Tissue-Specific Rhamnogalacturonan I Forms the Gel with Hyperelastic Properties
P. V. Mikshina*, A. A. Petrova, D. A. Faizullin, Yu. F. Zuev, and T. A. Gorshkova
Kazan Institute of Biochemistry and Biophysics, Kazan Scientific Center, Russian Academy of Sciences, 420111 Kazan, Russia; fax: +7 (843) 292-7347; E-mail: p.mikshina@gmail.com* To whom correspondence should be addressed.
Received February 8, 2015; Revision received February 27, 2015
Rhamnogalacturonans I are complex pectin polysaccharides extremely variable in structure and properties and widely represented in various sources. The complexity and diversity of the structure of rhamnogalacturonans I are the reasons for the limited information about the properties and supramolecular organization of these polysaccharides, including the relationship between these parameters and the functions of rhamnogalacturonans I in plant cells. In the present work, on the example of rhamnogalacturonan I from flax gelatinous fibers, the ability of this type of pectic polysaccharides to form at physiological concentrations hydrogels with hyperelastic properties was revealed for the first time. According to IR spectroscopy, water molecules are more tightly retained in the gelling rhamnogalacturonan I from flax fiber cell wall in comparison with the non-gelling rhamnogalacturonan I from primary cell wall of potato. With increase in strength of water binding by rhamnogalacturonan I, there is an increase in elastic modulus and decrease in Poisson’s ratio of gel formed by this polysaccharide. The model of hyperelastic rhamnogalacturonan I capture by laterally interacting cellulose microfibrils, constructed using the finite element method, confirmed the suitability of rhamnogalacturonan I gel with the established properties for the function in the gelatinous cell wall, allowing consideration of this tissue- and stage-specific pectic polysaccharide as an important factor in creation of gelatinous fiber contractility.
KEY WORDS: gelatinous fibers, polysaccharides, rhamnogalacturonan I, water sorption, gel, elastoplastic propertiesDOI: 10.1134/S000629791507010X
Abbreviations: RGf, flax fiber rhamnogalacturonan I before incorporation into the cell wall; RGfcw, rhamnogalacturonan I of flax fiber cell wall; RGp, rhamnogalacturonan I of potato primary cell wall; RH, relative humidity.
Plant cell wall is the source of the great number of carbohydrate
structures that are built mainly on the base of ten types of
polysaccharide backbones. The diversity of cell wall polysaccharides,
including polysaccharides with the same type of backbone apparently
correlates with their “functional fitting” [1]. However, the criteria defining cell wall
polysaccharide “functional fitting” have not yet been
determined. The ability to form supramolecular structures with certain
physical, chemical, and mechanical properties could serve as one of
these criteria. The result of this ability, in particular, is the
formation of various types of gels by individual polysaccharides. The
way of gelation and the properties of the formed gel can differ not
only for structurally distant polysaccharides, but even among
polysaccharides formed based on the same type of backbone [2-5].
Pectins including polygalacturonic acid and rhamnogalacturonan I and II take a key place among the well-characterized gel-forming polysaccharides of higher plants. The ability of pectin to form gel is mainly related with the presence of high- and low-methoxylated polygalacturonic acid in their structure [6-8]. It is considered that rhamnogalacturonans of higher plants, in the absence of homogalacturonan structure fragments, do not form gels [9], although for some of these polysaccharides the ability to aggregate due to modifying groups and/or neutral side chains was demonstrated [10-13]. Unusual associates of pectin molecules that do not contain polygalacturonan fragments were characterized for tissue- and stage-specific flax fiber rhamnogalacturonan I present at the stage of tertiary cell wall formation. Such cell walls are typical for many plant fibers and named as “gelatinous” due to their gel-like image. A peculiarity of the spatial organization of the cell wall rhamnogalacturonan I associates of flax fiber is that the charged polysaccharide backbone, built of alternating dimers [→4)-α-D-GalpA-(1→2)-α-L-Rhap(1→], is located at the surface, and the neutral galactan chains, interacting with each other, form the core and hold the molecules in associates [13]. In gelatinous layers of the cell wall, such associates are “entrapped” between laterally interacting cellulose microfibrils. This causes the effective microfibril tension and as a result the appearance of contractile properties that are characteristic for fibers [14-18]. Localization and function of rhamnogalacturonan I under pressure, occurring as a result of cellulose microfibril interaction, suggest that this polysaccharide has certain elastoplastic properties. Due to the special type of secretion of Golgi vesicle content during tertiary cell wall formation [19], rhamnogalacturonan I can also be isolated before incorporation into the cell wall [20].
The aim of this work was to establish the physicochemical and mechanical features of tissue-specific rhamnogalacturonan I, determining the polysaccharide “functional fitting” as an element of tertiary cell wall of gelatinous fibers. For this, we compared the characteristics of the polysaccharide and high molecular weight rhamnogalacturonan I isolated before incorporation into the gelatinous cell wall of flax fibers, as well as rhamnogalacturonan I of thin primary cell walls of potato that does not form associates of a particular type. The ability of the analyzed rhamnogalacturonans I to gel is characterized including features of sorption properties of these polysaccharides with respect to water as a key factor in polysaccharide hydrogel formation.
MATERIALS AND METHODS
Plant material. Flax (Linum usitatissimum L., var. Mogilevski) plants from the collection of the All-Russian Flax Research Institute (Torzhok, Russia) were used. The plants were grown in natural condition, in boxes with a 50-cm soil layer, in the open air, under natural light, and with daily watering. Stem peels (fibrous parts) of 10-cm-long segments located below the snap point [21] were taken at the period of rapid growth (40 days after sowing) and used for rhamnogalacturonan I isolation. Fibers isolated from mature flax stems (100 days after the sowing) were used for extraction of rhamnogalacturonan I from the cell wall.
Rhamnogalacturonan I isolation and purification. Flax fiber rhamnogalacturonan I, which is not incorporated in the cell wall (RGf), was isolated as a high molecular weight polymer from the supernatant obtained after tissue homogenization in buffer (10 mM NaOAc, pH 5.0, 10 ml per g of tissue). Polymers present in the clarified homogenate were filtered and precipitated with ethanol (final concentration 80%); the pellet was dried, redissolved, and chromatographed on a column with Sepharose CL-4B (12 × 400 mm; Pharmacia, Sweden). The eluent was 0.01 M pyridine/acetic acid solution, pH 4.5, flow rate 0.25 ml/min, volume of collected fractions 1 ml. Fractions corresponding to 700-2000 kDa were taken for analysis [20, 22]. Sugar content in each fraction was measured by the phenol–sulfuric acid assay [23]. Pullulans with Mw 1660, 380, 100, and 48 kDa (Showa Denko, Japan) with low index of polydispersity (1.09-1.19) were used to calibrate the column.
Rhamnogalacturonan I of gelatinous cell wall (RGfcw) was isolated from flax fibers and pre-washed by 1% ammonium oxalate and 4 M KOH according to the procedure developed by Gurjanov et al. [22]. For total cellulose microfibril destruction, solution of 8% LiCl (Merck, Germany) in N,N-dimethylacetamide (AppliChem, Germany) dehydrated using molecular sieves and cellulase (Cellusoft-L; Novo Nordisk Bioindustrrie SA, France; 750 EGU/G) were used. Rhamnogalacturonan I that is the bulk of the polymer fractions was purified by gel filtration on a Sepharose CL-4B column (12 × 400 mm; Pharmacia) under the same conditions that were used for the buffer-extraction polymer separation. Fractions corresponding to 100-400 kDa were collected for analysis [22, 24].
Commercial (Megazyme, Ireland) rhamnogalacturonan I (RGp) from potato primary cell wall was purified from low molecular mass contaminants using Sephadex G-25 column (Pharmacia).
Rhamnogalacturonan I gel preparation. Three approaches were used for preparation of gels from rhamnogalacturonans I of flax fibers and potato [25]: 1) the dried polysaccharide sample was saturated by water (polysaccharide–water ratio 4 : 1 with subsequent increase in the proportion of water in the sample); 2) polysaccharide solution was heated on a water bath at 90°C for 5 min and cooled to room temperature; 3) polysaccharide solution was heated in a microwave oven at 584 W for 1 min, then filled into blisters with cell diameter of 10.3 mm, and cooled at room temperature until solidification.
Experiments on uniaxial compression. To determine Young’s modulus and Poisson’s ratio, the gels were subjected to uniaxial compression (Fig. 1). Measurements were performed by micrometer with a digital computing device (MCC-25-0.001; Russia), evaluating the change in the height of the gel on application of pressure. Various weights (1, 2, 5, 10, 20, 100, and 200 g) were used as the source of pressure. Before measurement, the system was kept for 30 s in the pressure-free state (recovery of initial gel block height) (Fig. 1a). After placing a weight on the gel block, the system was held for 20 s to come to equilibrium; then the magnitude of steady load was determined (Fig. 1b). The force applied to the gel block was calculated as the product of the weight mass and free fall acceleration product, and the pressure was calculated from the force magnitude and the gel block surface area.
Fig. 1. Equipment for experiments on uniaxial compression. Gel block of rhamnogalacturonan I from flax fiber cell wall with corresponding micrometer readings before (a) and after (b) pressuring.
Young’s modulus and Poisson’s ratio were calculated according to formulas (1) and (2), respectively:
E = Fl/SΔl, (1)
where E is Young’s modulus, F the force applied to the gel block, S gel surface area on which the force is distributed, l gel block height, Δl module of gel block height change as a result of elastic deformation (measured in the same units as the length l);
µ = │ε′/ε│, (2)
where µ is Poisson’s ratio, ε′ gel block deformation in the transverse direction, ε gel block deformation in longitudinal direction.
IR spectroscopy. IR spectra were obtained using IR Affinity 1 spectrophotometer (Shimadzu, Japan) in the range of 700-4000 cm–1 with resolution of 4 cm–1; 128 spectral scans were averaged. Rhamnogalacturonan I samples were dissolved in water at concentration 10 mg/ml, and 5 µl of a solution was loaded on the surface of the germanium crystal of a total internal reflection accessory MIRacle ATR and dried. The film was placed in a sealed chamber through which the air stream of controlled H2O or D2O relative humidity (RH) was passed. The temperature during the sample preparation and measurement was 25°C.
To determine rhamnogalacturonan I sorption properties in relation to water, IR spectra of films at two RH values were taken: close to saturation (99%) and close to zero (over phosphorus pentoxide). The amount of tightly bound water was determined by the intensity of the residual band of deformation oscillations of OH-bonds in water at 1640 cm–1. For this purpose, the spectra of the individual components corresponding to water and polysaccharide absorption bands were resolved. The amount of weakly bound water was determined by the 1640 cm–1 absorption band area obtained by subtracting the “dry” sample spectrum from the sample spectrum at saturating RH.
Accessibility of polysaccharide OH groups and hydration water for the solvent was evaluated by deuterium exchange rate. Films of the samples were moistened to saturation in H2O vapors, and then sample was placed in saturating D2O vapors. Spectra were recorded during the time required for registration of the total kinetic curve of H-D exchange.
Computer modeling. Rhamnogalacturonan I cellulose microfibril entrapping was modeled by a finite element method using the AnsysWorkbench 15.0 software system. During model construction, a two-dimensional structure was designed, including rhamnogalacturonan I, located between two cellulose microfibrils, one of which was under pressure corresponding to cell turgor pressure (0.3 MPa) [26]. Since the cellulose microfibrils have linear elasticity, the literature characteristics for flax cellulose were used in the course of a given construction modeling: microfibril length 6 µm, diameter 3 nm [27, 28], Young’s modulus 130 GPa [29], Poisson’s ratio 0.4 [30]. Necessary parameters for rhamnogalacturonan I modeling (Young’s modulus, Poisson’s ratio, the Mooney–Rivlin constants) were obtained from uniaxial compression experiments; rhamnogalacturonan associate diameter – 40 nm [13].
To determine the C01 and C10 Mooney–Rivlin constants, formulae (3) and (4) were used:
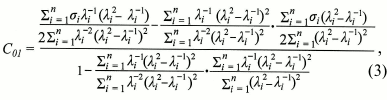
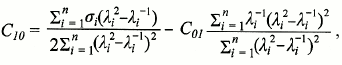 (4)
(4)where σ is experimental strain generated in the sample (defined by formula (5)), λ the degree of the sample deformation (λ = h/h0), h0 the initial height of the gel block before pressuring, h the height of the gel block after pressuring, n the number of experimental points;
where Felast is an elastic force arising during the gel deformation (defined by formula (6)), S gel block surface area through which the force is distributed;
where k is gel stiffness (defined by formula (7)), Δl deformation value;
where E is Young’s modulus, S gel block surface area through which the force is distributed, L0 gel block height.
RESULTS
Establishing rhamnogalacturonan I gel-forming ability. On saturation of flax fiber and potato rhamnogalacturonans I with water in the ratio 4 : 1, all polysaccharides absorbed water completely without dissolution. Addition of more water to samples resulted in rhamnogalacturonan dissolution; in this case, gels were not formed even after keeping a sample at 4°C, regardless of the polysaccharide concentration. However, flax and potato rhamnogalacturonan I solutions obtained through water saturation were visually different: RGp solution was a liquid regardless of the concentration, but RGf formed a viscous syrup at water ratio 1 : 2.5, and RGfcw formed gel-like grainy pastes. The polysaccharides behaved similarly during water bath heating.
After electromagnetic radiation exposure in a microwave oven, the flax fiber rhamnogalacturonans I formed gels at room temperature, the minimal polysaccharide concentration required for gel formation being 4%. Visually, the gel formed by RGf was softer than the gel from RGfcw. The state of potato rhamnogalacturonan I after microwave heating did not change. This polysaccharide did not form gel either after subsequent sample cooling and keeping at 4°C, or addition of Ca2+ (0.62 mM) to the polysaccharide solution [31], as well as maximal increase in the concentration by drying. In all cases, for RGp only two states were revealed – a viscous solution or a film.
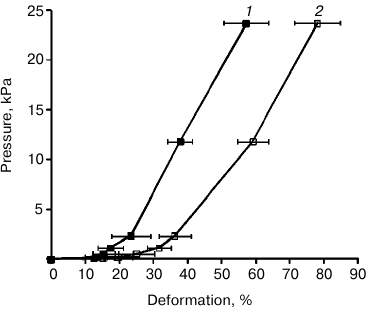
Elastoplastic properties of rhamnogalacturonan I gels of flax fibers. To establish elastoplastic properties of flax fiber rhamnogalacturonan I, an uniaxial compression experiment was performed. The parameters of the factors applied on RGf and RGfcw gels are presented in Table 1. Based on the results of the experiment, the dependence of the degree of gel deformation on the magnitude of the applied pressure was established, as well as the Young’s modulus (a physical quantity that characterizes the properties of the material to resist stress/strain during elastic deformation) and Poisson’s ratio (the ratio of the relative transverse compression to the relative longitudinal extension) were calculated.
Table 1. Deformation characteristics of
rhamnogalacturonan I gels of flax fibers (RGf and RGfcw) in experiments
on uniaxial compression
Stress–strain curves for flax fiber rhamnogalacturonan I gel have concave shape (Fig. 2) typical for water gels, in particular for gels based on hyaluronic acid [32]. This curve is characterized for materials that are destroyed before beginning to flow under pressure, such as elastomers.
Fig. 2. Stress–strain curves for gel blocks of RGfcw (1) and RGf (2) samples.
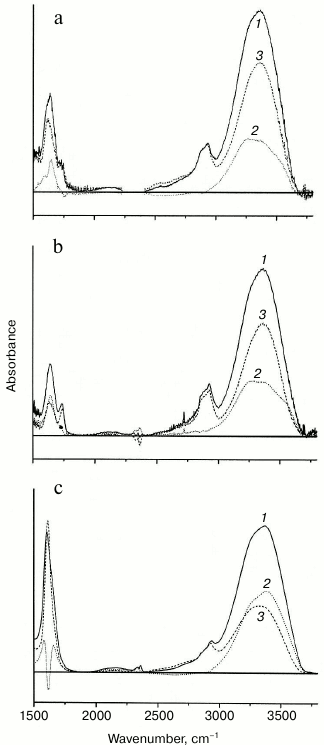
Water sorption by rhamnogalacturonans I. IR spectroscopy was used to compare rhamnogalacturonans I with different gel-forming capability according to their water sorption characteristics. Figure 3 shows the spectra of the dry and wet films of RGf, RGfcw, and RGp samples. The spectra of adsorbed water were obtained by determining the difference between the spectra of dry and wet samples. The form and intensity of the absorption bands in the area of valence vibrations of the adsorbed water can be considered as the sum of at least three components with maximum frequency in area of 3240, 3400, and 3560 cm–1 (Fig. 3). The positions of the maximums are due to differences in the strength of hydrogen bonds formed by water molecules in the polysaccharide matrix [34]. In the RGf and RGfcw samples, the relative intensity and number of spectral components of weakly removable water are almost identical. The intensity of the RGp water absorption is significantly higher, and the band form is distorted by extended absorption in the low-frequency region up to 2500 cm–1, which is characteristic for water adsorbed on charged acidic groups [35]. Intense bands at frequencies 1608 and 1410 cm–1 indicate high content of ionized acid residues in RGp. At the same time, the amount of tightly bound water, determined by the residual absorbance at 1640 cm–1 in the dry sample spectra, was less in RGp than in RGf and RGfcw.
Fig. 3. IR spectra of films of samples: a) RGfcw; b) RGf; c) RGp. 1) Wet sample; 2) dry sample; 3) difference between spectra of wet and dry samples.
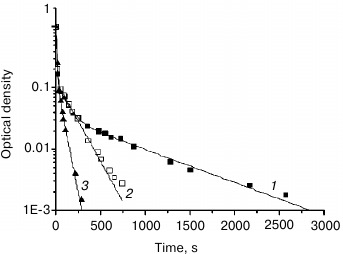
Fig. 4. Normalized dependence of H-D exchange kinetics according to the band at 3400 cm–1 for films of samples: RGfcw (1), RGf (2), RGp (3). The approximations by exponential functions are designated by solid lines (see Table 2).
Table 2. Approximation parameters of the
kinetic curves of H-D exchange by exponential components. Band of
valence vibrations of OH groups (3400 cm–1) in
RGf, RGfcw, and RGp samples normalized to the band maximum
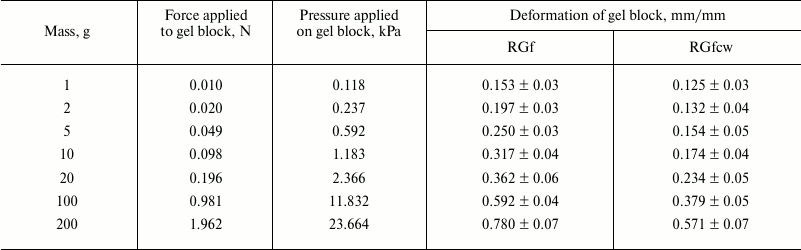
The number and relative weights of the components for flax fiber and potato rhamnogalacturonans I are significantly different. So, RGf and RGp approximation parameters of the H-D exchange kinetic curves are described by two exponential components, while RGfcw are describe by three. The overlap of the absorption bands of OH groups of water and polysaccharides in the 3400 cm–1 region makes difficult component classification. However, the absorption in the 1640-cm–1 region is due only to the contribution of water absorption; the exchange kinetics recorded at this band has only one component with time constant close to the lowest value given in Table 2. Thus, the most rapid proton exchange reaction occurs in the weakly bound water molecules, and probably in part of the polysaccharide hydrated OH groups that are not included in a system of hydrogen bonds. Slower exchange is characteristic for polysaccharide and water OH groups involved in hydrogen bond formation. The third, largest, value of time constant is observed only in the RGfcw sample and describes proton replacement in very slowly exchangeable OH groups.
Approximation parameters of the corresponding kinetic curve for RGp indicate weak intermolecular binding of OH groups in this polysaccharide, while in RGf about a third of OH groups, and in RGfcw about half of OH groups were apparently involved in hydrogen bond formation, and exchanged at medium rate (Table 2). More complex form and higher duration of deuterium exchange kinetics in RGfcw (Fig. 4 and Table 2) indicates the presence of very slowly exchangeable protons, along with quickly and slowly exchangeable in these conditions protons of OH groups of the polysaccharide. Their number, estimated by comparing the intensity of the OH group absorption bands in the initially dry and wet samples with high degree of deuterium exchange, indicates that about 10% of all OH groups in RGfcw are sterically inaccessible for the solvent.
Thus, the IR spectroscopy shows that in flax fiber gel-forming rhamnogalacturonan I samples, water molecules that are more tightly retained by polysaccharides as compared with a non-gel-forming rhamnogalacturonan I of potato primary cell wall are present. The increase in water retention by rhamnogalacturonan I in this case is connected with increase in strength of the formed gel and might be associated with the presence of densely packed areas in the sample structure.
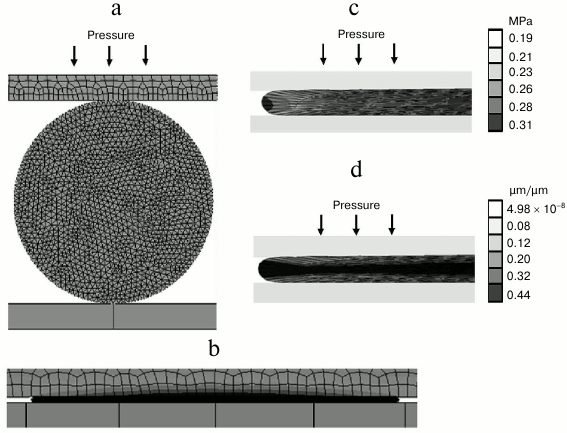
Modeling of entrapping of rhamnogalacturonan I by microfibrils of cellulose. To check the suitability of the rhamnogalacturonan I gel with defined properties for functioning in a gelatinous cell wall, a model was built which included the polysaccharide being sandwiched between two cellulose microfibrils (Fig. 5a). One of the microfibrils was put under pressure corresponding to the cell turgor pressure (0.3 MPa) [26]. Due to the fact that polysaccharide undergoes significant deformation under such pressure, the Mooney–Rivlin model for hyperelastic materials was used during simulation [37]. The Mooney–Rivlin C01 and C10 constants needed for modeling were calculated using formulas (3) and (4) and were 395.8 and 2751.2 Pa, respectively, for seven points of the experimental data.
Fig. 5. Modeling of deformation of rhamnogalacturonan I from flax fiber cell wall on positioning between two cellulose microfibrils, one of which is under pressure corresponding to the cell turgor pressure. a) Finite-element model of RGfcw; b) finite element model of RGfcw after pressuring; c) stress in longitudinal direction for RGfcw (MPa); d) RGfcw deformation in the longitudinal direction (µm/µm). Elastoplastic properties of RGfcw used in the modeling were determined in the experiments on uniaxial compression.
DISCUSSION
Rhamnogalacturonans I, in terms of structures, are the most complexly organized class of plant polysaccharides, and they are widely represented in various sources and cell wall types [15, 38-42]. The described structures of supramolecular rhamnogalacturonan I are formed mainly with the involvement of modifying groups and/or neutral side chains [10-12]. The principal difference of flax gelatinous fiber rhamnogalacturonan I from rhamnogalacturonans I of other cell wall types is the ability to self-association, because of which supramolecular structure of a special type is formed: the backbone is located on the surface, and the interacting galactan chains form the associate core [13]. Such spatial structure of rhamnogalacturonan I from gelatinous fibers provides: (i) the ability of the polysaccharide to maintain hydrodynamic volume after a decrease in molecular mass by enzymatic digestion (side chains, not crucial for the associate maintenance, undergo hydrolysis); (ii) the presence of a charged surface (GalpA carboxyl groups).
It was shown that at concentrations above 0.08%, flax fiber rhamnogalacturonan I associates are able to aggregate to larger particles (142-192 nm) [13], and at a concentration of ~4% this polysaccharide forms a hydrogel with hyperelastic properties, being classified as strong physical gels [25, 43]. The ability to form such gel distinguishes fibers of rhamnogalacturonan I from other rhamnogalacturonans I, including polysaccharide from primary cell wall (similar in composition, but functionally distinct) “gluing” neighboring cells to each other. In this regard, rhamnogalacturonan I gel-forming ability can be considered as one of the factors of polysaccharide “functional fitting” in plant fibers.
The mechanism of physical hydrogel formation is based on the formation of a spatial network of interlacing polymer chains with the participation of water molecules as a solvent. In vitro, gels from flax fiber rhamnogalacturonans I were obtained only when the polysaccharides were exposed to electromagnetic radiation in the decimeter range (microwave). Microwave radiation is widely used for promoting chemical reactions in organic chemistry. It is believed that the main factor of microwave radiation exposure, in these cases, is the thermal effect due to motion of water molecules and polar groups of organic compounds [44, 45]. In the flax fiber rhamnogalacturonan I samples, the presence of water molecules, which are more tightly retained in comparison with water molecules in the potato rhamnogalacturonan I sample, can provide polysaccharide gel-forming ability. Moreover, the increase in the binding force of water is associated with the increase in elastic modulus and decrease in Poisson’s ratio of the gel formed by rhamnogalacturonan I. It is known that rhamnogalacturonans I with galactan side chains have higher water-retaining capacity than those with arabinan ones [46, 47]. The presence of strongly bound water in the sample of flax fiber cell wall rhamnogalacturonan I could contribute to the formation of locally ordered zones between polysaccharide galactan chains involved in gel formation. This hypothesis is confirmed by the IR spectroscopy results, demonstrating increase in the degree of structuring of rhamnogalacturonan I of flax fiber cell wall, which forms the most dense gel.
The elastoplastic properties of flax fiber rhamnogalacturonan I gel are different from the properties of classical pectin gel from polygalacturonic acid. Values of Young’s modulus of the gel formed of polygalacturonan vary depending on the temperature and the degree of polysaccharide methoxylation, but do not exceed 10 kPa [2, 3, 48, 49]. At the same time, Poisson’s ratio of the polygalacturonan gels is close to that determined for flax fiber rhamnogalacturonan I one and is 0.4-0.5 [50]. These differences demonstrate that rhamnogalacturonan I gel is more suitable (more elastic and incompressible in comparison with polygalacturonan) for the key role of matrix polysaccharide functioning under high-pressure conditions.
This work was supported in part by the Russian Foundation for Basic Research (project No. 14-04-31462 and 15-04-02560).
REFERENCES
1.Gorshkova, T. A., Kozlova, L. V., and Mikshina, P.
V. (2013) Spatial structure of plant cell wall polysaccharides and its
functional significance, Biochemistry (Moscow), 78, 836-853.
2.Vithanage, C. R., Grimson, M. J., Wills, P. R.,
Harrison, P., and Smith, B. G. (2010) Rheological and structural
properties of high-methoxyl esterified, low-methoxyl esterified and
low-methoxyl-amidated pectin gels, J. Texture Studies,
41, 899-927.
3.Ngouemazong, E. D., Jolie, R. P., Cardinaels, R.,
Van Loey, A., Moldenaers, P., and Hendrickx, M. (2012) Stiffness of
Ca2+-pectin gels: combined effect of degree and pattern of
methyl esterification for various Ca2+ concentrations,
Carbohydr. Res., 348, 69-76.
4.Mitsumata, T., Honda, A., Kanazawa, H., and Kawai,
M. (2012) Magnetically tunable elasticity for magnetic hydrogels
consisting of carrageenan and carbonyl iron particles, J. Phys.
Chem., 116, 12341-12348.
5.Brenner, T., Wang, Z., Achayuthakan, P., Nakajima,
T., and Nishinari, K. (2013) Rheology and synergy of
κ-carrageenan/locust bean gum/konjac glucomannan gels,
Carbohydr. Polymers, 98, 754-760.
6.Grant, G. T., Morris, E. R., Rees, D. A., Smith, P.
J. C., and Thom, D. (1973) Biological interactions between
polysaccharides and divalent cations: the egg-box model, FEBS
Lett., 32, 195-198.
7.Fishman, M. L., and Cooke, P. H. (2009) The
structure of high-methoxyl sugar acid gels of citrus pectin as
determined by AFM, Carbohydr. Res., 344, 1792-1797.
8.Yapo, B. M., and Gnakri, D. (2014) Pectic
polysaccharides and their functional properties,
Polysaccharides; DOI: 10.1007/978-3-319-03751-6-62-1.
9.Yapo, B. M. (2011) Rhamnogalacturonan-I: a
structurally puzzling and functionally versatile polysaccharide from
plant cell walls and mucilages, Polymer Rev., 51,
391-413.
10.Oosterveld, A., Beldman, G., Schols, H. A., and
Voragen, A. G. (2000) Characterization of arabinose and ferulic acid
rich pectic polysaccharides and hemicelluloses from sugar beet pulp,
Carbohydr. Res., 328, 185-197.
11.Sengkhamparn, N., Sagis, L. M. C., de Vries, R.,
Schols, H. A., Sajjaanantakul, T., and Voragen, A. G. J. (2010) Food
Hydrocolloids, 24, 35-41.
12.Morris, G. A., Ralet, M.-C., Bonnin, E.,
Thibault, J.-F., and Harding, S. E. (2010) Physical characterization of
the rhamnogalacturonan and homogalacturonan fractions of sugar beet
(Beta vulgaris) pectin, Carbohydr. Polymers, 82,
1161-1167.
13.Mikshina, P. V., Idiyatullin, B. Z., Petrova, A.
A., Shashkov, A. S., Zuev, Y. F., and Gorshkova, T. A. (2015)
Physicochemical properties of complex rhamnogalacturonan I from
gelatinous cell walls of flax fibers, Carbohydr. Polymers,
117, 853-861.
14.Mellerowicz, E. J., Immerzeel, P., and Hayashi,
T. (2008) Xyloglucan: the molecular muscle of trees, Ann.
Botany, 102, 659-665.
15.Gorshkova, T. A., Gurjanov, O. P., Mikshina, P.
V., Ibragimova, N. N., Mokshina, N. E., Salnikov, V. V., Ageeva, M. V.,
Amenitskii, S. I., Chernova, T. E., and Chemikosova, S. B. (2010)
Specific type of secondary cell wall formed by plant fibers, Russ. J.
Plant Physiol., 57, 328-341.
16.Gorshkova, T., Brutch, N., Chabbert, B.,
Deyholos, M., Hayashi, T., Lev-Yadun, S., Mellerowicz, E. J., Morvan,
C., Neutelings, G., and Pilate, G. (2012) Plant fiber formation: state
of the art, recent and expected progress, and open questions, Crit.
Rev. Plant Sci., 31, 201-228.
17.Mellerowicz, E. J., and Gorshkova, T. A. (2012)
Tensional stress generation in gelatinous fibers: a review and possible
mechanism based on cell-wall structure and composition, J. Exp.
Bot., 63, 551-565.
18.Mikshina, P. V., Chernova, T. E., Chemikosova, S.
B., Ibragimova, N. N., Mokshina, N. Y., and Gorshkova, T. A. (2013) in
Cellulose (van de Ven, T., and Godbout, L., eds.) InTech,
Rijeka, pp. 91-112.
19.Salnikov, V. V., Ageeva, M. V., and Gorshkova, T.
A. (2008) Homofusion of Golgi secretory vesicles in flax phloem fibers
during formation of the gelatinous secondary cell wall,
Protoplasma, 233, 269-273.
20.Gorshkova, T. A., Wyatt, S. E., Salnikov, V. V.,
Gibeaut, D. M., Ibragimov, M. R., Lozovaya, V. V., and Carpita, N. C.
(1996) Cell wall polysaccharides of developing flax plants, Plant
Physiol., 110, 721-729.
21.Gorshkova, T. A., Salnikov, V. V., Chemikosova,
S. B., Ageeva, M. V., Pavlencheva, N. V., and van Dam, J. E. G. (2003)
Snap point: a transient point in Linum usitatissimum bast fiber
development, Ind. Crops Products, 18, 213-221.
22.Gurjanov, O. P., Ibragimova, N. N., Gnezdilov, O.
I., and Gorshkova, T. A. (2008) Polysaccharides, tightly bound to
cellulose in the cell wall of flax bast fiber: isolation and
identification, Carbohydr. Polymer, 72, 719-729.
23.Dubois, M., Gilles, K. A., and Hamilton, J. K.
(1956) Colorimetric method for determination of sugars and related
substances, Anal. Chem., 28, 350-356.
24.Mikshina, P. V., Gurjanov, O. P., Mukhitova, F.
K., Petrova, A. A., Shashkov, A. S., and Gorshkova, T. A. (2012)
Structural details of pectic galactan from the secondary cell walls of
flax (Linum usitatissimum L.) phloem fibers, Carbohydr.
Polymers, 87, 853-861.
25.Gulrez, S. K. H., Al-Assaf, S., and Phillips, G.
O. (2011) Hydrogels: methods of preparation, characterization and
applications, in Progress in Molecular and Environmental
Bioengineering – From Analysis and Modeling to Technology
Applications (Carpim, A., ed.) InTech, Rijeka, pp. 117-150.
26.Zimmermann, U., Husken, D., and Schulze, E.-D.
(1980) Direct turgor pressure measurements in individual leaf cells of
Tradescantia virginiana, Planta, 149, 445-453.
27.Perez, S., and Mazeau, K. (2005) in
Polysaccharides: Structural Diversity and Functional
Versatility, 2nd Edn. (Dumitriu, S., ed.) Marcel Dekker, N. Y., pp.
41-68.
28.Ioelovich, M. (2008) Cellulose as a
nanostructured polymer: a short review, BioResources, 3,
1403-1418.
29.Sturcova, A., Davies, G. R., and Eichhorn, S. J.
(2005) The elastic modulus and stress-transfer properties of tunicate
cellulose whiskers, Biomacromolecules, 6, 1055-1061.
30.Bos, H. L. (2004) The Potential of Flax Fibers
as Reinforcement for Composite Materials, Technische Universiteit
Eindhoven, Proefschrift, Eindhoven.
31.Kastner, H., Einhorn-Stoll, U., and Senge, B.
(2012) Structure formation in sugar containing pectin gels –
influence of Ca2+ on the gelation of low-methoxylated pectin
at acidic pH, Food Hydrocolloids, 27, 42-49.
32.Jha, A. K., Malik, M. S., Farach-Carson, M. C.,
Duncanb, R. L., and Jia, X. (2010) Hierarchically structured,
hyaluronic acid-based hydrogel matrices via the covalent integration of
microgels into macroscopic networks, RSC J., 6,
5045-5055.
33.Robertson, C. G., Bogoslovov, R., and Roland, C.
M. (2007) Effect of structural arrest on Poisson’s ratio in
nanoreinforced elastomers, Phys. Rev., 75,
051403-1-7.
34.Sekine, Y., Takagi, H., Sudo, S., Kajiwara, Y.,
Fukazawa, H., and Ikeda-Fukazawa, T. (2014) Dependence of structure of
polymer side chain on water structure in hydrogels, Polymer,
55, 6320-6324.
35.Zundel, G. (1969) Hydration and Intermolecular
Interaction: Infrared Investigations with Polyelectrolyte
Membranes, Academic Press, Germany.
36.Hofstetter, K., Hinterstoisser, B., and Salmen,
L. (2006) Moisture uptake in native cellulose – the roles of
different hydrogen bonds: a dynamic FT-IR study using deuterium
exchange, Cellulose, 13, 131-145.
37.Korobeinikov, S. N. (2000) Non-linear
Deformation of Solids [in Russian], Izd-vo SO RAN, Novosibirsk.
38.McNeil, M., Darvill, A. G., and Albersheim, P.
(1980) Structure of plant cell walls: X. Rhamnogalacturonan I, a
structurally complex pectic polysaccharide in the walls of
suspension-cultured sycamore cells, Plant Physiol., 66,
1128-1134.
39.Goubet, F., Bourlard, T., Girault, R., Alexandre,
C., Vandevelde, M. C., and Morvan, C. (1995) Structural features of
galactans from flax fibers, Carbohydr. Polymers, 27,
221-227.
40.Ridley, B. L., O’Neill, M. A., and Mohnen,
D. (2001) Pectins: structure, biosynthesis and
oligogalacturonide-related signaling, Phytochemistry, 57,
929-967.
41.Western, T. L., Skinner, D. J., and Haughn, G. W.
(2000) Differentiation of mucilage secretory cells of the
Arabidopsis seed coat, Plant Physiol., 122, 345-355.
42.Naran, R., Chen, G., and Carpita, N. C. (2008)
Novel rhamnogalacturonan I and arabinoxylan polysaccharides of flax
seed mucilage, Plant Physiol., 148, 132-141.
43.Ahmed, E. M. (2015) Hydrogel: preparation,
characterization, and applications, J. Adv. Res., 6,
105-121.
44.Richel, A., and Paquot, M. (2012) in
Carbohydrates – Comprehensive Studies on Glycobiology and
Glycotechnology (Chang, Ch-F., ed.) InTech, Rijeka, pp. 21-36.
45.Bezakova, Z., Hermannova, M., Drımalova,
E., Malovıkova, A., Ebringerova, A., and Velebny, V. (2008)
Effect of microwave irradiation on the molecular and structural
properties of hyaluronan, Carbohydr. Polymers, 73,
640-646.
46.Ulvskov, P., Wium, H., Bruce, D., Jorgensen, B.,
Qvist, K. B., Skjot, M., Hepworth, D., Borkhardt, B., and Sorensen, S.
O. (2005) Biophysical consequences of remodeling the neutral side
chains of rhamnogalacturonan I in tubers of transgenic potatoes,
Planta, 220, 609-620.
47.Larsen, F. H., Byg, I., Damager, I., Diaz, J.,
Engelsen, S. B., and Ulvskov, P. (2011) Residue specific hydration of
primary cell wall potato pectin identified by solid-state
13C single-pulse MAS and CP/MAS NMR spectroscopy,
Biomacromolecules, 12, 1844-1850.
48.Cardenas, A., Goycoolea, F. M., and Rinaudo, M.
(2008) On the gelling behavior of “nopal” (Opuntia
ficusindica) low methoxyl pectin, Carbohydr. Polymers,
73, 212-222.
49.Ngouemazong, E. D., Nkemamin, N. F., Cardinaels,
R., Jolie, R. P., Fraeye, I., Van Loey, A. M., Moldenaers, P., and
Hendrickx, M. E. (2012) Rheological properties of Ca2+-gels
of partially methyl-esterified polygalacturonic acid: effect of
“mixed” patterns of methyl-esterification, Carbohydr.
Polymers, 88, 37-45.
50.Park, H., Park, K., and Shalaby, W. S. W. (1993)
Biodegradable Hydrogels for Drug Delivery, Technomic Publishing
Company, Lancaster, Pennsylvania, USA.