REVIEW: Viral Vectors for Gene Therapy: Current State and Clinical Perspectives
A. N. Lukashev1,2* and A. A. Zamyatnin, Jr.3,4
1Chumakov Institute of Poliomyelitis and Viral Encephalitides, 142782 Moscow, Russia; fax: +7 (495) 841-9321; E-mail: alexander_lukashev@hotmail.com2Sechenov First Moscow State Medical University, Martsinovsky Institute of Medical Parasitology and Tropical Medicine, 119991 Moscow, Russia
3Sechenov First Moscow State Medical University, Institute of Molecular Medicine, 119991 Moscow, Russia
4Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119991 Moscow, Russia; E-mail: zamyat@belozersky.msu.ru
* To whom correspondence should be addressed.
Received April 19, 2016
Gene therapy is the straightforward approach for the application of recent advances in molecular biology into clinical practice. One of the major obstacles in the development of gene therapy is the delivery of the effector to and into the target cell. Unfortunately, most methods commonly used in laboratory practice are poorly suited for clinical use. Viral vectors are one of the most promising methods for gene therapy delivery. Millions of years of evolution of viruses have resulted in the development of various molecular mechanisms for entry into cells, long-term survival within cells, and activation, inhibition, or modification of the host defense mechanisms at all levels. The relatively simple organization of viruses, small genome size, and evolutionary plasticity allow modifying them to create effective instruments for gene therapy approaches. This review summarizes the latest trends in the development of gene therapy, in particular, various aspects and prospects of the development of clinical products based on viral delivery systems.
KEY WORDS: DNA, RNA, viral vector, molecular targeting, smart drugsDOI: 10.1134/S0006297916070063
Abbreviations: AAV, adeno-associated virus; GT, gene therapy; HIV, human immunodeficiency virus; HSC, hematopoietic stem cells; MLV, murine leukemia virus; MVA, modified vaccinia virus Ankara.
During the recent decades, gene therapy (GT) has become one of the most
actively developing and most promising branches of medicine. Gene
therapy has a number of potential applications, such as:
– treatment of hereditary genetic diseases, primarily monogenic ones;
– anticancer therapy;
– treatment of infectious diseases;
– treatment of common therapeutic diseases.
Moreover, recombinant vaccines can also be regarded as gene therapy, because the way they are developed and produced is closer to GT approaches rather than to the development of classical vaccines.
METHODS OF GENE THERAPY
A characteristic feature of a gene therapy is the use of a nucleic acid to provide a specific effect in a cell. Generally, therapeutic effect is achieved through the expression of a gene encoded by this nucleic acid. It can code either for a protein, or for RNA, initiating RNA interference. Moreover, it should be noted that functions of nucleic acids are not limited only to storage and expression of genetic information. Newly developed “smart drugs” can be nucleic acids that not only contain genetic information, but also have special 3D-structures and enzymatic activities. Such nucleic acids are able to change their conformation in response to certain external stimuli, activating a ribozyme that excises, for example, microRNA, which initiates RNA interference [1-3].
Discovery of CRISPR-Cas systems allowing targeted changing (editing) of a genome [4] stimulated studies aimed at optimization of the delivery of CRISPR-Cas system components to a particular organ and further inside its cells. Scientists are still looking for alternative CRISPR-Cas systems with smaller sizes of genes/proteins that would fit into viral vectors [5].
GENE THERAPY DELIVERY SYSTEMS
An unprotected nucleic acid (RNA or DNA) is not stable for a long time in a biological environment. Moreover, nucleic acid is not able to enter by itself into the cytoplasm (where RNA can realize its function) or into the nucleus, where DNA is transcribed and the cell genome can be modified. Thus, one of the main problems of gene therapy is the delivery of an effector into cells. Viral vectors are considered as one of the most promising means of gene therapy delivery.
During evolution, viruses developed various mechanisms of entry into cells, of prolonged preservation of their own genetic material inside cells, and of suppression or modification of protective mechanisms in the host organism. Reverse genetics (methods for genome modification and generation of recombinant viruses) has been developing for about 40 years. For the majority of viruses, recombinant variants can be generated, and most of them, at least theoretically, have been considered as vectors for gene therapy, but only a limited number of viral delivery systems are being used. In this review, we discuss the latest trends in gene therapy development, in particular various aspects and perspectives of clinical development of viral delivery systems.
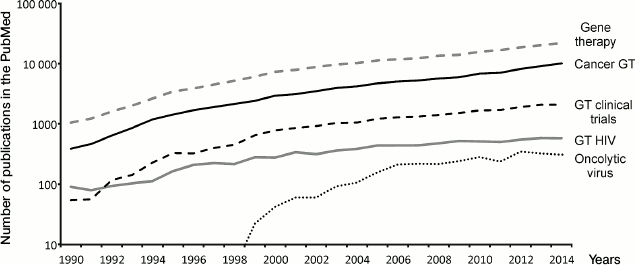
To visualize the milestones of gene therapy development, we analyzed the dynamics of publications in the main fields. According to the PubMed database, 22,000 articles on gene therapy were published in the world in 2014. The number of publications is constantly growing (Fig. 1). Nearly half of these publications (about 10,000) discuss the potential of gene therapy approaches in oncology, but only a small part describes actual attempts to achieve tumor regression by means of gene therapy. More often, the possibility of gene therapy or the possibility of affecting the dynamics of tumor progression by changing signal pathways are discussed (proof-of-concept). The number of articles describing oncolytic viruses is about 20-fold less (330 per year) and has not grown since 2012. This may be explained by the development of new approaches in immunotherapy of malignancies aimed at overcoming immune system tolerance [6, 7]. Another popular trend of gene therapy is the development of methods to treat human immunodeficiency virus (HIV) infection via ex vivo modification of T-lymphocytes (see below). The number of publications on this topic reached a plateau in 2005 and is almost constant since then. Thus, we can clearly see growing interest for gene therapy and the expansion of areas of its possible application. Moreover, the set of tools being used for gene therapy delivery has significantly changed in recent years.
Fig. 1. Dynamics of publications on different aspects of gene therapy from 1990 to 2014. Number of publications in the PubMed database (y-axis, logarithmic scale) according to year (x-axis) for the several search criteria specified at the right side of the plot.
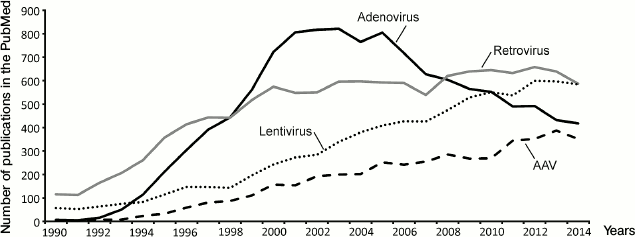
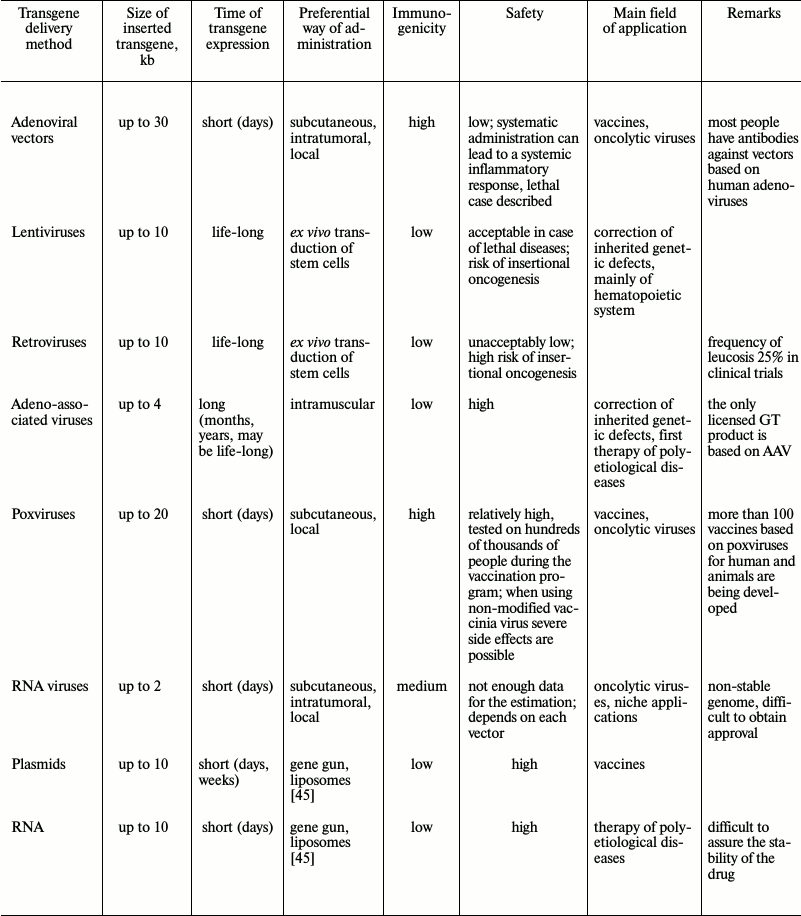
Up to now, a number of gene therapy delivery techniques have been developed. Viruses are regarded as the most promising gene therapy delivery system for clinical use. Many data concerning different viral-based vectors have accumulated (Fig. 2). The choice of the safest and most efficient delivery system depends on the task that needs to be solved by a new gene therapy approach (table).
Fig. 2. Dynamics of publications on different viral vectors in gene therapy from 1990 to 2014. Number of publications in the PubMed database (y-axis) according to year (x-axis). AAV, adeno-associated viruses.
Major routes of delivery of gene therapy products
ADENOVIRAL VECTORS
Adenoviruses were among the first thoroughly studied viruses and were proposed for use in gene therapy more than 20 years ago [8]. Adenoviruses are non-enveloped viruses with a double-stranded DNA genome of about 35 kb. To be used as vectors for gene therapy, adenoviruses are attenuated by deletion of a genome fragment coding for early proteins. Various levels of attenuation can be achieved by removal of different numbers of genes: only one E1B gene (first generation vectors), the majority of early genes (second generation vectors), and even full deletion of all genetic information of an adenovirus (so-called gutless vectors) [9]. Viruses with small deletions can be propagated in cultured cells with genetic defects allowing virus reproduction. Production of gutless vectors requires special producer cell lines. The large size of the genome and the possibility to delete a major part of it provide high coding capacity for these vectors: 1-2 kb can be inserted in early generation vectors, and up to 30 kb in gutless vectors. It is important to note that the adenoviral genome does not integrate into the genome of the host cell, which makes the vectors rather safe. On the other hand, the viral life cycle is not adapted for increased duration of transgene expression.
Originally, adenoviruses were planned to be used for a broad spectrum of clinical tasks, from therapy to regenerative medicine [10]. Later, it turned out that even genetically inert adenoviral particles have extremely immunogenic capsids. Systemic application of adenoviral vectors is complicated by the fact that many viral components bind nonspecifically to blood components (proteins of the coagulation cascade, complement proteins, erythrocytes, platelets), leading to inactivation of the virus [11, 12]. Moreover, systemic administration of high doses of adenovirus can lead to systemic inflammatory response, which can be lethal in some extreme cases [13]. Thus, adenoviruses can be used as gene therapy delivery system in applications when local administration is possible and an immune response is required, i.e. in therapy of malignant tumors or in development of vaccines.
Another drawback of adenoviruses is that they are widespread and often persist in human tonsils. From 50 to 80% of adults have antibodies against the most common adenovirus serotypes [14]. The most widespread serotype, adenovirus C5, was also used as a backbone for the majority of adenoviral vectors. Even a single administration of such vectors will lead with high probability to the development of a secondary immune response. To overcome this limitation, simian adenoviruses have been isolated and studied. Humans do not have antibodies to simian adenoviruses; therefore their structural proteins can be used to create chimeric adenoviruses [15].
Adenoviral vectors are now less used in gene therapy (Fig. 2) because of side effects, complexity of systematic administration, large genome size, and relative complexity of design.
RETROVIRUSES AND LENTIVIRUSES
Retroviruses are RNA viruses. Replication of retroviruses has an obligatory step of RNA copying into DNA (reverse transcription) and integration into the genome of a host cell. Early studies of the possible use of retroviruses for gene therapy began in the 1980s (Fig. 2). Before the discovery of CRISPR-Cas systems (see below), retroviruses were the only possible way to modify a patient’s genome. Naturally, retroviral vectors were first applied to curing monogenic disorders caused by a defect in a particular gene. The first gene therapy clinical trials using retroviruses started in the early 1990s were aimed at treatment of severe combined immunodeficiency caused by the lack of adenosine-deaminase [16-18]. This and some other genetic defects of the immune system (see below) were chosen as targets for gene therapy not only because of the possibility to cure them by correction of a single gene, but also because gene therapy could be applied ex vivo on isolated stem cells or on early precursors of blood cells. Vectors based on murine leukemia virus (MLV) from the group of γ-retroviruses were used in this study. The important feature of retroviruses is that they have a preferential site for genome integration. From this point of view, MLV turned out to be a poor choice, since it is more likely to integrate into actively transcribed genome loci close to the start of a transcription unit, which can lead, with high probability, to dysregulation of oncogene expression control [19-21]. In the first clinical trial, such integration led to the development of leucosis in five out of 20 patients [22].
Side effects of gene therapy caused by insertional mutagenesis were called “genotoxicity”. From the point of view of genotoxicity, lentiviruses – a group of retroviruses that include HIV – are much better vectors for gene therapy. One of their important advantages is the absence of preferential integration close to the start of transcription units [20]. Similarly to MLV, lentiviruses integrate in actively transcribed genome loci, but prefer 3′-regions of genes. Experimental data reveal that lentiviral vectors induced oncogenesis significantly less frequently than retroviral vectors [23]. The first lentiviral vector system was created only in 1996 [24]. Though technically lentiviral vectors are retroviral vectors, in practice the latter term is used only for vectors based on γ-retroviruses (primarily MLV).
To assure safe transgene delivery into cells by means of HIV-based vectors, one can use a system of several plasmids or a vector packaging cell line that expresses lentiviral proteins and produces viral capsids containing essential viral proteins, but only the transgene encoding RNA [25]. Moreover, noncoding sequences on the ends of lentiviral vectors are modified to reduce genotoxicity (auto-inactivating vectors) [26]. Despite much higher safety, lentiviral vectors cannot exclude a possibility of oncogenesis; clinical trials reveal cases of transduced cell transformation (usually without the development of clinically detected leucosis) (see below).
ADENO-ASSOCIATED VIRUSES
Adeno-associated viruses (AAV) are small non-enveloped viruses with single-stranded DNA that belong to the family Parvoviridae. AAV are non-autonomous parvoviruses, which means that they are not able to replicate in the absence of adenovirus. In nature, AAV infect humans and stay inactive in the cell nucleus. Most viral genomes do not integrate into the genome (they stay as episomes). A small part of genomes (about 0.1%) can be integrated into the host cell genome, with integration occurring specifically at a single site of chromosome 19. AAV based vectors created up to now are not able to integrate into a genome and, thus, do not have a genotoxic effect. One of the drawbacks related to such life cycle is that the number of AAV genomes in dividing cells decreases gradually, which, in turn, leads to decrease of a transgene expression level. For this reason, AAV are the best choice for transfection of slowly dividing cells, such as myocytes, cardiomyocytes, etc. [9]. It should also be mentioned that the AAV capsid is less immunogenic than those of adenoviruses or poxviruses. Severe systemic inflammatory response was not observed upon use of AAV; the virus is rather stable in blood, though an immune response was still observed. From 10 to 30% of humans are AAV seropositive [27]. This does not exclude the possibility of single-gene therapy application using AAV, especially in the case of local administration of the virus. Temporary immunosuppression or antibody traps based on empty AAV capsids can be used if needed [28]. To achieve longer effect, a number of strategies can be used, such as controlled release of small amounts of vector from adapted carriers [29]. Safety, facility of generation, and production made AAV based vectors, appearing only in the mid-1990s, as popular as adenovirus-based vectors (Fig. 2); they are considered as the most perspective way of temporary gene expression for gene therapy [30].
POXVIRUSES
Viruses of the Poxviridae family are the largest and most complex of viruses causing human disease. These viruses contain a double-stranded DNA genome of about 180-220 kb. One of the best-known representatives of poxviruses is the smallpox virus. The main tool for eradication of smallpox, achieved worldwide in 1980, was a live vaccine based on vaccinia virus – a poxvirus of unknown origin (most likely isolated from horses). Use of this vaccine allowed the accumulation of much information concerning the side effects of the virus; moreover, it was known that the initial vaccinia virus was very reactogenic. Shortly before the end of the global immunization program, new variants of attenuated virus were obtained. One the most perspective variants – Modified Virus Ankara (MVA) – was obtained upon 570 passages of vaccinia virus in cell culture [31]. As a result, MVA lost about 15% of its genome, including many genes responsible for in vivo pathogenesis and immune system repression. MVA was used as a vaccine against smallpox and was administrated to more than 120,000 people. Thus, the safety of this vector was studied clinically on a very large population. MVA is now being used for development of about 50 vaccines against viral, bacterial, and parasitic diseases [31].
OTHER VIRUSES
Reverse genetics (genome modification and generation of genetically engineered virus) is now possible for almost all human viruses and for a number of viruses of birds and other animals. RNA-containing viruses are not very widely used in gene therapy. Their small genome is not very well adapted for large-scale modifications and easily loses heterologous inserts. A large number of mutations appear upon replication, which complicates drug standardization. Production is difficult to scale-up. Oncolytic therapy is the only field where RNA viruses are being used. Many RNA viruses, which are not very pathogenic for humans, replicate easily in cancer cells, but not in normal cells, and even unmodified viruses of different groups have a very strong oncolytic effect [32-35].
There are several examples of using herpes virus as a basis for gene therapy vectors. Being neurotropic, these viruses are mainly used for the development of gene therapy approaches for treatment of CNS dysfunctions [36].
NON-VIRAL DELIVERY
Non-viral delivery systems comprise DNA molecules (primarily plasmids) and RNA molecules, as well as autologous or allogeneic cells that have undergone genetic modification ex vivo before transplantation. One of the main problems of these systems, unlike their viral-based counterparts, is the delivery of gene therapy material to an organ and into cells.
The simplest examples are DNA fragments (most commonly, plasmids) that can express a gene that will induce the synthesis of a needed protein or interfering RNA in the cell. Special chemical and physical methods can be used to improve the efficiency of DNA delivery to the cell [37]. DNA-based constructs are relatively easy to generate and standardize. However, some negative aspects, such as low stability in the ambient environment and problematic targeted delivery to organs, as well as low stability in cells, and, thus, short-lasting effects, should be considered. DNA-based constructs are generally used in laboratory studies. They are also used for the development of DNA vaccines for veterinary use, because safety requirements for animal vaccines are less stringent than for human vaccines and the level of acceptable costs for development and production of vaccines is also lower.
RNA has a number of obvious drawbacks for use in gene therapy, and one of the major ones is low stability. Nevertheless, RNA molecules can still be used for certain gene therapy applications. For example, an efficient local short-lasting expression of therapeutic proteins can be achieved using RNA [38]. Progress in increasing stability and decreasing immunogenicity of RNA in vivo using modified nucleotides will allow broadening the spectrum of gene therapy using RNA [39].
GENE THERAPY CLINICAL TRIALS
Tens of thousands of articles on gene therapy are publisher each year (Fig. 1). A considerable number of these publications describe fundamental biological studies, where gene therapy is only mentioned as a potential application. The number of real clinical trials is several orders of magnitude lower, nevertheless significant.
The most obvious application of gene therapy is in the correction of monogenic diseases. The first attempt to cure β-thalassemia by means of gene therapy using a plasmid expressing normal hemoglobin was undertaken in 1980. This attempt failed because the transgene expression was only temporary. At that time, the procedure to obtain research approval was not appropriate, which discredited gene therapy for a long time [40]. Approximately in 1990 attempts to correct inherited immunodeficiencies through ex vivo transduction of hematopoietic stem cells with γ-retroviruses bearing a missing gene were done. The first study ended not quite successfully because of nonspecific integration and insertional oncogenesis. Five out of 20 patients developed leucosis, and one patient died. On the other hand, this attempt can be considered as rather successful, since two patients were partially and 17 patients were totally cured [16, 17]. The clinical cure rate and mortality after gene therapy was better than that after standard treatment by allogeneic bone marrow transplantation. In later studies with γ-retroviruses, the genotoxicity risk was reduced, and no leucosis development was detected, although an asymmetric expansion of transduced clones was observed, which might reflect the beginning of malignant transformation of lymphocytes. Now γ-retroviral vectors are almost totally replaced by lentiviral vectors, and for the majority of inherited immunodeficiencies the risk of genotoxicity is now much lower than the potential benefit for the patient.
Gene therapy of hematopoietic disorders and immunodeficiencies seems most promising, because it is possible to isolate hematopoietic stem cells (HSC) of a patient, to grow them ex vivo, to transduce them, to suppress the remaining HSC in the organism by medical treatment, and to inject transduced cells. Thus, cells bearing a genetic defect are efficiently replaced by transduced cells. For the majority of other genetic defects this scheme cannot be used, but this approach is suitable for treatment of genetic defects of neuroglia, since HSC can differentiate into neuroglia.
Retroviruses are the best choice for the transduction of dividing cells, such as stem cells, as they integrate into the genome and remain there indefinitely. If transduction ex vivo is not possible, non-integrating vectors should be used from the point of view of safety. Since cell division will lead to the disappearance of a transgene, infrequently dividing cells are the best target. In the 1990th, adenoviruses were considered as one of the main methods of transient expression. In 1999, an 18-year-old patient died after administration of an adenoviral vector aimed to correct ornithine-transcarbamylase deficiency [41]. It turned out that systemic administration of a high dose of adenovirus causes systemic inflammatory response. Although a number of methods to decrease such risk have been developed, adenoviruses became less popular (Fig. 2). At present, vectors based on adeno-associated viruses are preferentially used to assure the transient expression of a transgene in treatment of genetic and therapeutic diseases. Although this tool appeared rather recently, and by 2011, AAVs were used only for 10% of clinical trials, the first licensed drug for gene therapy is based on an AAV. The alipogene tiparvovec, marketed under the trade name Glibera – an AAV bearing a gene of lipoprotein lipase – is used for treatment of a rare form of hereditary dyslipidemia; EU granted marketing authorization for this drug in 2012.
The costs of treatment can be one of the factors slowing the development of gene therapy. For example, Glibera was announced to cost one million euros per treatment. Even taking into account the possibility of complete recovery of hopeless patients, which will allow avoiding expensive substitutive therapy, this price level represents a problem even for health care systems of major economies. One of the reasons why gene therapy is so expensive is that extremely expensive development and marketing authorization is divided by a small number of potential patients. One way to decrease costs of gene therapy would be to simplify gene therapy technique licensing and to work out scientific and legal background for the development of platform medicines using standard vector and production protocols.
Today many gene therapy trials are being conducted in the world (first of all, in the USA). In the ClinicalTrials.gov database, 485 clinical trials related to gene therapy are registered. Two hundred seventy-three trials are aimed at treatment of cancer. Such preponderance can be explained by much better risk/effect ratio for severely affected cancer patients. Only 186 trials (38%) use viral vectors, but the portion of trials using viral vectors is much higher among trials at phases II and III. Only 40 trials are aimed at HIV treatment. Only 24 trials reached phase III. There are just two approved drugs in the world up to now: Glibera (see above) and first generation oncolytic adenovirus (ONYX-15), authorized in China for local treatment of head and neck cancers [42], but recognized as unpromising in other countries.
REGULATORY ASPECTS OF CLINICAL APPLICATIONS
The constant technological progress is one of the main difficulties legal bodies responsible for registration and use of viral-based drugs are facing. New approaches for the development of viral vectors emerge constantly, and new data about virus–host interactions are accumulating. Thus, the possibility to change the legislation for registration and circulation of viral vector-based medicine according to the results of the latest research has to be foreseen.
Existing legislation regulating the development and circulation of drugs for gene therapy is based, generally, on legislation regulating the development and circulation of traditional drugs [43].
On one hand, such approach unifies procedures necessary to prove safety and efficiency of a new drug. On the other hand, this approach is characterized by strict regulation, which can create certain difficulties when some modifications in regulatory documentation should be made. It should also be mentioned that in different countries the legislation regulating the development and circulation of medical and healthcare products is much more detailed for traditional drugs. Introduction of special procedures regulating the development and circulation of GT products relies on legislation for classical drugs; in cases when such legislation is temporarily absent (as now in Russia), the regulatory base for the development and circulation of medicinal products is used [44].
Gene therapy technologies started to be developed already several decades ago. Despite the first seeming progress, developers have faced a whole range of technical problems limiting the application of GT products either because of insufficient safety, or because of its low efficiency. Nevertheless, the majority of technical problems can be solved, and the discovery of RNA interference mechanisms or directed genome editing systems has expanded even more the possible GT applications. Up to now, viral based vectors are considered as one of the most efficient means of nucleic acid delivery to a cell. We believe that in the future GT products will occupy a significant place among medical products used for treatment of a broad range of diseases.
In Russia, clinical trials of gene therapy are not conducted; moreover, it is not possible to produce pilot GT products because of the absence of legislation regulating clinical trials. At present, the annual number of publications on gene therapy by scientists from Russia is 30 times lower than the number of publications by scientists from the USA or China, and two times lower than that by scientists from Iran. Taking into account the perspectives of using GT technologies, maximum efforts should be made to overcome the critical lag of Russia in this field of molecular medicine.
We are grateful to R. A. Zinovkin for critical discussion.
This work was done with financial support by the Russian Foundation for Basic Research (project No. 15-04-99543).
REFERENCES
1.Rudchenko, M. N., and Zamyatnin, A. A., Jr. (2015)
Perspectives of using of self-assambling structures based on nicleic
acids, Biochemistry (Moscow), 80, 391-399.
2.Benenson, Y., Gil, B., Ben-Dor, U., Adar, R., and
Shapiro, E. (2004) An autonomous molecular computer for logical control
of gene expression, Nature, 429, 423-429.
3.Kahan-Hanum, M., Douek, Y., Adar, R., and Shapiro,
E. (2013) A library of programmable DNAzymes that operate in a cellular
environment, Sci. Rep., 3, 1535.
4.Jinek, M., Chylinski, K., Fonfara, I., Hauer, M.,
Doudna, J. A., and Charpentier, E. (2012) A programmable
dual-RNA-guided DNA endonuclease in adaptive bacterial immunity,
Science, 337, 816-821.
5.Savitskaya, E. E., Musharova, O. S., and Severinov,
K. V. (2016) Diversity of mechanisms of CRISPR-Cas systems of adaptiv
immunity of procaryotes and possibilities to use them in biotechnology,
Biochemistry (Moscow), 81, 653-661.
6.Drake, C. G., Lipson, E. J., and Brahmer, J. R.
(2014) Breathing new life into immunotherapy: review of melanoma, lung
and kidney cancer, Nat. Rev. Clin. Oncol., 11, 24-37.
7.Mellman, I., Coukos, G., and Dranoff, G. (2011)
Cancer immunotherapy comes of age, Nature, 480,
480-489.
8.Quantin, B., Perricaudet, L. D., Tajbakhsh, S., and
Mandel, J. L. (1992) Adenovirus as an expression vector in muscle cells
in vivo, Proc. Natl. Acad. Sci. USA, 89,
2581-2584.
9.Giacca, M. (2010) Gene Therapy,
Springer-Verlag, Italy.
10.Tsai, S. Y., Schillinger, K., and Ye, X. (2000)
Adenovirus-mediated transfer of regulable gene expression, Curr.
Opin. Mol. Ther., 2, 515-523.
11.Lyons, M., Onion, D., Green, N. K., Aslan, K.,
Rajaratnam, R., Bazan-Peregrino, M., Phipps, S., Hale, S., Mautner, V.,
Seymour, L. W., and Fisher, K. D. (2006) Adenovirus type 5 interactions
with human blood cells may compromise systemic delivery, Mol.
Ther., 14, 118-128.
12.Hendrickx, R., Stichling, N., Koelen, J., Kuryk,
L., Lipiec, A., and Greber, U. F. (2014) Innate immunity to adenovirus,
Hum. Gene Ther., 25, 265-284.
13.Raper, S. E., Chirmule, N., Lee, F. S., Wivel, N.
A., Bagg, A., Gao, G. P., Wilson, J. M., and Batshaw, M. L. (2003)
Fatal systemic inflammatory response syndrome in a ornithine
transcarbamylase deficient patient following adenoviral gene transfer,
Mol. Genet. Metab., 80, 148-158.
14.Nwanegbo, E., Vardas, E., Gao, W., Whittle, H.,
Sun, H., Rowe, D., Robbins, P. D., and Gambotto, A. (2004) Prevalence
of neutralizing antibodies to adenoviral serotypes 5 and 35 in the
adult populations of The Gambia, South Africa, and the United States,
Clin. Diagn. Lab. Immunol., 11, 351-357.
15.Roy, S., Clawson, D. S., Lavrukhin, O., Sandhu,
A., Miller, J., and Wilson, J. M. (2007) Rescue of chimeric adenoviral
vectors to expand the serotype repertoire, J. Virol. Methods,
141, 14-21.
16.Blaese, R. M., Culver, K. W., Miller, A. D.,
Carter, C. S., Fleisher, T., Clerici, M., Shearer, G., Chang, L.,
Chiang, Y., Tolstoshev, P., Greenblatt, J. J., Rosenberg, S. A., Klein,
H., Berger, M., Mullen, C. A., Ramsey, W. J., Muul, L., Morgan, R. A.,
and Anderson, W. F. (1995) T-lymphocyte-directed gene therapy for
ADA-SCID: initial trial results after 4 years, Science,
270, 475-480.
17.Bordignon, C., Notarangelo, L. D., Nobili, N.,
Ferrari, G., Casorati, G., Panina, P., Mazzolari, E., Maggioni, D.,
Rossi, C., Servida, P., Ugazio, A. G., and Mavilio, F. (1995) Gene
therapy in peripheral blood lymphocytes and bone marrow for
ADA-immunodeficient patients, Science, 270, 470-475.
18.Kohn, D. B., Weinberg, K. I., Nolta, J. A.,
Heiss, L. N., Lenarsky, C., Crooks, G. M., Hanley, M. E., Annett, G.,
Brooks, J. S., El-Khoureiy, A., Lawrence, K., Wells, D., Moen, R. C.,
Bastian, J., Williams-Herman, D. E., Elder, M., Wara, D., Bowen, T.,
Hershfield, M. S., Mullen, C. A., Blaese, R. M., and Parkman, R. (1995)
Engraftment of gene-modified umbilical cord blood cells in neonates
with adenosine deaminase deficiency, Nat. Med., 1,
1017-1023.
19.Cattoglio, C., Facchini, G., Sartori, D.,
Antonelli, A., Miccio, A., Cassani, B., Schmidt, M., Von Kalle, C.,
Howe, S., Thrasher, A. J., Aiuti, A., Ferrari, G., Recchia, A., and
Mavilio, F. (2007) Hot spots of retroviral integration in human
CD34+ hematopoietic cells, Blood, 110,
1770-1778.
20.Cattoglio, C., Pellin, D., Rizzi, E., Maruggi,
G., Corti, G., Miselli, F., Sartori, D., Guffanti, A., Di Serio, C.,
Ambrosi, A., De Bellis, G., and Mavilio, F. (2010) High-definition
mapping of retroviral integration sites identifies active regulatory
elements in human multipotent hematopoietic progenitors, Blood,
116, 5507-5517.
21.Wu, X., Li, Y., Crise, B., and Burgess, S. M.
(2003) Transcription start regions in the human genome are favored
targets for MLV integration, Science, 300, 1749-1751.
22.Hacein-Bey-Abina, S., Garrigue, A., Wang, G. P.,
Soulier, J., Lim, A., Morillon, E., Clappier, E., Caccavelli, L.,
Delabesse, E., Beldjord, K., Asnafi, V., Macintyre, E., Dal Cortivo,
L., Radford, I., Brousse, N., Sigaux, F., Moshous, D., Hauer, J.,
Borkhardt, A., Belohradsky, B. H., Wintergerst, U., Velez, M. C.,
Leiva, L., Sorensen, R., Wulffraat, N., Blanche, S., Bushman, F. D.,
Fischer, A., and Cavazzana-Calvo, M. (2008) Insertional oncogenesis in
4 patients after retrovirus-mediated gene therapy of SCID-X1, J.
Clin. Invest., 118, 3132-3142.
23.Montini, E., Cesana, D., Schmidt, M., Sanvito,
F., Ponzoni, M., Bartholomae, C., Sergi Sergi, L., Benedicenti, F.,
Ambrosi, A., Di Serio, C., Doglioni, C., Von Kalle, C., and Naldini, L.
(2006) Hematopoietic stem cell gene transfer in a tumor-prone mouse
model uncovers low genotoxicity of lentiviral vector integration,
Nat. Biotechnol., 24, 687-696.
24.Naldini, L., Blomer, U., Gallay, P., Ory, D.,
Mulligan, R., Gage, F. H., Verma, I. M., and Trono, D. (1996) In
vivo gene delivery and stable transduction of nondividing cells by
a lentiviral vector, Science, 272, 263-267.
25.Poeschla, E., Corbeau, P., and Wong-Staal, F.
(1996) Development of HIV vectors for anti-HIV gene therapy, Proc.
Natl. Acad. Sci. USA, 93, 11395-11399.
26.Iwakuma, T., Cui, Y., and Chang, L. J. (1999)
Self-inactivating lentiviral vectors with U3 and U5 modifications,
Virology, 261, 120-132.
27.Halbert, C. L., Miller, A. D., McNamara, S.,
Emerson, J., Gibson, R. L., Ramsey, B., and Aitken, M. L. (2006)
Prevalence of neutralizing antibodies against adeno-associated virus
(AAV) types 2, 5, and 6 in cystic fibrosis and normal populations:
implications for gene therapy using AAV vectors, Hum. Gene
Ther., 17, 440-447.
28.Tse, L. V., Moller-Tank, S., and Asokan, A.
(2015) Strategies to circumvent humoral immunity to adeno-associated
viral vectors, Expert Opin. Biol. Ther., 15, 845-855.
29.Rey-Rico, A., and Cucchiarini, M. (2016)
Controlled release strategies for rAAV-mediated gene delivery, Acta
Biomater., 29, 1-10.
30.Kotterman, M. A., and Schaffer, D. V. (2014)
Engineering adeno-associated viruses for clinical gene therapy, Nat.
Rev. Genet., 15, 445-451.
31.Sanchez-Sampedro, L., Perdiguero, B.,
Mejias-Perez, E., Garcia-Arriaza, J., Di Pilato, M., and Esteban, M.
(2015) The evolution of poxvirus vaccines, Viruses, 7,
1726-1803.
32.Hata, Y., Etoh, T., Inomata, M., Shiraishi, N.,
Nishizono, A., and Kitano, S. (2008) Efficacy of oncolytic reovirus
against human breast cancer cells, Oncol. Rep., 19,
1395-1398.
33.Goetz, C., Everson, R. G., Zhang, L. C., and
Gromeier, M. (2010) MAPK signal-integrating kinase controls
cap-independent translation and cell type-specific cytotoxicity of an
oncolytic poliovirus, Mol. Ther., 18, 1937-1946.
34.Borrego-Diaz, E., Mathew, R., Hawkinson, D.,
Esfandyari, T., Liu, Z., Lee, P. W., and Farassati, F. (2012)
Pro-oncogenic cell signaling machinery as a target for oncolytic
viruses, Curr. Pharm. Biotechnol., 13, 1742-1749.
35.Esfandyari, T., Tefferi, A., Szmidt, A., Alain,
T., Zwolak, P., Lasho, T., Lee, P. W., and Farassati, F. (2009)
Transcription factors down-stream of Ras as molecular indicators for
targeting malignancies with oncolytic herpes virus, Mol. Oncol.,
3, 464-468.
36.Kantor, B., Bailey, R. M., Wimberly, K.,
Kalburgi, S. N., and Gray, S. J. (2014) Methods for gene transfer to
the central nervous system, Adv. Genet., 87, 125-197.
37.Alsaggar, M., and Liu, D. (2015) Physical methods
for gene transfer, Adv. Genet., 89, 1-24.
38.Sergeeva, O. V., Koteliansky, V. E., and
Zatsepin, T. S. (2016) Using mRNAs for therapy – achivements
and perspectives, Biochemistry (Moscow), 81, 709-722.
39.Li, B., Luo, X., and Dong, Y. (2016) Effects of
chemically modified messenger RNA on protein expression, Bioconj.
Chem., 27, 849-853.
40.Wade, N. (1981) Gene therapy caught in more
entanglements, Science, 212, 24-25.
41.Marshall, E. (1999) Gene therapy death prompts
review of adenovirus vector, Science, 286, 2244-2245.
42.Yu, W., and Fang, H. (2007) Clinical trials with
oncolytic adenovirus in China, Curr. Cancer Drug Targets,
7, 141-148.
43.Mironov, A. N., Vasiliev, A. N., Goryachev, D.
V., Gavrishina, E. V., and Niyasov, R. R. (2014) Medicines for advanced
therapies: scientific approaches to validate quality, safety and
efficacy, Remedium, Nos. 7-8, 16-24.
44.Galli, M. C., and Serabian, M. (2014) Regulatory
aspects of gene therapy and cell therapy products: a global
perspective, Adv. Exp. Med. Biol., 871, 1-221.
45.Villemejane, J., and Mir, L. M. (2009) Physical
methods of nucleic acid transfer: general concepts and applications,
Br. J. Pharmacol., 157, 207-219.