, and Berthold Detection Systems GmbH from Germany; New Horizons Diagnostic Corp. and Pierce from USA; BioThema from Sweden; Kikkoman Corp. from Japan; Roche Diagnostics Ltd. from Switzerland; Lumtek from Russia, and others) offer various kits for analysis that contain reagents, disposables, and luminometers. The Hygiena International, for example, offers single-use devices for sampling and measuring of total ATP content in combination with a portable luminometer. The method was tested in food industry [56] for monitoring of microbial contamination on hands and household surfaces [57], for assessing hygienic conditions of technological surfaces in food-handling facilities [58], and in hospitals [59]. The method is quite effective for real-time monitoring of the cleanliness of medical equipment, such as endoscopes, in the process of their disinfection, which allows prevention of cross-infection among patients [60].
However, it has been mentioned in a number of publications that low correlation was observed between the results of control microbiological tests and bioluminescence assay while the method was validated in industry and large amounts of data have been statistically processed [56]. Various factors could cause false negative results in determination of intracellular ATP: incomplete cell disruption or incomplete extraction, low rate of extraction, decrease in intracellular ATP concentration due to its hydrolysis by ATPases during extraction, low stability of the bioluminescence signal, ATP disruption during storage of extract, etc. In the development of an ATP extraction technique for a particular sample, it is very important to make sure that the method yields correct data on intracellular ATP content in the sample. Observed false-positive results are usually related to a high level of the extracellular ATP, which occurs when cells are already disrupted but not removed. In this case, it is necessary to differentiate total ATP content into extracellular (disrupted cells) and intracellular (live cells) ATP.
Determination of extracellular ATP content. The amount of extracellular or so-called “free” ATP in a cell suspension is usually one or two orders of magnitude lower than the intracellular ATP. For example, the extracellular ATP content in a culture of metabolically active E. coli and Salmonella cells constituted no more than 5% of the total ATP pool [61], and it was defined by the growth phase of the cells. The level of extracellular ATP increased during the logarithmic phase and decreased during the stationary phase. The sole exception was the Acinetobacter junii AJ4970 cells, for which the ratio of extracellular to intracellular ATP was found to be above 0.5. It was shown that the bacterial cells rapidly consume extra free ATP with rate of approximately 5 µmol/h. According to [61], an artificial increase in ATP concentration in nutrient medium resulted in better survival of cells during prolonged incubation (7 days).
The extracellular ATP level can increase significantly under external stresses due to escape of intracellular ATP into the environment. The extracellular ATP content can be indicative of the number of destroyed cells upon, for example, freezing, in the process of lyophilization, under unfavorable storage conditions, etc. Live and disrupted cells can be distinguished by the ratio of extra- and intracellular ATP. For example, a method is described in [62] for quantitative determination of bacteria sensitivity to nisin – a broad spectrum antibiotic affecting Gram-positive bacteria that is widely used as a preservative. Under the action of nisin, cells release ATP, ADP, and AMP into the environment. Using the enhanced sensitivity of Lactococcus cremoris cells to this antibiotic, it was shown that the nisin concentration could be determined with the bioluminescence method from the amount of the extracellular ATP.
As shown in [34], the amount of extracellular ATP secreted by a series of Gram-positive and Gram-negative bacteria depended on the nature of the surface in contact with the microorganisms: contact with a surface unfavorable for cells stimulated the level of ATP release, and the level of extracellular ATP for Gram-negative bacteria was on average higher than for Gram-positive bacteria.
Importance of extracellular ATP removal. Hence, to correctly determine intracellular ATP, it is necessary to first remove extracellular ATP, followed by extraction of the intracellular ATP. Two main methods are used for removal of extracellular ATP: a) filtration of cell suspension through a membrane filter during which the extracellular ATP is removed together with the filtrate [63]; b) enzymatic hydrolysis of the extracellular ATP [64]. Selection of one method or the other is based on peculiarities of the analyzed suspension of microorganisms. Filtration is used in cases when the determined concentrations of intracellular ATP are less than 1 pM (at low concentrations of cells, which is, for example, lower than 102 CFU/ml for Gram-negative microorganisms), which at the same time results in concentration of cells on the filter and increase in the measured ATP concentration. The enzymatic hydrolysis of ATP is preferable when the cell concentrations are sufficiently high (above 104 CFU/ml).
Special 300-µl cuvettes with filtering bottom having pore size of 0.45 µm (Filtravette™; New Horizons Diagnostic Corp., USA) were used in [65] for determination of total microbiological contamination of food products. The removal of extracellular ATP, concentration of bacterial cells, ATP extraction, and measuring of its content using the bioluminescence method is conducted sequentially in the same cuvette. In the case when the sample contains more than 1000 CFU/ml (for E. coli), the analysis time is no more than 5-10 min. Preincubation of the sample for 6 h in nutrient medium to increase metabolic status of the cells and their propagation allowed detection of 1 CFU/ml in drinking water [66]. The same approach was used in [67] for control of sterility in health care facilities. The lack of bioluminescence signal following 6 h of incubation of rinses from different surfaces in nutrient medium indicated sterility of the object. In the case when a positive bioluminescence signal was observed after 6 h of incubation, the microbial contamination of the sample was 1-10 CFU/100 cm2. Microbial contamination above 100 CFU/100 cm2 was detected without incubation. The correlation coefficient between the ATP and CFU was 0.8-0.95. The problem of high content of non-bacterial ATP also presents challenges for analysis of purity of soya milk. The possible sources of non-bacterial ATP in soya milk are fibers and proteins in its composition [68]. Pretreatment of such a sample with a mixture of nonionic detergents [68] and removal of non-target ATP allowed using the bioluminescence method for sterility testing of such products.
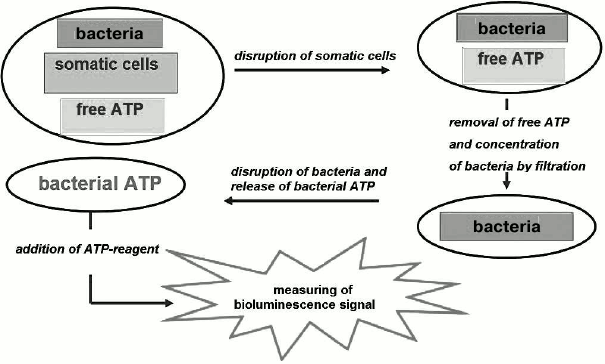
Total bacterial counts or quantity of mesophilic aerobic and facultative anaerobic microorganisms (QMAFAnM) represent one of the main indicators of the sanitary state of raw milk, which determines ways for further processing of the milk and affects the final price of the product. The generally recognized microbiological method according to State Standard GOST 9225-84 (Russia) allows determination of QMAFAnM in milk following 72 h incubation of plated samples; hence, express methods for evaluation of product quality, including the bioluminescence ATP assay with total analysis time of 20 min and detection limit 1000 CFU/ml, are of particular interest. For this purpose, a milk sample is incubated with a mixture of detergents and protease to destroy somatic cells and protein micelles in the milk. Next, the non-microbial ATP is removed by filtration through a bacterial filter, the bacteria are disrupted with DMSO, and the concentration of intracellular ATP is measured [69] (Fig. 3). By way of illustration, if the concentration of microbial ATP is less than 0.6 pmol/ml, the sample contains up to 5·104 CFU/ml, and if above 70 pmol/ml – more than 4·106 CFU/ml.
Fig. 3. Scheme of bioluminescence determination of intracellular ATP in a biological sample.
A combined method for determination of spore contamination of food products was suggested in [70]. In the first step of the analysis, the authors used bioluminescence ATP assay in the sample: the sample was concentrated in a Filtravette™, somatic cells were disrupted by treatment with a mild lytic agent, and both somatic and free ATP was removed. To increase the intracellular ATP concentration, germination of spores was activated in nutrient medium. If the results were positive, indicating the presence of bacterial contamination, the samples were further analyzed for the presence of sporulation gene spo0A with real time PCR using SYBR Green dye.
Sensitivity of bioluminescence ATP assay can be increased not only by concentration of cells on bacterial filters or their enrichment in suitable nutrient medium. The ATP content in a cell is related to the content of ADP and AMP, which form a system of adenine nucleotides in the cell. The reversible equilibrium reaction catalyzed by adenylate kinase enzyme plays an important role in maintaining equilibrium between them in the system of adenine nucleotides in the cell: ATP + AMP = 2 ADP.
The authors in [71] suggested destroying single live cells collected on a filter following removal of extracellular and somatic ATP and using the hybrid protein adenylate kinase–polyphosphate kinase to convert the endogenous ATP in the presence of AMP and polyphosphate into double the amount of ADP and again to ATP. The method allowed detection of 75 CFU of S. aureus in 0.5 ml of milk. The sensitivity of the analysis increases 10,000-fold in the process and allows detecting 10–18 M ATP.
CELL VIABILITY ASSESSMENT IN VACCINES BASED ON LYOPHILIZED
MYCOBACTERIA CELLS
An important problem of modern microbiology is development and optimization of methods for extended storage of cultures of microorganisms, specifically lyophilization. Survival of cells following lyophilization depends on a number of parameters such as composition of the nutrient medium, initial cell concentration, availability and type of cryoprotectors, mode of lyophilization, and method of subsequent rehydration [72]. A method for rapid assessment of viability of microbial cells is required for optimization of these parameters. Traditionally plating is the method of choice, but bioluminescence analysis, especially suitable for viability testing of slowly growing organisms, can serve this function. A number of publications describe application of bioluminescence analysis for assessment the viability of lyophilized BCG vaccines [73, 74].
The viability of microbial cells in vaccines based on lyophilized cells of mycobacteria is a main indicator of their quality. When the standard microbiological method is used, the suspension of rehydrated cells is inoculated onto solid nutrient medium, and the number of formed colonies is counted following incubation. Mycobacteria are slow-growing organisms, and the incubation period could last up to five weeks. Moreover, the standard method is very variable and often hard to reproduce because mycobacterial cells are prone to aggregation. All this complicates the production and testing of vaccine preparations. The bioluminescence method provides high sensitivity, speed, accuracy, and repeatability of determination of viable cells in vaccine. This is the reason why the first studies on application of bioluminescence ATP assay for quantitative determination of mycobacteria were published already in the 1970s [75].
Extraction of intracellular ATP. The extraction of ATP is an important step for determination of intracellular ATP in cells of mycobacteria. The extraction method must release the intracellular ATP completely in one step and ensure minimal destruction of ATP during extraction. It is rather difficult to extract ATP from mycobacteria because the cell walls are very strong and “wax”-like. The problem of ATP extraction from mycobacteria has received a considerable amount of attention from many researchers.
Lysis of the cells with boiling Tris-EDTA buffer is the most common method for extracting ATP from mycobacteria. The ATP is determined with the bioluminescence method after cooling the lysate. The relationship between ATP concentration and CFU/ml for serial dilutions of a sample of Mycobacterium tuberculosis H37Rv culture was determined in [76], and high correlation was observed (r = 0.993). Nevertheless, when similar measurements were conducted for eight different samples of the same strain taken following different incubation time, the observed correlation coefficient was slightly lower (r = 0.846). The authors concluded that indeed the growth of the mycobacterial culture could be monitored with the bioluminescence method. High bioluminescence signal was observed already after 7 days of cell growth, while the signal was lacking in sterile medium.
The authors of [77] compared three methods for ATP extraction: 1) treatment with boiling Tris-EDTA buffer; 2) treatment with hot chloroform; 3) treatment with butanol at room temperature. Using fresh culture of the BCG variant Moreau strain of mycobacteria, the ATP content was determined with the ATP bioluminescence method, and number of bacteria from the mass of wet cells and concentration of viable cells (CFU/ml) in the wet culture was determined by plating serial dilutions and colony counting after 3-4 weeks of incubation. The intracellular ATP extraction with butanol was found to be the most effective. This simple and rapid technique was used by the authors in further work. Correlation between the ATP concentration and CFU/ml with correlation coefficient r = 0.99 was obtained for suspensions containing (1-50)⋅107 CFU/ml. ATP content per CFU/ml was compared for cells after 7, 9, and 12 days of growth. It was found that the total ATP content in the culture and the number of viable cells were the same for the seven- and nine-day cultures. The total ATP content was approximately 10-fold lower for the “aged” culture (12 and 16 days). However, the ATP content of a single cell depended only slightly on the culture age and varied in the range from 2.03 to 2.63 fg of ATP per CFU. This constitutes approximately (3.7-4.8)⋅10–18 mol ATP per cell. It must be noted that ATP content per cell can differ somewhat for different strains of mycobacteria because it depends on the size of the cell and its energy status. The storage of cells at different temperatures as well as lyophilization can result in decrease in intracellular ATP content.
The list of lytic agents was extended in [78]. The following extractants were used: 1) boiling 0.1 M Tris-EDTA buffer; 2) boiling 0.1 M Tris-EDTA buffer with addition of detergent – 1, 2, or 4% dodecyltrimethylammonium bromide (DTAB); 3) 10% TCA; 4) 1 M NaOH; 5) DMSO. For extraction, an aliquot of extractant (0.9 ml) was added to 0.1 ml of cell suspension, mixed quickly, and either heated, when boiling buffer was used, or incubated for a short time at room temperature. The resulting extracts were stored for a few hours on ice, and then were frozen for long-term storage. An internal standard was used during the ATP measurement (ATP solution with concentration 100 nM). Comparison of extracts showed that the NaOH released more than 90% of the ATP, but it could not be used as it was hazardous. Other extractants released around 50% of ATP, and only extraction with the use of boiling buffer with addition of 2% DTAB resulted in release of 100% of the ATP. The results, however, cast some doubt as the extraction method that uses TCA is considered as classic for intracellular ATP extraction. Moreover, the correlation between the ATP concentration and CFU for different extraction methods is not presented in the work. The extraction method described in [78] was optimized by the authors in [79], and it was shown that the addition of 10% cyclodextrin into the ATP-reagent decreased the inhibition of luciferase by the DTAB.
The most significant work on the use of the bioluminescence ATP assay for viability testing of the BCG vaccine is a study of Danish scientists [80], where not only the method for quantitative determination of the viability of the BCG vaccine based on the amount of intracellular ATP was described, but the method was thoroughly validated. They used industrial samples of lyophilized BCG vaccine (strain 1331 from the Danish collection). Separate batches of the vaccine were stored for various periods at different temperatures to obtain samples with low (storage for 28 day at 37°C), medium (storage at 2-8°C for 12-18 months), and high viability (storage at 2-8°C for 1-3 months). The lyophilized vaccine was resuspended in diluted Sauton solution SSI, and apyrase solution was added to remove extracellular ATP. Following short-term incubation, the intracellular ATP was extracted using boiling Tris-EDTA buffer and the ATP concentration was measured. Simultaneously, the number of CFU per vial was determined by plating of serial dilutions, and the dependences of the ATP content in the vial on the number of CFU per vial were constructed.
The level of ATP in the vaccine analyzed immediately after reconstruction was low. For example, the ATP content was practically independent of the CFU value with CFU changing from 105 to 106 CFU/ml and was 2-4 ng per vial (~10–18 mol ATP per CFU). This was explained by the fact that the freezing and lyophilization represent a strong stress for the cells, which results in a sharp drop of their metabolic activity and, consequently, in decrease in the level of intracellular ATP. For restoring metabolic activity of the cells, certain conditions are required after rehydration of the cells. For this purpose, the authors used preincubation for one day at 37°C in Dubos nutrient medium containing Tween and albumin additives. This rich medium restored metabolic activity of the cells, but the growth of the cells was not yet initiated. Following incubation, the ATP content increased 5-10-fold in comparison with the non-incubated cells, while the number of CFU remained the same. The average ATP content per CFU increased to ~10–17 mol. The conducted validation showed that the method is sufficiently robust to accommodate minor changes in incubation and ATP extraction conditions. Very high linear correlation (r ≥ 0.99) was observed between the ATP content within the range of 1-25 pmol ATP per vial, and the number of CFU per vial for different samples of the same batch of the vaccine with high viability. Good correlation (r = 0.928) between the ATP contents in the range 8-45 ng per vial and the CFU numbers was also observed for samples of vaccine batches with different viability. This range of ATP content corresponds to the range of viable cells, (0.7-7.6)⋅106 CFU per vial.
The Sauton solution SSI is used most often for regeneration of lyophilized mycobacterial cells [81]. According to [80], additional incubation in nutrient medium for one day was required to restore cell viability completely and to obtain good correlation between ATP content and CFU number. However, it was shown in [82] that a 30-min incubation of the lyophilized vaccine rehydrated in Sauton solution was sufficient for restoration of viability of the lyophilized mycobacteria, and satisfactory correlation between the ATP concentration and the CFU numbers was observed. Hence, the one-day incubation in the nutrient medium is not mandatory. This simplifies the process of the vaccine pretreatment significantly.
Removal of extracellular ATP. A certain part of microbial cells is destroyed during freezing and lyophilization. The intracellular ATP is released into the solution in the process. Moreover, media used for biomass production also can contain free, extracellular ATP. Hence, the removal of extracellular ATP is an important step in the pretreatment of reconstructed vaccine. In the case of vaccine, the best method for removal of ATP from solution is treatment with apyrase enzyme solution [83]. Usually the apyrase is isolated from potato tubers [83]. The reaction catalyzed by apyrase is described by the following scheme:
ATP → ADP + Pi; (1)
ADP → AMP + Pi. (2)
Neither ADP nor AMP is a substrate of firefly luciferase; hence, decomposition of ATP to ADP and AMP results in 1000-fold decrease in the emission. Two different apyrases were isolated from different potato species: apyrase A, its activity in reaction (1) being ~10-fold higher than in reaction (2); apyrase B, in contrast demonstrates 10-fold higher activity in reaction (2) rather than in reaction (1).
The use of apyrase A is necessary for the degradation of ATP, as it is exactly this enzyme that ensures fast removal of free ATP from the suspension of cells. It must be emphasized that apyrase does not penetrate inside the cells, like other proteins, and therefore it does not affect the intracellular ATP content. According to the literature, 10-min incubation of the cell suspension in dilute apyrase solution decreases the concentration of the extracellular ATP by hundred-folds [80]. In study [82], the apyrase was added into the Sauton solution. The removal of extracellular ATP occurred concurrently with the restoration of metabolic activity of the cells.
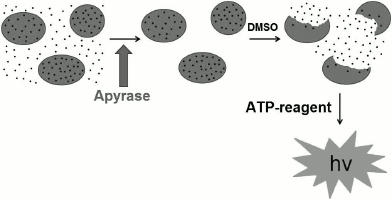
Hence, the scheme for analysis of the rehydrated BCG vaccine is as follows: in the first step the removal of extracellular ATP occurs, in the second – extraction of intracellular ATP, and in the third – determination of the intracellular ATP concentration using the bioluminescence method (Fig. 4).
Fig. 4. Scheme of bioluminescence analysis of BCG vaccine.
In conclusion, the literature data and data from our own studies on the bioluminescence method for determination of cell viability and its application in various areas of microbiology, sanitary control, and ecology were analyzed in this review. The general characteristics of the bioluminescence method for determination of extracellular and intracellular ATP, methods for determination of ATP concentration, and peculiarities of the determination of intracellular ATP in individual and mixed cultures of microorganisms were considered, and the efficiency of ATP extraction from cells using various lytic agents was analyzed. The intracellular ATP content in cells of different types was compared. The effect of different factors on the level of intracellular and extracellular ATP was shown. Different examples of the application of bioluminescence ATP assay for monitoring of food quality, hygienic state of health care facilities and industrial clean rooms, for investigation of different processes in cell systems (necrosis and apoptosis of cells, peculiarities of cell growth and degradation, effect of cellular stress on metabolic activity) were presented. The advantages of the bioluminescence method for control of the specific activity of vaccines based on live cells of lyophilized microorganisms were shown.
REFERENCES
1.Lehninger, A. (1972) Biochemistry, Worth
Publishers, New York.
2.Ugarova, N. N., Brovko, L. Yu., Trdatian, I. Yu.,
and Rainina, E. I. (1987) Bioluminescent methods of analysis in
microbiology, Appl. Biochem. Microbiol. (Moscow),
23, 11-20.
3.Ugarova, N. N. (1993) Bioanalytical applications of
firefly luciferase, Appl. Biochem. Microbiol. (Moscow),
29, 135-144.
4.McElroy, W. D. (1947) The energy source for
bioluminescence in isolated systems, Proc. Natl. Acad. Sci. USA,
33, 342-348.
5.Ando, Y., Niwa, K., Yamada, N., Enomoto, T., Irie,
T., Kubota, H., Ohmiya, Y., and Akiyama, H. (2008) Firefly
bioluminescence quantum yield and color change by pH-sensitive green
emission, Nature Photon, 2, 44-47.
6.Berthold, F., and Tarkkanen, V. (2013) Luminometer
development in the last four decades: recollections of two
entrepreneurs, Luminescence, 28, 16.
7.Lundin, A. (2000) Use of firefly luciferase in
ATP-related assays of biomass, enzymes, and metabolites, Methods
Enzymol., 305, 346-370.
8.Ugarova, N. N., Maloshenok, L. G., Moroz, N. A.,
and Lomakina, G. Yu. (2004) Reagent for determination of adenosine
5′-triphosphate, RF Patent 2268943.
9.Ugarova, N. N., Koksharov, M. I., and Lomakina, G.
Yu. (2009) Reagent for determination of adenosine
5′-triphosphate, RF Patent 2420594.
10.Koksharov, M. I., and Ugarova, N. N. (2009)
Increased thermal stability of luciferase of lighting beetles
Luciola mingrelica by random mutagenesis, Moscow Univ. Chem.
Bull., 64, 18-22.
11.Koksharov, M. I., and Ugarova, N. N. (2011)
Thermostabilization of firefly luciferase by in vivo directed
evolution, Prot. Eng. Des. Select., 24, 835-844.
12.Guardigli, M., Lundin, A., and Roda, A. (2011) in
Chemiluminescence and Bioluminescence: Past, Present and Future
(Roda, A., ed.) Royal Society of Chemistry, Cambridge, pp.
143-190.
13.Narsaiah, K., Jha, S. N., Jaiswal, P., Singh, A.
K., Gupta, M., and Bhardwaj, R. (2012) Estimation of total bacteria on
mango surface by using ATP bioluminescence, Sci. Hortic.
(Amsterdam), 146, 159-163.
14.Wang, H.-Y., Bhunia, A. K., and Lu, C. (2006) A
microfluidic flow-through device for high throughput electrical lysis
of bacterial cells based on continuous dc voltage, Biosens.
Bioelectron., 22, 582-588.
15.Romanova, N. A., Brovko, L. Yu., and Ugarova, N.
N. (1997) Comparative assessment of methods of intracellular ATP
extraction from different types of microorganisms for bioluminescent
determination of microbial cells, Appl. Biochem. Microbiol.,
33, 306-311.
16.Luo, J., Liu, X., Tian, Q.,Yue, W., Zeng, J.,
Chen, G., and Cai, X. (2009) Disposable bioluminescence-based biosensor
for detection of bacterial count in food, Anal. Biochem.,
394, 1-6.
17.Hattori, N., Sakakibara, T., Kajiyama, N.,
Igarashi, T., Maeda, M., and Murakami, S. (2003) Enhanced microbial
biomass assay using mutant luciferase resistant to benzalkonium
chloride, Anal. Biochem., 319, 287-295.
18.Rakotonirainy, M. S., Heraud, C., and Lavedrine,
B. (2003) Detection of viable fungal spores contaminant on documents
and rapid control of the effectiveness of an ethylene oxide
disinfection using ATP assay, Luminescence, 18,
113-121.
19.Hett, E. C., and Rubin, E. J. (2008) Bacterial
growth and cell division: a mycobacterial perspective, Microbiol.
Mol. Biol. Rev., 72, 126-156.
20.Hoffner, S., Jimenez-Misas, C., and Lundin, A.
(1999) Improved extraction and assay of mycobacterial ATP for rapid
drug susceptibility testing, Luminescence, 14,
255-261.
21.Kapoor, R., and Yadav, J. S. (2010) Development
of a rapid ATP bioluminescence assay for biocidal susceptibility
testing of rapidly growing mycobacteria, J. Clin. Microbiol.,
48, 3725-3728.
22.Chida, J., Yamane, K., Takei, T., and Kido, H.
(2012) An efficient extraction method for quantitation of adenosine
triphosphate in mammalian tissues and cells, Anal. Chim. Acta,
727, 8-12.
23.Kamidate, T., Yanashita, K., Tani, H., Ishida,
A., and Notani, M. (2006) Firefly bioluminescent assay of ATP in the
presence of ATP extractant by using liposomes, Anal. Chem.,
78, 337-342.
24.Ishida, A., Yoshikawa, T., Nakazawa, T., and
Kamidate, T. (2002) Enhanced firefly bioluminescence assay of ATP in
the presence of ATP extractants by using diethylaminoethyl-dextran,
Anal. Biochem., 305, 236-241.
25.Shama, G., and Malik, D. J. (2013) The uses and
abuses of rapid bioluminescence-based ATP assays, Int. J. Hyg.
Environ. Health, 216, 115-125.
26.Venkateswaran, K., Hattori, N., La Duc, M. T.,
and Kern, R. (2003) ATP as a biomarker of viable microorganisms in
clean-room facilities, J. Microbiol. Methods, 52,
367-377.
27.Hammes, F., Goldschmidt, F., Vital, M., Wang, Y.,
and Egli, T. (2010) Measurement and interpretation of microbial
adenosine triphosphate (ATP) in aquatic environments, Water
Res., 44, 3915-3923.
28.Buckstein, M. H., He, J., and Rubin, H. (2008)
Characterization of nucleotide pools as a function of physiological
state in Escherichia coli, J. Bacteriol., 190,
718-726.
29.Yaginuma, H., Kawai, S. V., Tabata, K.,
Tomiyama, K., Kakizuka, A., Komatsuzaki, T., Noji, H., and Imamura, H.
(2014) Diversity in ATP concentrations in a single bacterial cell
population revealed by quantitative single-cell imaging, Sci.
Rep., 4, 6522.
30.Hamilton, R. D., and Holm-Hansen, O. (1967)
Adenosine triphosphate content of marine bacteria, Limnol.
Oceanogr., 12, 319-324.
31.Vogel, S. J., Tank, M., and Goodyear, N. (2014)
Variation in detection limits between bacterial growth phases and
precision of an ATP bioluminescence system, Lett. Appl.
Microbiol., 58, 370-375.
32.Paciello, L., Falco, F. C., Landi, C., and
Parascandola, P. (2013) Strengths and weaknesses in the determination
of Saccharomyces cerevisiae cell viability by ATP-based
bioluminescence assay, Enzyme Microb. Technol.,
52, 157-162.
33.Hong, Y., and Brown, D. G. (2009) Variation in
bacterial ATP level and proton motive force due to adhesion to a solid
surface, Appl. Environ. Microbiol., 75, 2346-2353.
34.Ivanova, E. P., Alexeeva, Y. V., Pham, D.
K., Wright, J. P., and Nicolau, D. V. (2006) ATP level variations in
heterotrophic bacteria during attachment on hydrophilic and hydrophobic
surfaces, Int. Microbiol., 9, 37-46.
35.Jamwal, S., Midha, M. K., Verma, H. N., Basu, A.,
Rao, K. V., and Manivel, V. (2013) Characterizing virulence-specific
perturbations in the mitochondrial function of macrophages infected
with Mycobacterium tuberculosis, Sci. Rep.,
3, 1328.
36.Finger, S., Wiegand, C., Buschmann, H.-J., and
Hipler, U.-C. (2013) Antibacterial properties of
cyclodextrin-antiseptics-complexes determined by microplate laser
nephelometry and ATP bioluminescence assay, Int. J. Pharm.,
452, 188-193.
37.Aragones, L., Escude, C., Visa, P., Salvi, L.,
and Moce-Llivina, L. (2012) New insights for rapid evaluation of
bactericidal activity: a semi-automated bioluminescent ATP assay, J.
Appl. Microbiol., 113, 114-125.
38.Paloque, L., Vidal, N., Casanova, M., Dumetre,
A., Verhaeghe, P., Parzy, D., and Azas, N. (2013) A new, rapid and
sensitive bioluminescence assay for drug screening on
Leishmania, J. Microbiol. Methods, 95,
320-323.
39.Shridhar, S., Hassan, K., Sullivan, D. J., Vasta,
G. R., and Fernandez Robledo, J. A. (2013) Quantitative assessment of
the proliferation of the protozoan parasite Perkinsus marinus
using a bioluminescence assay for ATP content, Int. J. Parasitol.
Drugs Resist., 3, 85-92.
40.Gallez, F., Fadel, M., Scruel, O., Cantraine, F.,
and Courtois, P. (2000) Salivary biomass assessed by bioluminescence
ATP assay related to (bacterial and somatic) cell counts, Cell
Biochem. Funct., 18, 103-108.
41.Sanchez, M. C., Llama-Palacios, A., Marin, M. J.,
Figuero, E., Leon, R., Blanc, V., Herrera, D., and Sanz, M. (2013)
Validation of ATP bioluminescence as a tool to assess antimicrobial
effects of mouthrinses in an in vitro subgingival-biofilm model,
Med. Oral Patol. Oral Cir. Bucal., 18, e86-92.
42.Froundjian, V. G., Romanova, N. A.,
Dement’yeva, E. I., Brovko, L. Yu., Ugarova, N. N., Novikov, K.
N., Brusovanik, V. I., Kobyalko, V. O., and Meshkov, N. A. (1997) ATP
content in neutrophils and whole blood of people of Altai region
exposed to nuclear test radiation, Radiats. Biol. Radioekol.,
37, 13-19.
43.Chollet, R., Kukuczka, M., Halter, N., Romieux,
M., Marc, F., Meder, H., Beguin, V., and Ribault, S. (2008) Rapid
detection and enumeration of contaminants by ATP bioluminescence using
the Milliflex® rapid microbiology detection and enumeration system,
J. Rapid Methods Automat. Microbiol., 16, 256-272.
44.Dewi Puspita, I., Kamagata, Y., Tanaka, M.,
Asano, K., and Nakatsu, C. H. (2012) Are uncultivated bacteria really
uncultivable? Microbes Environ., 27, 356-366.
45.Lomakina, G. Y., and Ugarova, N. N. (2010)
Monitoring of the microbial contamination of air by bioluminescent
ATP-assay, Luminescence, 25, 192-193.
46.Park, C. W., Park, J.-W., Lee, S. H., and Hwang,
J. (2014) Real-time monitoring of bioaerosols via cell lysis by air ion
and ATP bioluminescence detection, Biosens. Bioelectron.,
52, 379-383.
47.Haghighi, F., Mohammadi, S. R., Mohammadi, P.,
Eskandari, M., and Hosseinkhani, S. (2012) The evaluation of Candida
albicans biofilms formation on silicone catheter, PVC and glass
coated with titanium dioxide nanoparticles by XTT method and ATPase
assay, Bratisl. Lek. Listy, 113, 707-711.
48.Kajiyama, S., Tsurumoto, T., Osaki, M.,
Yanagihara, K., and Shindo, H. (2009) Quantitative analysis of
Staphylococcus epidermidis biofilm on the surface of
biomaterial, J. Orthop. Sci., 14, 769-775.
49.Oulahal-Lagsir, N., Martial-Gros, A., Bonneau,
M., and Blum, L. J. (2000) Ultrasonic methodology coupled to ATP
bioluminescence for the non-invasive detection of fouling in food
processing equipment – validation and application to a dairy
factory, J. Appl. Microbiol., 89, 433-441.
50.Sule, P., Wadhawan, T., Carr, N. J., Horne, S.
M., Wolfe, A. J., and Pruβ, B. M. (2009) A combination of assays
reveals biomass differences in biofilms formed by Escherichia
coli mutants, Lett. Appl. Microbiol., 49,
299-304.
51.Romanova, N. A., Froundjian, V. G., and Ugarova,
N. N. (1994) in Bioluminescence and Chemiluminescence. Fundamental
and Applied Aspects. Proc. 8th Int. Symp. (Campbell, A. K., Kricka,
L. J., and Stanley, P. E., eds.) John Wiley & Sons, Chichester, pp.
434-437.
52.Hunter, D. M., Leskinen, S. D., Magana, S.,
Schlemmer, S. M., and Lim, D. V. (2011) Dead-end ultrafiltration
concentration and IMS/ATP-bioluminescence detection of Escherichia
coli O157:H7 in recreational water and produce wash, J.
Microbiol. Methods, 87, 338-342.
53.Minikh, O. A., Brovko, L. Yu., Griffiths, M. W.,
and Ugarova, N. N. (2010) Specific determination of E. coli
B using bacteriophage T4, nanofilters, and ATP assay, Moscow
Univ. Chem. Bull., 65, 202-205.
54.Romanova, N. A., Brovko, L. Yu., Moore, L.,
Pometun, E. V., Savitsky, A. P., and Ugarova, N. N. (2003) Assessment
of photodynamic destruction of Escherichia coli O157:H7 and
Listeria monocytogenes by using ATP bioluminescence, Appl.
Environ. Microbiol., 69, 6393-6398.
55.Romanova, N. A., Pometun, E. V., Savitski, A. P.,
and Ugarova, N. N. (2000) Bioluminescent determination of
adenosine-5′-triphosphate effuent from yeast cells because of
their photodynamic injuries, Vestnik Mosk. Univ., Khimiya,
41, 404-407.
56.Costa, P. D., Andrade, N. J., Brandao, S. C. C.,
Passos, F. J. V., and de Soares, N. F. F. (2006) ATP-bioluminescence
assay as an alternative for hygiene-monitoring procedures of stainless
steel milk contact surfaces, Brazil. J. Microbiol., 37,
345-349.
57.Larson, E., Aiello, A., Gomez-Duarte, C., Lin,
S., Lee, L., Della-Latta, P., and Lindhardt, C. (2003) Bioluminescence
ATP monitoring as a surrogate marker for microbial load on hands and
surfaces in the home, Food Microbiol., 20, 735-739.
58.Aycicek, H., Oguz, U., and Karci, K. (2006)
Comparison of results of ATP bioluminescence and traditional hygiene
swabbing methods for the determination of surface cleanliness at a
hospital kitchen, Int. J. Hyg. Environ. Health, 209,
203-206.
59.Griffith, C. J., Cooper, R. A., Gilmore, J.,
Davies, C., and Lewis, M. (2000) An evaluation of hospital cleaning
regimes and standards, J. Hosp. Infect., 45, 19-28.
60.Obee, P. C., Griffith, C. J., Cooper, R. A.,
Cooke, R. P., Bennion, N. E., and Lewis, M. (2005) Real-time monitoring
in managing the decontamination of flexible gastrointestinal
endoscopes, Am. J. Infect. Control, 33, 202-206.
61.Mempin, R., Tran, H., Chen, C., Gong, H., Kim Ho,
K., and Lu, S. (2013) Release of extracellular ATP by bacteria during
growth, BMC Microbiol., 13, 301.
62.Valat, C., Champiat, D., N’Guyen, T. T.,
Loiseau, G., Raimbault, M., and Montet, D. (2003) Use of ATP
bioluminescence to determine the bacterial sensitivity threshold to a
bacteriocin, Luminescence, 18, 254-258.
63.Trudil, D. P., Tartal, J., and Trudil, C. (2002)
in Bioluminescence and Chemiluminescence: Progress and Current
Applications (Stanley, P. E., and Kricka, L. J., eds.) World
Scientific Publishing Co., Singapore, pp. 369-372.
64.Thore, A., Ansehn, S., Lundin, A., and Bergman,
S. (1975) Detection of bacteria by luciferase assay of adenosine
triphosphate, J. Clin. Microbiol., 1, 1-8.
65.Frundzhyan, V., and Ugarova, N. (2007)
Bioluminescent assay of total bacterial contamination of drinking
water, Luminescence, 22, 241-244.
66.Froundjian, V. G., Ugarova, N. N., and Trudil, D.
P. (2002) in Bioluminescence and Chemiluminescence. Progress and
Current Applications (Campbell, A. K., Kricka, L. J., and Stanley,
P. E., eds.) John Wiley and Sons, Chichester, pp. 475-478.
67.Frundzhyan, V. G., Ugarova, N. N., Gabriyelyan,
N. I., Aref’eva, L. I., and Preobrazhenskaya, T. B. (2005) in
13th Int. Symp. Biolumin. Chemilumin. Prog. Perspect. (Kricka,
L. J., and Stanley, P. E., eds.) John Wiley and Sons, Chichester, pp.
389-392.
68.Shinozaki, Y., Sato, J., Igarashi, T., Suzuki,
S., Nishimoto, K., and Harada, Y. (2013) Evaluation of an improved
bioluminescence assay for the detection of bacteria in soy milk,
Biocontr. Sci., 18, 1-7.
69.Frundzhyan, V. G., and Ugarova, N. N (2005) Rapid
method for determining the number of mesophilic aerobic and facultative
anaerobic microorganisms in raw milk, Pererabotka Moloka,
No. 3, 14-15.
70.Ratphitagsanti, W., Park, E., Lee, C. S., Amos
Wu, R.-Y., and Lee, J. (2012) High-throughput detection of spore
contamination in food packages and food powders using tiered approach
of ATP bioluminescence and real-time PCR, LWT–Food Sci.
Technol., 46, 341-348.
71.Satoh, T., Kato, J., Takiguchi, N., Ohtake, H.,
and Kuroda, A. (2004) ATP amplification for ultrasensitive
bioluminescence assay: detection of a single bacterial cell, Biosci.
Biotechnol. Biochem., 68, 1216-1220.
72.Morgan, C. A., Herman, N., White, P. A., and
Vesey, G. (2006) Preservation of microorganisms by drying; a review,
J. Microbiol. Methods, 66, 183-193.
73.Kolibab, K., Derrick, S. C., Jacobs, W. R., and
Morris, S. L. (2012) Characterization of an intracellular ATP assay for
evaluating the viability of live attenuated mycobacterial vaccine
preparations, J. Microbiol. Methods, 90, 245-249.
74.Jensen, S. E., Hubrechts, P., Klein, B. M., and
Haslov, K. R. (2008) Development and validation of an ATP method for
rapid estimation of viable units in lyophilised BCG Danish 1331
vaccine, Biologicals, 36, 308-314.
75.Crispen, R. G. (1971) Rapid testing of freeze
dried BCG vaccine for stability and viability, Symp. Ser.
Immunobiol. Stand., 17, 205-210.
76.Beckers, B., Lang, H. R., Schimke, D., and
Lammers, A. (1985) Evaluation of a bioluminescence assay for rapid
antimicrobial susceptibility testing of mycobacteria, Eur. J. Clin.
Microbiol., 4, 556-561.
77.Janaszek, W., Aleksandrowicz, J., and Sitkiewicz,
D. (1987) The use of the firefly bioluminescent reaction for the rapid
detection and counting of mycobacterium BCG, J. Biol. Stand.,
15, 11-16.
78.Hoffner, S. E., Jimenez-Misas, C. A., and Lundin,
A. (1994) Improved extraction of mycobacterial ATP, in
Bioluminescence and Chemiluminescence. Fundamental and Applied
Aspects (Campbell, A. K., Kricka, L. J., and Stanley, P. E., eds.)
John Wiley and Sons, Chichester, pp. 442-445.
79.Hoffner, S., Jimenez-Misas, C., and Lundin, A.
(1987) Improved extraction and assay of mycobacterial ATP for rapid
drug susceptibility testing, Luminescence, 14,
255-261.
80.Jensen, S. E., Hubrechts, P., Klein, B. M., and
Haslow, K. R. (2008) Development and validation of an ATP method for
rapid estimation of viable units in lyophilized BCG Danish 1331
vaccine, Biologicals, 36, 308-314.
81.Deinse, F. V., and Senechal, F. (1950) BCG on
Sauton medium. Effect of a long series of subcultures on the
morphological and biological properties of BCG cultures, Bull.
World Health Org., 2, 347-354.
82.Kolibab, K., Derrick, S. C., Jacobs, W. R., and
Morris, S. L. (2012) Characterization of an intracellular ATP assay for
evaluating the viability of live attenuated mycobacterial vaccine
preparations, J. Microbiol. Meth., 90, 245-249.
83.Kettlun, A. M., Uribe, L., Calvo, V., Silva, S.,
Rivera, J., Mancilla, M., Valenzuela, M. A., and Traverso-Cori, A.
(1982) Properties of two apyrases from Solanum tuberosum,
Phytochemistry, 21, 551-558.