Better Understanding of Phosphoinositide 3-Kinase (PI3K) Pathways in Vasculature: Towards Precision Therapy Targeting Angiogenesis and Tumor Blood Supply
D. Tsvetkov1#, A. Shymanets2#, Yu Huang3, K. Bucher2, R. Piekorz4, E. Hirsch5, S. Beer-Hammer2, C. Harteneck2&, M. Gollasch1*, and B. Nürnberg2*
1Charité University Medicine Berlin, Experimental and Clinical Research Center, Section Nephrology/Intensive Care, 13125 Berlin, Germany; E-mail: maik.gollasch@charite.de2Eberhard Karls University Hospitals and Clinics, and Interfaculty Center of Pharmacogenomics and Drug Research, Institute of Experimental and Clinical Pharmacology and Toxicology, Department of Pharmacology and Experimental Therapy, 72074 Tübingen, Germany; E-mail: bernd.nuernberg@uni-tuebingen.de
3Chinese University of Hong Kong, School of Biomedical Sciences, 223A, Lo Kwee-Seong Integrated Biomedical Sciences Building, Area 39, Hong Kong, China
4Heinrich Heine University Düsseldorf, Institute for Biochemistry and Molecular Biology II, 40225 Düsseldorf, Germany
5University of Torino, Molecular Biotechnology Centre, Department of Molecular Biotechnology and Health Sciences, 10126 Turin, Italy
# These authors contributed equally to this work.
& Deceased.
* To whom correspondence should be addressed.
Received February 26, 2016; Revision received April 12, 2016
The intracellular PI3K-AKT-mTOR pathway is involved in regulation of numerous important cell processes including cell growth, differentiation, and metabolism. The PI3Kα isoform has received particular attention as a novel molecular target in gene therapy, since this isoform plays critical roles in tumor progression and tumor blood flow and angiogenesis. However, the role of PI3Kα and other class I isoforms, i.e. PI3Kβ, γ, δ, in the regulation of vascular tone and regional blood flow are largely unknown. We used novel isoform-specific PI3K inhibitors and mice deficient in both PI3Kγ and PI3Kδ (Pik3cg–/–/Pik3cd–/–) to define the putative contribution of PI3K isoform(s) to arterial vasoconstriction. Wire myography was used to measure isometric contractions of isolated murine mesenteric arterial rings. Phenylephrine-dependent contractions were inhibited by the pan PI3K inhibitors wortmannin (100 nM) and LY294002 (10 µM). These vasoconstrictions were also inhibited by the PI3Kα isoform inhibitors A66 (10 µM) and PI-103 (1 µM), but not by the PI3Kβ isoform inhibitor TGX 221 (100 nM). Pik3cg–/–/Pik3cd–/–-arteries showed normal vasoconstriction. We conclude that PI3Kα is an important downstream element in vasoconstrictor GPCR signaling, which contributes to arterial vasocontraction via α1-adrenergic receptors. Our results highlight a regulatory role of PI3Kα in the cardiovascular system, which widens the spectrum of gene therapy approaches targeting PI3Kα in cancer cells and tumor angiogenesis and regional blood flow.
KEY WORDS: p110α, phenylephrine, α1-adrenergic receptor, PI3K/AktDOI: 10.1134/S0006297916070051
Abbreviations: α1-AR, α1-adrenergic receptors; GPRC, G-protein coupled receptors; PI3Ks, phosphoinositide 3-kinases; RTK, receptor tyrosine kinases; VDCC, voltage-dependent Ca2+ channels; VSMCs, vascular smooth muscle cells.
Phosphoinositide 3-kinases (PI3Ks) comprise a family of lipid kinases
catalyzing phosphorylation of the 3-position hydroxyl group of the
inositol ring of phosphoinositides, which in turn are considered to act
as second messengers [1, 2].
Activation of PI3K pathways is commonly observed in human cancer and is
critical for tumor progression and resistance to chemotherapy [3]. Analyses of the biochemical and structural
properties of PI3K enzymes have led to the discovery and synthesis of
several compounds that are currently being tested as novel therapeutics
in clinical trials [4]. The application of these
drugs is hampered by side effects, which may depend on PI3K isoform
selectivity.
Classification, subunit composition, and functions of PI3Ks have been reviewed elsewhere [5]. In general, based on homology, substrate specificity, and structure, PI3Ks can be divided into three different classes: PI3K I, II, and III. The most widely studied class I PI3K subfamily comprises heterodimeric enzymes that are further subdivided into PI3K IA catalytic isoforms (i.e. p110α, p110β, p110δ), which are associated with p85-like regulatory/adapter subunits, and one PI3K IB catalytic subunit (i.e. p110γ), which binds to p87 (also termed p84) or p101 non-catalytic subunits. The members of class I PI3Ks also differ in their sensitivity towards upstream regulators involving either receptor tyrosine kinases (RTK) or/and G-protein coupled receptors (GPRC). Thus, p110β and p110γ are the only isoforms known to be directly activated by GPCRs via Gβγ subunits, where p110β, as well as p110α and δ, are sensitive towards RTKs via direct interaction via p85 adapter subunits.
Among all members of PI3Ks, PI3Kα (catalytic subunit: p110α) has received particular attention as a novel molecular target in gene therapy, since PI3Kα is critically involved in tumor progression, tumor blood flow regulation, and angiogenesis, at least in breast cancer and glioblastoma [6-8]. In addition to its oncological and vascular implications, PI3Kα seems to play significant roles in cardiac function, since transgenic mice overexpressing p110α display cardiac hypertrophy [9], whereas activation of p110α protects the heart against myocardial infarction-induced heart failure [10]. Hence, the PI3Kα isoform appears a promising target for precision therapy in cardiovascular medicine. However, the role of PI3Kα and other isoforms in vascular signaling and regulation of vascular tone is largely unknown. Therefore, we studied the contribution of PI3K isoforms in the regulation of arterial contraction via α1-adrenergic receptors (α1-AR) using pharmacological and genetic tools and approaches.
MATERIALS AND METHODS
Materials. Chemicals obtained from Sigma-Aldrich (Germany) were of the highest analytical grade. Phenylephrine (Sigma Aldrich) and serotonin (5-HT) (Research Biochemical International, USA) were freshly dissolved on the day of the experiment with distillated water according to the supplier’s recommendations. Agonists were ranged: phenylephrine from 0.1 to 100 µM, 5-HT from 0.1 to 3 µM. PI3K inhibitors were purchased from Sigma-Aldrich (wortmannin), Calbiochem (LY294002 and TGX 221), Axon Medchem (A66), and Enzo Life Sciences (PI-103). These compounds were dissolved using DMSO; the maximal DMSO concentration in the bath solution did not exceed 0.5%. Stock solutions of wortmannin (10 mM), LY294002 (10 mM), A66 (10 mM), PI-103 (10 mM), and TGX 221 (1 mM) were stored at –80°C. All PI3K inhibitors were applied at effective concentrations as described previously [11-15].
Mouse model. The double deficient mouse strain lacking both p110γ and p110δ (Pik3cg–/–/Pik3cd–/–) was generated from crossbreeding singly deficient p110γ and p110δ mice [16]. For breeding, animals were kept in individually ventilated cages (IVC) under standardized conditions with an artificial 12-h dark–light cycle with free access to water and food according to national guidelines for animal care at the animal facilities of the University of Tübingen or Charité. Male mice used in this study were between 12-16 weeks old and on a C57BL/6N genetic background (Charles River). All experimental protocols were approved by local authorities (Landesamt für Gesundheit und Soziales Berlin, LAGeSo). We followed American Physiological Society guidelines for animal care.
Wire myography. Mesenteric arteries were removed immediately after sacrificing the mice with isoflurane anesthesia, quickly transferred to cold (4°C), oxygenated (95% O2/5% CO2) physiological salt solution (PSS) containing (in mM) 119 NaCl, 4.7 KCl, 1.2 KH2PO4, 25 NaHCO3, 1.2 MgSO4, 11.1 glucose, 1.6 CaCl2, and dissected into 2 mm rings, as previously described [17]. Perivascular fat and connective tissue were removed without damaging the adventitia. Each ring was placed between two stainless steel wires (diameter 0.0394 mm) in a 5-ml organ bath of a Mulvany Small Vessel Myograph (DMT 610 M; Danish Myo Technology, Denmark). The organ bath was filled with PSS. The bath solution was continuously oxygenated with a gas mixture of 95% O2 and 5% CO2, and kept at 37°C (pH 7.4). The arterial rings were placed under a tension equivalent to that generated at 0.9 times the diameter of the vessel at 100 mm Hg by stepwise distending the vessel using a LabChart DMT normalization module. This normalization procedure was performed to obtain the passive diameter of the vessel at 100 mm Hg [17]. The Chart5 software (AD Instruments Ltd., Germany) was used for data acquisition and display. After 60 min incubation time, arteries were pre-contracted with isotonic external 60 mM KCl until a stable resting tension was acquired. The composition of the 60 mM KCl solution (in mM) was 63.7 NaCl, 60 KCl, 1.2 KH2PO4, 25 NaHCO3, 1.2 MgSO4, 11.1 glucose, and 1.6 CaCl2. Drugs were added to the bath solution if not indicated otherwise. Tension is expressed as a percentage of the steady-state tension (100%) obtained with isotonic external 60 mM KCl.
Statistics. Data presented are mean ± SEM. Statistical significance was determined by Student’s t-test, and p-values were corrected by the Bonferroni procedure. EC50 values were calculated using the Hill equation: T = (B0 – Be)/(1 + ([D]/EC50)n) + Be, where T is the tension in response to the drug (D); Be is the maximum response induced by the drug; B0 is a constant; EC50 is the concentration of the drug that elicits the half-maximal response [18]. Curves were fit using Prism 6 software applying nonlinear regression. The extra sum-of-squares F-test was performed for comparison of concentration–response curves; p values < 0.05 were considered statistically significant; n represents the number of arteries tested.
RESULTS
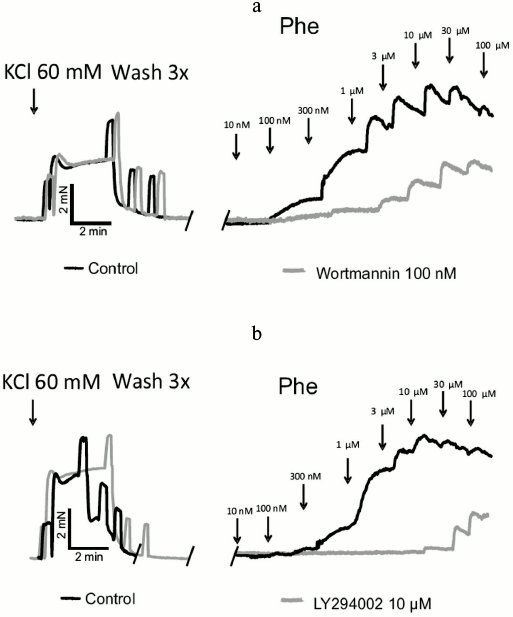
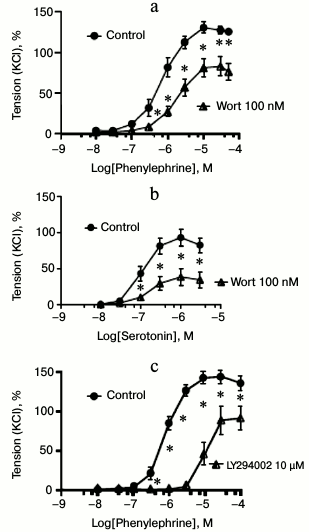
Effects of the pan PI3K inhibitors wortmannin and LY294002. First, we studied the effects of two structurally distinct and mechanistically differently acting pan class I PI3K inhibitors, namely wortmannin and LY294002, on agonist-induced vasocontractions of murine mesenteric arterial rings at maximally effective concentrations [11, 12]. Figures 1a and 2a show recordings and concentration–response curves, respectively, demonstrating that wortmannin inhibited phenylephrine-induced vasocontractions (EC50 of control and wortmannin treated rings were 0.70 ± 0.12 and 1.69 ± 0.08 µM (p < 0.05), respectively). Similar results were obtained with serotonin as vasoconstrictor (EC50 of control and wortmannin-treated rings were 0.10 ± 0.06 and 0.16 ± 0.04 µM (p < 0.05), respectively) (Fig. 2b). LY294002 also inhibited phenylephrine-induced vasocontractions (EC50 of control and LY294002 treated rings were 0.80 ± 0.14 and 10.0 ± 1.0 µM (p < 0.05), respectively) (Figs. 1b and 2c). These results strongly suggest that class I PI3Ks are involved in GPCR-induced vasocontractions, at least via α1-AR GPCRs.
Fig. 1. Effects of pan PI3K inhibitors wortmannin and LY294002 on vasocontractions. Original recordings of isometric contractions of isolated vessels in response to increasing concentrations of phenylephrine (Phe) are shown. Vessels from the control group versus vessels preincubated either with wortmannin (100 nM, 30 min after 60 mM KCl application) (a) or LY294002 (10 µM, 10 min after 60 mM KCl application (b).
Fig. 2. Effects of pan PI3K inhibitors wortmannin and LY294002 on vasocontractions. a) Inhibition of phenylephrine-dependent contractions by wortmannin (Wort, 100 nM) (n = 6 control, n = 6 Wort treated arterial rings). b) Inhibition of serotonin-dependent contractions by wortmannin (Wort, 100 nM) (n = 7 control, n = 6 Wort arterial treated rings). c) Inhibition of phenylephrine-dependent contractions by LY294002 (10 µM) (n = 5 control, n = 6 LY294002 treated arterial rings); * p < 0.05.
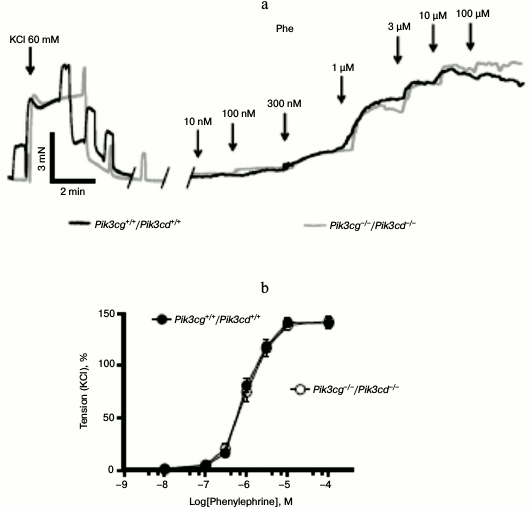
PI3Kγ and PI3Kδ do not regulate vasoconstriction downstream of α1-adrenergic receptors. Next, we sought to determine the PI3K isoform involved in α1-AR-dependent vasocontraction. Of the four class I isoforms, PI3Kα and PI3Kβ are ubiquitously expressed, whereas PI3Kγ and PI3Kδ exhibit more restricted expression with highest levels in the hematopoietic system. To check for a role of the latter PI3K isoforms, we analyzed mesenteric arterial rings obtained from mice deficient in both p110γ and p110δ (Pik3cg−/−/Pik3cd−/−). As depicted in Fig. 3a, phenylephrine induced normal vasoconstrictions regardless whether PI3Kγ and PI3Kδ were present or not. Concentration–response curves revealed similar EC50 (EC50 were 0.98 ± 0.05 and 0.93 ± 0.05 µM in wild type and Pik3cg−/−/Pik3cd−/− arteries, respectively) (Fig. 3b). Therefore, we assume that these two isoforms have no substantial role in the α1-AR pathway.
Fig. 3. Normal vasocontractions of Pik3cg−/−/Pik3cd−/− mesenteric arteries. a) Original recordings of isometric contractions of isolated vessels in response to increasing concentrations of phenylephrine (Phe). b) Phe-dependent contractions of arterial rings isolated from wildtype (Pik3cg+/+/Pik3cd+/+) (n = 11 arterial rings) and Pik3cg−/−/Pik3cd−/− mice (n = 16 arterial rings).
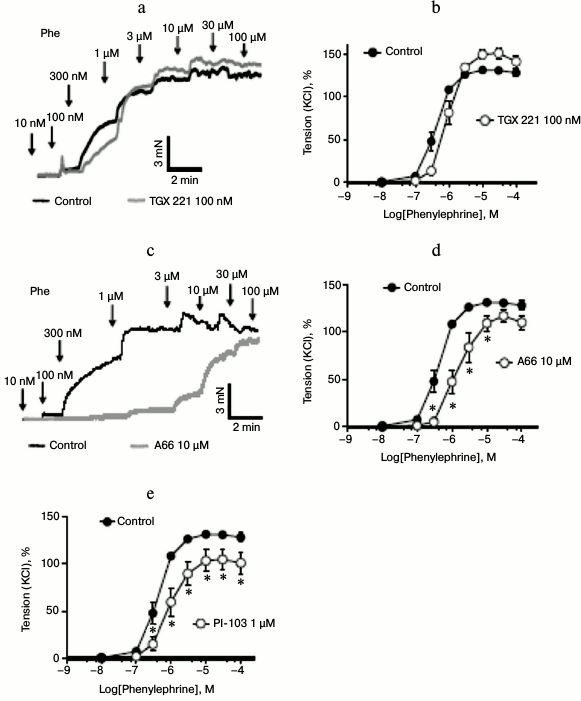
Selective vasoconstrictive role of PI3Kα. Of the remaining PI3K isoforms, PI3Kβ is directly coupled to Gβγ complexes that are under control of GPCRs [19, 20]. We therefore tested the PI3Kβ isoform-specific inhibitor TGX 221 at maximally effective concentration [13]. The concentration–response curve of phenylephrine-induced contraction of mesenteric artery rings displayed a negligible effect of the PI3Kβ-specific inhibitor (Fig. 4a). This makes PI3Kβ unlikely to play a major role in the α1-AR pathway leading to vasoconstriction.
Fig. 4. Involvement of PI3Kα isoform in vasocontractions. a) Original recordings of isometric contractions of isolated vessels in response to increasing concentrations of phenylephrine (Phe). Control vessels versus vessels preincubated with the PI3Kβ inhibitor TGX 221 (100 nM, 30 min after 60 mM KCl application). b) Effects of PI3Kβ inhibitor TGX 221 (100 nM) on phenylephrine-dependent contractions (n = 7 control, n = 7 TGX 221 treated arterial rings). c) Original recordings of isometric contractions of isolated vessels in response to increasing concentrations of phenylephrine (Phe). Control vessels versus vessels preincubated with the PI3Kα inhibitor A66 (10 µM, 30 min after 60 mM KCl application). d) Effects of A66 (10 µM) on phenylephrine-dependent contractions (n = 7 control rings, n = 7 A66 treated arterial rings). e) Effects of PI3Kα inhibitor PI-103 (1 µM) on phenylephrine-dependent contractions (n = 7 control, n = 6 PI-103 treated arterial rings); * p < 0.05.
In contrast, the PI3Kα isoform-specific inhibitor A66 significantly blunted agonist-stimulated vasoconstriction (Figs. 4, c and d). The EC50 values were 0.41 ± 0.14 and 1.34 ± 0.11 µM in control and A66 treated rings (p < 0.05), respectively. Results with the structurally distinct PI3Kα-isoform inhibitor PI-103 [13, 14] confirmed our findings on phenylephrine-induced vasocontractions with A66. The EC50 values were 0.41 ± 0.14 and 0.85 ± 0.11 µM in control and PI-103 treated rings (p < 0.05), respectively.
Taken together, our data suggest that the PI3Kα isoform specifically contributes to α1-adrenergic arterial vasocontraction, whereas we consider PI3Kβ, γ, and δ to be not involved.
DISCUSSION
The PI3Kα isoform is found to be constitutively activated following oncogenic mutations in many malignant tumors (cancers) [6, 21]. This has attracted broad interest in identifying inhibitors of the PI3K pathway and in particular those blocking the p110α catalytic subunit of PI3Kα. In addition, increasing evidence suggests the involvement of class I PI3-kinases in regulation of the vascular tone [22, 23]. Therefore, inhibition of PI3K-activity may have a significant impact on tumor blood flow regulation. As a consequence, understanding not only the underlying mechanisms of tumor initiation and progression by the oncogenic mutations but also elucidating the mechanisms regulating tumor blood supply by PI3K is of particular interest to design a precision therapy. Here, we focused on the impact of different PI3K isoforms in the regulation of arterial vascular tone following agonist-stimulated-α1-AR-induced vasocontraction.
Effects of pan class I PI3K inhibitors on vasocontractions. Therefore, we first performed a set of experiments using wortmannin and LY294002 to test if class I PI3-kinases contribute to α1-AR-induced contraction of murine small resistance-sized mesenteric arteries. Wortmannin and LY294002 are first generation PI3K inhibitors exhibiting a broad-spectrum, which greatly contributed to our understanding of PI3K pathways [24]. In our experiments, we applied both drugs at concentrations assumed to be specific for class I PI3Ks. Under these conditions, they induced a strong rightward shift of concentration–response curves of mesenteric artery rings following stimulation by phenylephrine and serotonin. The results suggest that at least one class I PI3K contributes to the regulation of arterial tone via α1-AR. Our data are consistent with previous results obtained by Sinagra et al., who reported that LY294002 inhibits phenylephrine-induced contractions in rat femoral arteries [25]. Nevertheless, experiments with broad-spectrum PI3K inhibitors should be interpreted with caution for several reasons. First, preclinical studies revealed little to no selectivity of wortmannin and LY294002 for individual PI3K isoforms [24]. Second, due to their mechanism of action on the ATP-binding site, they may also be critical for function of other kinases, including myosin light change kinase [26]. Third, LY294002 may inhibit Cav1.2 L-type Ca2+ channels (also known as voltage-dependent Ca2+ channels (VDCC)) in arterial myocytes [27]. Theoretically, the latter affect may explain putative vascular side effects of certain PI3K inhibitors, such as decreased systemic blood pressure.
Role of PI3Kα in vascular tone regulation. Next, we have specifically addressed the involvement of the four members of class I PI3K isoforms by using either isoform-selective pharmacological inhibitors and by studying mesenteric arteries from mice deficient in PI3Kγ and PI3Kδ (Pik3cg−/−/Pik3cd−/−). Although some studies have suggested a role of PI3Kγ in angiotensin II-dependent vasotoxic, hypertensive effects [28] and regulation of ATP-sensitive K+ channels activated by α1 adrenergic stimuli [29], we failed to detect any difference in contraction induced by phenylephrine in arterial rings isolated from Pik3cg−/−/Pik3cd−/− mice, compared to rings from wild-type control mice. Interestingly, p110γ and p110δ are predominantly expressed in leukocytes, where their genetic deletion lead to impaired immune functions and target-organ damage [30-33]. Moreover, in our experiments we observed no effect of the specific p110β inhibitor TGX-221 on phenylephrine-induced contractions. Therefore, we conclude that the three isoforms β, γ, and δ are unlikely to be involved in α1-AR-induced vasocontraction.
In contrast, our experiments using the selective PI3Kα isoform inhibitors A66 and PI-103 point to the PI3Kα isoform transmitting α1-adrenergic-induced arterial vasocontraction. Our conclusion is in line with several other findings. First, the p110α isoform is ubiquitously expressed including vascular smooth muscle cells (VSMCs). Surprisingly, by transcriptome analysis, only the Pik3r1 gene (phosphoinositide-3-kinase, regulatory subunit 1, p85α) was shown to be differently expressed in arteries isolated from obese rats compared to arteries from normal rats [34]; in contrast, the expression pattern of other PI3K isoforms did not differ. As the obese rats show increased blood pressure compared to normal-weight controls [35], it is intriguing to speculate that blood pressure changes could be due to changes in PI3Kα activity. One unanswered question is how the PIK3α isoform can be controlled by GPCRs. In this scenario, it may couple to Gq/11, which is known to mediate α1-AR contractions. So far, such a direct activation of PI3K by GPCR has only been demonstrated for PI3Kβ and -γ isoforms through interaction with Gβγ and Ras or Rho family members [19, 36-39]. Instead, PI3Kα is believed to be directly activated mainly through tyrosine kinase receptor (TKR) pathways, which apparently contradicts our main findings.
However, recent studies have demonstrated that α1-AR-induced contraction of rat aorta involves transactivation of epidermal growth factor receptor (EGFR) mediated via a PI3K- and ERK1/2-dependent pathway [40]. Based on these findings, it is possible that PI3Kα will represent the PI3K isoform contributing to such transactivation, i.e. crosstalk between PI3K, RTK, and GPCR signaling in arterial smooth muscle. Moreover, inhibition of α1-AR by doxazosin is known to interfere with the PI3K-AKT pathway to inhibit tumor growth and angiogenesis [41]. These data are in line with recent findings indicating that angiogenesis selectively requires the p110α isoform of PI3K to control endothelial cell migration [42]. In the latter study, p110α was shown to exert this critical endothelial cell-autonomous function by regulating endothelial cell migration through the small GTPase RhoA. Clearly, future studies should shed more light on intra- and intercellular interactions between p110α signaling pathways in the vasculature contributing to tumor blood supply.
In conclusion, our findings suggest for the first time that the PI3Kα isoform is integrated into the α1-AR signaling pathway regulating the contractility of mesenteric arteries. Our findings make the PI3Kα isoform a promising target for precision therapy fighting tumors, not only because of targeting directly cancer cells, but also by its ability to regulate regional blood flow, which is expected to contribute to angiogenesis within the tumor. Novel CRISPR technologies may reveal new information about molecular components of PI3Kα-pathways involved in this scenario and even represent innovative therapeutic approaches to target PI3K.
Our research was supported by the Deutsche Akademische Austauschdienst (DAAD), Dr. Werner Jackstädt Stiftung, the Interfaculty Center of Pharmacogenomics and Drug Research (ICePhA), and Deutsche Forschungsgemeinschaft (DFG).
REFERENCES
1.Fruman, D. A., and Rommel, C. (2014) PI3K and
cancer: lessons, challenges and opportunities, Nat. Rev. Drug
Discov., 13, 140-156.
2.Balla, T. (2013) Phosphoinositides: tiny lipids
with giant impact on cell regulation, Physiol. Rev.,
93, 1019-1037.
3.Courtney, K. D., Corcoran, R. B., and Engelman, J.
A. (2010) The PI3K pathway as drug target in human cancer, J. Clin.
Oncol., 28, 1075-1083.
4.Rodon, J., Dienstmann, R., Serra, V., and
Tabernero, J. (2013) Development of PI3K inhibitors: lessons learned
from early clinical trials, Nat. Rev. Clin. Oncol., 10,
143-153.
5.Vadas, O., Burke, J. E., Zhang, X., Berndt, A., and
Williams, R. L. (2011) Structural basis for activation and inhibition
of class I phosphoinositide 3-kinases, Sci. Signal., 4,
re2.
6.Samuels, Y., Wang, Z., Bardelli, A., Silliman, N.,
Ptak, J., Szabo, S., Yan, H., Gazdar, A., Powell, S. M., Riggins, G.
J., Willson, J. K. V., Markowitz, S., Kinzler, K. W., Vogelstein, B.,
and Velculescu, V. E. (2004) High frequency of mutations of the
PIK3CA gene in human cancers, Science, 304,
554.
7.Parsons, D. W., Jones, S., Zhang, X., Lin, J.
C.-H., Leary, R. J., Angenendt, P., Mankoo, P., Carter, H., Siu, I.-M.,
Gallia, G. L., Olivi, A., McLendon, R., Rasheed, B. A., Keir, S.,
Nikolskaya, T., Nikolsky, Y., Busam, D. A., Tekleab, H., Diaz, L. A.,
Hartigan, J., Smith, D. R., Strausberg, R. L., Marie, S. K. N., Shinjo,
S. M. O., Yan, H., Riggins, G. J., Bigner, D. D., Karchin, R.,
Papadopoulos, N., Parmigiani, G., Vogelstein, B., Velculescu, V. E.,
and Kinzler, K. W. (2008) An integrated genomic analysis of human
glioblastoma multiforme, Science, 321, 1807-1812.
8.Yuan, T. L., Choi, H. S., Matsui, A., Benes, C.,
Lifshits, E., Luo, J., Frangioni, J. V., and Cantley, L. C. (2008)
Class 1A PI3K regulates vessel integrity during development and
tumorigenesis, Proc. Natl. Acad. Sci. USA, 105,
9739-9744.
9.Shioi, T., Kang, P. M., Douglas, P. S., Hampe, J.,
Yballe, C. M., Lawitts, J., Cantley, L. C., and Izumo, S. (2000) The
conserved phosphoinositide 3-kinase pathway determines heart size in
mice, EMBO J., 19, 2537-2548.
10.Lin, R. C. Y., Weeks, K. L., Gao, X.-M.,
Williams, R. B. H., Bernardo, B. C., Kiriazis, H., Matthews, V. B.,
Woodcock, E. A., Bouwman, R. D., Mollica, J. P., Speirs, H. J., Dawes,
I. W., Daly, R. J., Shioi, T., Izumo, S., Febbraio, M. A., Du, X.-J.,
and McMullen, J. R. (2010) PI3K(p110 alpha) protects against myocardial
infarction-induced heart failure: identification of PI3K-regulated
miRNA and mRNA, Arterioscler. Thromb. Vasc. Biol., 30,
724-732.
11.Shymanets, A., Prajwal, Vadas, O., Czupalla, C.,
LoPiccolo, J., Brenowitz, M., Ghigo, A., Hirsch, E., Krause, E.,
Wetzker, R., Williams, R. L., Harteneck, C., and Nürnberg, B.
(2015) Different inhibition of Gβγ-stimulated class IB
phosphoinositide 3-kinase (PI3K) variants by a monoclonal antibody.
Specific function of p101 as a Gβγ-dependent regulator of
PI3Kγ enzymatic activity, Biochem. J., 469,
59-69.
12.Quignard, J. F., Mironneau, J., Carricaburu, V.,
Fournier, B., Babich, A., Nürnberg, B., Mironneau, C., and Macrez,
N. (2001) Phosphoinositide 3-kinase γ mediates angiotensin
II-induced stimulation of L-type calcium channels in vascular myocytes,
J. Biol. Chem., 276, 32545-32551.
13.Chaussade, C., Rewcastle, G. W., Kendall, J. D.,
Denny, W. A., Cho, K., Gronning, L. M., Chong, M. L., Anagnostou, S.
H., Jackson, S. P., Daniele, N., and Shepherd, P. R. (2007) Evidence
for functional redundancy of class IA PI3K isoforms in insulin
signaling, Biochem. J., 404, 449-458.
14.Knight, Z. A., Gonzalez, B., Feldman, M. E.,
Zunder, E. R., Goldenberg, D. D., Williams, O., Loewith, R., Stokoe,
D., Balla, A., Toth, B., Balla, T., Weiss, W. A., Williams, R. L., and
Shokat, K. M. (2006) A pharmacological map of the PI3-K family defines
a role for p110alpha in insulin signaling, Cell, 125,
733-747.
15.Jamieson, S., Flanagan, J. U., Kolekar, S.,
Buchanan, C., Kendall, J. D., Lee, W.-J., Rewcastle, G. W., Denny, W.
A., Singh, R., Dickson, J., Baguley, B. C., and Shepherd, P. R. (2011)
A drug targeting only p110α can block phosphoinositide 3-kinase
signaling and tumor growth in certain cell types, Biochem. J.,
438, 53-62.
16.Beer-Hammer, S., Zebedin, E., Von Holleben, M.,
Alferink, J., Reis, B., Dresing, P., Degrandi, D., Scheu, S., Hirsch,
E., Sexl, V., Pfeffer, K., Nürnberg, B., and Piekorz, R. P. (2010)
The catalytic PI3K isoforms p110gamma and p110delta contribute to
B-cell development and maintenance, transformation, and proliferation,
J. Leukoc. Biol., 87, 1083-1095.
17.Fesus, G., Dubrovska, G., Gorzelniak, K., Kluge,
R., Huang, Y., Luft, F. C., and Gollasch, M. (2007) Adiponectin is a
novel humoral vasodilator, Cardiovasc. Res., 75,
719-727.
18.Bychkov, R., Gollasch, M., Steinke, T., Ried, C.,
Luft, F. C., and Haller, H. (1998) Calcium-activated potassium channels
and nitrate-induced vasodilation in human coronary arteries, J.
Pharmacol. Exp. Ther., 285, 293-298.
19.Maier, U., Babich, A., and Nürnberg, B.
(1999) Roles of non-catalytic subunits in Gβγ-induced
activation of class I phosphoinositide 3-kinase isoforms β and
γ, J. Biol. Chem., 274, 29311-29317.
20.Dbouk, H. A., Vadas, O., Shymanets, A., Burke, J.
E., Salamon, R. S., Khalil, B. D., Barrett, M. O., Waldo, G. L., Surve,
C., Hsueh, C., Perisic, O., Harteneck, C., Shepherd, P. R., Harden, T.
K., Smrcka, A. V., Taussig, R., Bresnick, A. R., Nürnberg, B.,
Williams, R. L., and Backer, J. M. (2012) G protein-coupled
receptor-mediated activation of p110beta by Gβγ is required
for cellular transformation and invasiveness, Sci. Signal.,
5, ra89.
21.Lee, J. W., Soung, Y. H., Kim, S. Y., Lee, H. W.,
Park, W. S., Nam, S. W., Kim, S. H., Lee, J. Y., Yoo, N. J., and Lee,
S. H. (2005) PIK3CA gene is frequently mutated in breast
carcinomas and hepatocellular carcinomas, Oncogene, 24,
1477-1480.
22.Viard, P., Exner, T., Maier, U., Mironneau, J.,
Nürnberg, B., and Macrez, N. (1999) G beta gamma dimers stimulate
vascular L-type Ca2+ channels via phosphoinositide 3-kinase,
FASEB J., 13, 685-694.
23.Macrez, N., Mironneau, C., Carricaburu, V.,
Quignard, J. F., Babich, A., Czupalla, C., Nürnberg, B., and
Mironneau, J. (2001) Phosphoinositide 3-kinase isoforms selectively
couple receptors to vascular L-type Ca2+ channels, Circ.
Res., 89, 692-699.
24.Liu, P., Cheng, H., Roberts, T. M., and Zhao, J.
J. (2009) Targeting the phosphoinositide 3-kinase pathway in cancer,
Nat. Rev. Drug Discov., 8, 627-644.
25.Sinagra, T., Tamburella, A., Urso, V., Siarkos,
I., Drago, F., Bucolo, C., and Salomone, S. (2013) Reversible
inhibition of vasoconstriction by thiazolidinediones related to
PI3K/Akt inhibition in vascular smooth muscle cells, Biochem.
Pharmacol., 85, 551-559.
26.Yoshioka, K., Sugimoto, N., Takuwa, N., and
Takuwa, Y. (2007) Essential role for class II phosphoinositide 3-kinase
alpha-isoform in Ca2+-induced, Rho- and Rho kinase-dependent
regulation of myosin phosphatase and contraction in isolated vascular
smooth muscle cells, Mol. Pharmacol., 71, 912-920.
27.Welling, A., Hofmann, F., and Wegener, J. W.
(2005) Inhibition of L-type Cav1.2 Ca2+ channels
by 2,(4-morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002) and
2-[1-(3-dimethyl-aminopropyl)-5-methoxyindol-3-yl]-3-(1H-indol-3-yl)maleimide
(Go6983), Mol. Pharmacol., 67, 541-544.
28.Vecchione, C., Patrucco, E., Marino, G.,
Barberis, L., Poulet, R., Aretini, A., Maffei, A., Gentile, M. T.,
Storto, M., Azzolino, O., Brancaccio, M., Colussi, G. L., Bettarini,
U., Altruda, F., Silengo, L., Tarone, G., Wymann, M. P., Hirsch, E.,
and Lembo, G. (2005) Protection from angiotensin II-mediated
vasculotoxic and hypertensive response in mice lacking PI3Kgamma, J.
Exp. Med., 201, 1217-1228.
29.Haba, M., Hatakeyama, N., Kinoshita, H., Teramae,
H., Azma, T., Hatano, Y., and Matsuda, N. (2010) The modulation of
vascular ATP-sensitive K+ channel function via the
phosphatidylinositol 3-kinase-Akt pathway activated by phenylephrine,
J. Pharmacol. Exp. Ther., 334, 673-678.
30.Jou, S. T., Carpino, N., Takahashi, Y., Piekorz,
R., Chao, J. R., Wang, D., and Ihle, J. N. (2002) Essential,
nonredundant role for the phosphoinositide 3-kinase p110delta in
signaling by the B-cell receptor complex, Mol. Cell. Biol.,
22, 8580-8591.
31.Konrad, S., Ali, S. R., Wiege, K., Syed, S. N.,
Engling, L., Piekorz, R. P., Hirsch, E., Nurnberg, B., Schmidt, R. E.,
and Gessner, J. E. (2008) Phosphoinositide 3-kinases gamma and delta,
linkers of coordinate C5a receptor-Fc-gamma receptor activation and
immune complex-induced inflammation, J. Biol. Chem., 283,
33296-33303.
32.Hirsch, E., Katanaev, V. L., Garlanda, C.,
Azzolino, O., Pirola, L., Silengo, L., Sozzani, S., Mantovani, A.,
Altruda, F., and Wymann, M. P. (2000) Central role for G
protein-coupled phosphoinositide 3-kinase gamma in inflammation,
Science, 287, 1049-1053.
33.Ji, H., Rintelen, F., Waltzinger, C., Bertschy
Meier, D., Bilancio, A., Pearce, W., Hirsch, E., Wymann, M. P., Ruckle,
T., Camps, M., Vanhaesebroeck, B., Okkenhaug, K., and Rommel, C. (2007)
Inactivation of PI3Kgamma and PI3Kdelta distorts T-cell development and
causes multiple organ inflammation, Blood, 110,
2940-2947.
34.Jenkins, N. T., Padilla, J., Thorne, P. K.,
Martin, J. S., Rector, R. S., Davis, J. W., and Laughlin, M. H. (2014)
Transcriptome-wide RNA sequencing analysis of rat skeletal muscle feed
arteries. I. Impact of obesity, J. Appl. Physiol., 116,
1017-1032.
35.Takeshige, Y., Fujisawa, Y., Rahman, A.,
Kittikulsuth, W., Nakano, D., Mori, H., Masaki, T., Ohmori, K., Kohno,
M., Ogata, H., and Nishiyama, A. (2016) A sodium-glucose co-transporter
2 inhibitor empagliflozin prevents abnormality of circadian rhythm of
blood pressure in salt-treated obese rats, Hypertens. Res., doi:
10.1038/hr.2016.2.
36.Stoyanov, B., Volinia, S., Hanck, T., Rubio, I.,
Loubtchenkov, M., Malek, D., Stoyanova, S., Vanhaesebroeck, B., Dhand,
R., Nürnberg, B., et al. (1995) Cloning and characterization of a
G protein-activated human phosphoinositide-3 kinase, Science,
269, 690-693.
37.Vadas, O., Dbouk, H. A., Shymanets, A., Perisic,
O., Burke, J. E., Abi Saab, W. F., Khalil, B. D., Harteneck, C.,
Bresnick, A. R., Nürnberg, B., Backer, J. M., and Williams, R. L.
(2013) Molecular determinants of PI3Kγ-mediated activation
downstream of G-protein-coupled receptors (GPCRs), Proc. Natl. Acad.
Sci. USA, 110, 18862-18867.
38.Fritsch, R., De Krijger, I., Fritsch, K., George,
R., Reason, B., Kumar, M. S., Diefenbacher, M., Stamp, G., and
Downward, J. (2013) RAS and RHO families of GTPases directly regulate
distinct phosphoinositide 3-kinase isoforms, Cell, 153,
1050-1063.
39.Kurig, B., Shymanets, A., Bohnacker, T., Prajwal,
Brock, C., Ahmadian, M. R., Schaefer, M., Gohla, A., Harteneck, C.,
Wymann, M. P., Jeanclos, E., and Nürnberg, B. (2009) Ras is an
indispensable coregulator of the class IB phosphoinositide 3-kinase
p87/p110gamma, Proc. Natl. Acad. Sci. USA, 106,
20312-20317.
40.Ulu, N., Gurdal, H., Landheer, S. W., Duin, M.,
Guc, M. O., Buikema, H., and Henning, R. H. (2010)
α1-Adrenoceptor-mediated contraction of rat aorta is partly
mediated via transactivation of the epidermal growth factor receptor,
Br. J. Pharmacol., 161, 1301-1310.
41.Park, M. S., Kim, B.-R., Dong, S. M., Lee, S.-H.,
Kim, D.-Y., and Rho, S. B. (2014) The antihypertension drug doxazosin
inhibits tumor growth and angiogenesis by decreasing VEGFR-2/Akt/mTOR
signaling and VEGF and HIF-1α expression, Oncotarget,
5, 4935-4944.
42.Graupera, M., Guillermet-Guibert, J., Foukas, L.
C., Phng, L.-K., Cain, R. J., Salpekar, A., Pearce, W., Meek, S.,
Millan, J., Cutillas, P. R., Smith, A. J. H., Ridley, A. J., Ruhrberg,
C., Gerhardt, H., and Vanhaesebroeck, B. (2008) Angiogenesis
selectively requires the p110alpha isoform of PI3K to control
endothelial cell migration, Nature, 453, 662-666.