REVIEW: Inflammatory Immune Infiltration in Human Tumors: Role in Pathogenesis and Prognostic and Diagnostic Value
A. V. Bogolyubova1,2* and P. V. Belousov1,2
1Engelhardt Institute of Molecular Biology, Russian Academy of Sciences, 119991 Moscow, Russia; E-mail: apollinariya.bogolyubova@gmail.com2Lomonosov Moscow State University, Biological Faculty, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received June 23, 2016; Revision received July 13, 2016
The cellular microenvironment directly and indirectly influences tumor development and possesses prognostic and in some cases diagnostic value. Over the years, understanding of structural organization of the immune/inflammatory moiety of neoplasms as well as in-depth phenotypic and transcriptomic profiling of its cellular components together provide more and more insights in both basic and translational medical science. In this review, we will discuss the specific roles of various stromal cells and their impact on neoplastic progression as well as address the use of quantitative and phenotypic analysis of immune/inflammatory infiltrate for diagnostics and predicting the clinical course of human malignancies.
KEY WORDS: cellular microenvironment of tumors, immune infiltration, melanoma, breast cancer, thyroid cancer, colorectal cancerDOI: 10.1134/S0006297916110043
Abbreviations: BC, breast cancer; CTL, cytotoxic T lymphocytes; IFN, interferon; IL, interleukin; ILC, innate lymphoid cells; MDSC, myeloid-derived suppressor cells; MSI, microsatellite instability; NCR, natural cytotoxicity receptors; Tregs, regulatory T cells.
Immune cells represent an integral component of the microenvironment of
neoplastic tissues. After entering a tumor, these cells may produce a
variety of proinflammatory cytokines, thereby maintaining chronic
inflammation and stimulating tumor growth and progression (e.g.
tumor-infiltrating macrophages are well-known to play role in
development of head and neck, gastric, colon and other cancers [1]). At the same time, various scenarios of antitumor
immune defense (antibody production, contact and complement-dependent
cytolysis, etc.) are executed primarily by the cells of lymphoid
lineage (T, B, NK and NKT cells) that dampen tumor growth in accordance
with the theory of immunological surveillance proposed by Burnet in the
1960s [2] and further transformed into cancer
immunoediting theory by Schreiber [3-5]. Thus, both the intensity and cellular composition
of immune infiltrate are important for deciphering of its prognostic
and diagnostic value.
IMMUNE CELLS INFILTRATING HUMAN TUMORS
One can find virtually any type of immune cells in tumor microenvironment [6], but in terms of influence on neoplastic progression only a few types were explicitly studied – namely macrophages, dendritic cells, and myeloid-derived suppressor cells (MDSC) representing myeloid lineage, as well as lymphoid cells such as T, mature B and plasma cells, as well as innate lymphoid cells (ILC) including NK cells.
In solid neoplasms, macrophages comprise a significant portion of tumor tissue [7]; however, ratio of M1- and M2-polarized macrophages varies largely depending on tumor histogenesis, stage, etc. [8, 9]. M2-polarized macrophages (NOS2–CD163+) [10] may be in vitro differentiated from monocytes in the presence of IL-4, IL-10, and IL-13 [11] and are able to suppress immune reactions; they are commonly detected in tumor tissues [12]. It is believed that M2-macrophages contribute to tumor growth and progression via local suppression of the immune response at the site of neoplastic transformation (e.g. production of high amounts of suppressive cytokine IL-10 in kidney cancer [13], suppression of activity of melanoma-infiltrating cytotoxic T cells [14], recruitment of regulatory T cells to neoplastic site in colorectal carcinoma [15], etc.). Zhang et al. [1] demonstrated negative prognostic effect of the presence of tumor-associated macrophages in gastric, breast, bladder, ovarian, thyroid cancer and laryngeal carcinoma; a similar phenomenon was also observed in cervical cancer [16]. At the same time, Zhang et al. [1] noted a positive correlation between macrophage infiltration and overall survival in patients with colorectal cancer, probably due to increased amount of M1-polarized macrophages (NOS2+CD163–) recruiting Th1 cells to the tumor site. Prognostic value of the presence of tumor-infiltrating macrophages also depends on their location inside the tumor as well as on the expression of phagocytosis-hindering molecule CD47 by colorectal cancer cells [17].
Dendritic cells being “professional” antigen-presenting cells are often found in tissues of various malignant neoplasms (melanoma [18], colon cancer [19], thyroid cancer [20], etc.). These cells can capture antigens releasing from apoptotic or necrotic tumor cells and present them to T cells, thereby triggering antitumor immune response [21]. However, this scenario requires the presence of proinflammatory microenvironment, in particular high concentrations of cytokines IFN-γ and TNF allowing maturation and consequently efficient functioning of dendritic cells. However, tumor tissues are usually characterized by increased amounts of IL-10, IL-6, M-CSF, etc. Despite clear proinflammatory activity demonstrated by some of them, the particular cytokine profile may inhibit maturation of tumor-infiltrating dendritic cells. It is known that immature dendritic cells present only small amounts of antigen in the absence of costimulatory signals, thereby leading to T cell anergy and development of immune tolerance for neoplastic cells-derived antigens [22]. Consequently, the presence of immature dendritic cells in the immune infiltrate of cancers is considered as a prognostically unfavorable factor (e.g. in non-small cell lung cancer [23], etc.), as it seems to contribute to evasion of the tumor from immune surveillance.
Myeloid-derived suppressor cells (MDSC) comprise a group of myeloid precursor cells (enriched in Lin–HLA-DR–CD33+ cell population) as well as immature mononuclear cells (CD14+HLA-DR–/lo), morphologically similar to monocytes, and immature polymorphonuclear cells (CD14–CD11b+CD33+CD15+ and CD66b+) [24]. MDSC can promote growth of primary tumors and developing of metastatic disease via protecting of tumor cells from the action of immune effectors, participating in remodeling of the tumor microenvironment and formation of a premetastatic niche. The majority of studies on the influence of MDSC on tumor growth were performed in murine models; however, there is also data describing a role of these cells in tumor microenvironment in humans. It particular, cells with MDSC phenotype displayed no immunosuppressive activity at the early stages of human lung cancer; probably, such cells are accumulated within tumor tissue in response to inflammatory stimuli and only later begin to influence tumor progression [25].
A requirement for simultaneous detection of multiple markers to reliably identify MDSC inside tumor tissues largely precludes their detection using the immunohistochemistry technique; therefore, only a few studies aimed at examining tumor-infiltrating MDSC in humans have been reported. Nonetheless, some data suggest the association between accumulation of MDSC within tumor stroma and poor prognosis in patients with a broad range of malignant neoplasms [26].
Preclinical and clinical studies suggest that detection of tumor-infiltrating lymphocytes may be associated with favorable prognosis in a broad range of solid tumors [27]. However, not all lymphocyte subsets exhibit antitumor activity [28]. A significant portion of tumor-infiltrating T cells is represented by antigen experienced (via mature dendritic cells in tumor-draining lymph nodes) cytotoxic T lymphocytes, recruited in neoplastic tissue by chemotactic molecules released by tumor and stromal cells [29]. Activated cytotoxic T lymphocytes (CTL) then are capable of destroying target cells via perforin/granzyme-dependent contact cytolysis.
Regulatory T cells (Tregs) characterized by membrane expression of various suppressor molecules (PD-1, CTLA-4, TIM-3, LAG-3, etc.) and secreting various suppressor cytokines represent another important subset of tumor-infiltrating T lymphocytes [30, 31]. After entering a tumor, Tregs exert suppressive effects on other immune cells residing inside tumor tissues, thereby promoting accelerated tumor progression [32]. However, some data suggest that the presence of Tregs correlates with favorable prognosis for patients with ovarian carcinoma, bladder cancer [33], head-and-neck cancer [34], and colorectal cancer [35]. This is probably related to the fact that depending on microenvironment and particular stage of a tumor, regulatory T cells may block antitumor activity of effector T cells, as well as lower the level of local protumorigenic inflammation, thereby resulting in opposite outcomes in these two scenarios [33].
Mature B cells and terminally differentiated plasma cells are also present in tumor stroma, being mostly abundant in medullary breast cancer and Warthin-like papillary thyroid carcinoma. The spatial distribution of mature B cells follows either diffuse or localized patterns, the latter being represented by B cell zones in the so-called tertiary lymphoid structures. Despite the fact that in many cancers a positive correlation between intensity of B cell infiltrate and survival rate was demonstrated, a pro- and anti-tumorigenic roles of B cells in each particular type of malignant neoplasms are still debated [36].
B cells may participate in maintaining chronic inflammation [37] as well as directly inhibit cytotoxic activity of T cells [38] and recruit Tregs to the sites of neoplastic growth by producing TGF-β and IL-10 [39, 40] (this data was obtained in murine models). However, tumor-infiltrating B cells in patients with metastatic melanoma demonstrate clonal amplification, somatic hypermutation, and immunoglobulin isotype switch, suggesting their involvement in antigen-dependent antitumor immune response [41]. Moreover, B cells may serve as efficient antigen-presenting cells to activate T lymphocytes and generate memory T cell pool [42] as well as reactivate T cells pre-primed by dendritic cells [43] (shown in murine models). In the case of hepatocellular carcinoma, tumor-infiltrating B cells were shown to exert independent cytolytic activity against cancer cells via granzyme B- and TRAIL-mediated mechanisms [44].
Recently, more attention has been given to tertiary lymphoid structures (TLS) – cell aggregates structurally resembling lymphoid follicles of the secondary lymphoid organs, usually located in the periphery of tumor growth at the invasion border. These structures contain a T-cell zone where T cells cluster with mature dendritic cells exerting efficient antigen presentation, and a B cell zone containing, in particular, naïve CD20+ B cells [45]. Moreover, a few macrophages as well as plasma cells may be found within the TLS [46]. These cell aggregates are highly vascularized, which is necessary for efficient immune response and probably play a central role in immune infiltration of tumor tissues and TLS development [36, 47]. Available data implicate TLS development as one of the efficient strategies for immune control of growth and progression of malignant neoplasms [48].
Innate lymphoid cells (ILC) represent a heterogeneous group of cell types that, according to experimental data obtained using murine models, may either promote or suppress tumor growth. Three types of ILC are usually distinguished: ILC1 (including classic NK cells), ILC2, and ILC3, each being characterized by a range of surface markers and produced cytokines. With exception of NK-cells, other ILC types were not studied in sufficient details to date. Nonetheless, it was demonstrated, that amount of NKp46/NKp30/NKp44 (members of the Natural Cytotoxicity Receptors family, NCR)-negative ILC1 and ILC2 did not differ between normal lung and non-small cell lung carcinoma tissues, whereas the amount of ILC3 in tumor tissue was substantially lower than in normal tissue. In contrast, NCR+ILC3 were found at much higher levels in tumor vs. normal tissue due to their selective enrichment within the tumor site. Upon activation, NCR+ILC3 can produce a broad range of cytokines with proinflammatory activity (IL-22, TNF), as well as those recruiting leukocytes and promoting their proliferation at the site of neoplastic transformation (IL-8, IL-2) [49].
The presence and intensity of NK cell infiltration in tumor stroma may have an impact on survival of patients with solid tumors, which was demonstrated in colorectal cancer [50], squamous cell lung cancer [51], etc. At the same time, the magnitude of NK cell antitumor activity significantly depends on cytokine profile within tumor tissues, and large amounts of NK cells in tumor stroma may not be proportional to their actual antitumor activity.
In the next section, we will discuss four types of malignant neoplasms wherein lymphocytic infiltrate is rather typical feature and/or was shown to play an important prognostic and diagnostic role.
MELANOMA
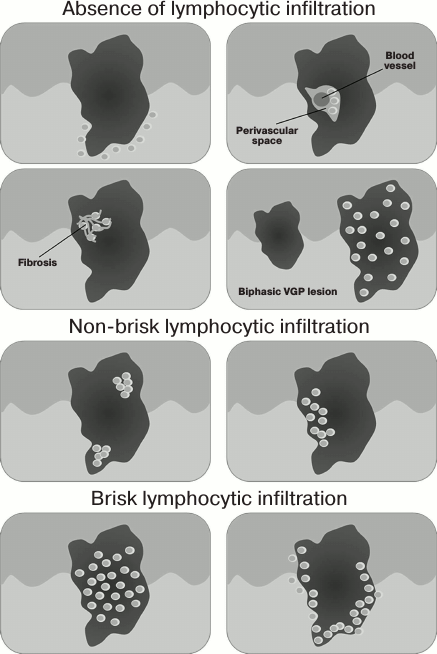
Lymphocytic infiltrate is found only in malignant melanocytic human neoplasms; therefore, it may serve as one of histological criteria to distinguish melanoma vs. melanocytic nevi. In 1969, Clark et al. described the presence of the immune cells in human skin melanoma [52], whereas in 1985 Elder et al. [53] proposed to classify immune infiltrate in melanomas according to the density and localization of immune cells as brisk, non-brisk, and absent (the good inter-rater agreement in assessing this criterion was demonstrated by Busam et al. [54]). These types of immune infiltration in the vertical growth phase of melanoma are schematically depicted in Fig. 1.
Fig. 1. Scheme depicting various types of immune infiltration in vertical growth phase of melanoma (according to Schatton et al. [55]).
Later, prognostic significance of melanoma-infiltrating immune cells was thoroughly investigated. In particular, Tuthill et al. [56] examined nine potential clinical and pathological prognostic factors in the cohort of 259 patients with localized primary melanoma. They found that the density of lymphocytic infiltrate was the most significant predictor of patients’ survival (p = 0.005). Moreover, 5- and 10-year survival rates were 100 and 93% vs. 71 and 55% in patients with brisk vs. absent lymphocytic infiltrate, respectively. In another cohort of 285 patients with localized primary melanoma, Clemente et al. [57] showed that 5- and 10-year survival rates were 77 and 55%, 53 and 45%, and 37 and 27% for patients with brisk, non-brisk, and absent lymphocytic infiltrate, respectively. Moreover, it was demonstrated that the abundance of immune cells in the primary tumor inversely correlated with the probability of developing metastatic disease in regional lymph nodes [57]. Furthermore, in the cohort of 99 patients with metastatic melanoma, Mihm et al. showed that 30-month event-free survival was higher in patients with brisk vs. non-brisk or absent lymphocytic infiltrate in melanoma metastases (81.3, 46.8, and 29.3%, respectively, p = 0.007) [58].
The cellular composition of melanoma lymphocytic infiltrate can significantly vary: studies performed using melanoma-infiltrating lymphocytes cultured in the presence of IL-2 demonstrated that CD4+/CD8+ T cell ratio may change within a broad range depending on the particular tumor specimen [59]. However, the majority of studies were focused on studying CD8+ T cells, as well as their antigenic specificity and cytotoxic activity both in vitro and in vivo. Effector T cell responses may be directed against epitopes derived from tumor antigens presented on MHC class I molecules; activated CD8+ T cells may then kill transformed cells via contact cytolysis [60]. Such antigens may be represented by autologous proteins with point mutations (e.g. β-catenin) and aberrantly expressed proteins (e.g. MAGE family antigens). In addition, many melanoma-specific antigens are normal proteins involved in melanin synthesis, including MART-1 (specific immune response against this protein is often detected in patients with melanoma), gp100, TRP-1, TRP-2, and tyrosinase [61, 62]. However, several studies reported that due to tumor suppressive microenvironment, infiltrating CD8+ T cells are mostly in inactive/anergic state [63-65].
On the other hand, melanocytic tumors are also infiltrated with CD4+ T cells, which due to their plasticity may exhibit both pro- and anti-inflammatory activity [66, 67]. Immunophenotyping of lymphocytic infiltrate demonstrated that the composition of CD4+ T cells may vary significantly depending on tumor size: in tumors with small thickness, the majority of CD4+ T cells secreted cytokines typical for Th1 phenotype – namely, IFN-γ, LTα, IL-2, and IL-15, whereas in tumors with larger thickness, these cells mostly produced TGF-β and IL-10 characteristic of Th2/Treg phenotype, thereby exerting immunosuppressive effects on tumor microenvironment [68, 69]. It was shown that melanoma antigen-specific CD4+ T cells can directly lyse transformed cells via an unknown mechanism [70]. Moreover, adoptive co-transfer of CD4+ and CD8+ T cells was more efficient compared to CD8+ T cells alone [71], and a long-term clinical response after adoptive transfer of NY-ESO-1-specific CD4+ T cells was also observed in a patient with metastatic melanoma [72].
B cell infiltrate in melanocytic tumor stroma follow either diffuse pattern or present in the form of aggregates resembling lymphoid follicles. It was noted that the presence of such infiltrate is associated with more favorable prognosis in melanoma patients [73]. Moreover, several studies demonstrated strong association between the presence of NY-ESO-1 – specific antibodies and NY-ESO-1-specific CD8+ T cells in peripheral blood of patients, both spontaneously [74] and in the context of NY-ESO-1 – specific immunotherapy. Apparently, tumor-specific antibodies opsonize tumor cells, contributing to their phagocytosis and antigen presentation, which in turn influences the activation of CD8+ T cells and better therapeutic outcome.
BREAST CANCER
Breast cancer (BC) is the most common cancer in women, with more than 1,380,000 new cases being diagnosed each year worldwide [75]. Several studies demonstrated that the intensity of lymphocytic infiltrate in tumor stroma may be a prognostically favorable sign regardless of its cellular composition in triple negative (ER–PR–HER2–) [76, 77] and HER2+ [78] BC. Moreover, patients with triple negative and HER2+ BC demonstrating abundant immune infiltration responded better to therapy with anthracyclines [77] and trastuzumab [79], respectively.
One can find virtually any type of immune cells in tissues of breast carcinomas, and many of them were implicated in better prognosis. The presence of CD8+ T cells [80], CD4+ T cells and CD20+ B cells [81, 82] is a predictor of complete pathological response to neoadjuvant therapy, whereas the presence of γδ T cells is associated with an increased amount of Tregs and consequently poor prognosis, tumor spread, and low survival [83]. The presence of T follicular helper cells (TFH) is associated with strong response to chemotherapy [84]. Perhaps this is related to the fact that TFH are found at the tumor border in tertiary lymphoid structures, which may represent the main sites concentrating cells exhibiting antitumor activity.
One of BC types known as medullary breast cancer is diagnosed in 3-5% of all BC patients. [85]. Despite a marked cancer cell anaplasia, this neoplasm is characterized by a very favorable prognosis (10-year survival reaches 84% compared to 63% in other infiltrative duct carcinomas [86]), which seems to be related to extremely abundant lymphocytic infiltration of tumor stroma.
Several hypotheses regarding the influence of the immune system on neoplastic cells in medullary breast cancer do exist. For instance, favorable prognosis of this BC type may be related to enhanced apoptosis of tumor cells [87], upregulated expression of factors inhibiting tumor metastatic activity [88] and the ICAM-1 molecule [89].
Lymphocytic infiltrate in medullary breast cancer was found to contain T, B, and plasma cells, with the apparent predominance of CD8+ T cell [90]. The lack of histiocytes (tissue macrophages and dendritic cells) and neutrophils suggests that lymphocytes accumulate within tumor tissues in response to certain specific stimuli (e.g. antigen-specific) rather than due to “nonspecific” inflammatory reaction against tumor necrotic cells or bacterial agents [91].
Several studies were aimed in identification of antigenic targets of the anti-tumor B cell response in this rare BC type. Using a library of variable immunoglobulin fragments derived from tumor-infiltrating plasma cells, delineating their specificity followed by FACS and ELISA assays, Kotlan et al. identified ganglioside D3 as a specific tumor-associated antigen of medullary BC [92]. Following demonstration of oligoclonality of tumor-infiltrating plasma cells, Ditzel et al. constructed and screened a cDNA-expression library of hypervariable IgH fragments to identify cytoskeletal protein β-actin as a specific antigenic target of medullary BC. This protein is apparently exposed on the surface of apoptotic bodies of medullary BC cells and undergoes a tumor-specific proteolysis involving granzyme B, thereby potentially predisposing it for being recognized by B-lineage cells [93].
THYROID CANCER
According to Tamai et al., a considerable proportion (~38%) of thyroid cancers (papillary and follicular types) contains an abundant lymphocytic infiltrate [94]. It was shown that a large number of tumor-infiltrating lymphocytes found in thyroid neoplastic tissues correlates with lower invasiveness, lowered risk of occurring distant metastases and recurrences, and consequently better patients’ survival. It was shown that the presence of lymphocytic infiltration is associated with significantly lower recurrence rate (2.8 vs. 18.6% for tumors with no infiltration). Although in both groups tumor size and frequency of metastatic disease in draining lymph nodes were similar, the degree of tumor invasion was higher in patients containing no lymphocytic infiltration [94].
By contrast, the presence of lymphocytic infiltrate had no impact on event-free survival (probably due to overall indolent behavior of differentiated thyroid neoplasms); however, this value was improved in patients infiltrated with proliferating lymphocytes [95]. Furthermore, the rate of long-term survival correlated with the presence of intratumoral phagocytes and dendritic cells capable of capturing and presenting tumor antigens to T lymphocytes, thereby activating them [96]. Interestingly, it was also reported that subpopulation contents of T lymphocytic pools in peripheral blood and intratumoral infiltrate were virtually identical; however, in functional tests, the latter demonstrated higher cytolytic activity, thus suggesting the participation of T cells in immune reactions against neoplastic cells [97].
Expression of co-stimulatory molecules on the surface of thyrocytes both in autoimmune (Graves disease and Hashimoto thyroiditis [98]) and neoplastic (follicular and papillary-patterned neoplasms [99]) conditions suggests that they are involved in activating specific T cell immune response against antigens associated with autoimmune disease and tumor progression.
Dendritic cells are not found in normal thyroid gland, but they can be recruited by chemokines produced during tumor growth or Hashimoto thyroiditis [100]. It was found that the majority of dendritic cells infiltrating thyroid tumor tissues demonstrated immature phenotype, since IL-10, IL-6, M-CSF, VEGF and gangliosides produced by neoplastic cells suppress their maturation and promote induction of apoptosis [101]. Immature dendritic cells may activate Tregs, thereby enhancing tumor-associated immunosuppression as well as secrete reactive oxygen species directly inhibiting T cell activity. Thus, in thyroid cancer, efficient presentation of tumor antigens probably occurs at low levels, as functioning of professional antigen-presenting cells is profoundly reduced; however, this function may be directly exerted by tumor cells per se [102].
In 1955, Dailey was the first to note the association between Hashimoto thyroiditis and thyroid cancer [103]; however, studies aimed at investigation of an impact of Hashimoto thyroiditis on the course and prognosis of thyroid carcinomas were published much later. It was observed that the presence of Hashimoto thyroiditis positively correlates with favorable prognosis in patients with thyroid cancer, but the same data suggest a potential role of chronic inflammation in triggering the growth of neoplastic tissue [22]. Kashima et al. studied 1533 cases of papillary thyroid cancer and demonstrated higher rates of relapse-free survival in a subgroup of 281 (18%) patients with Hashimoto thyroiditis [104]. Further studies showed that thyroid cancer patients with coexistent Hashimoto thyroiditis [105] demonstrated lower rates of extra-thyroid extension, regional and distant metastasis, as well as substantially lower risk of recurrence and mortality rate [106].
A question about cause/effect relationship between Hashimoto thyroiditis and thyroid cancer remains debated. On one hand, the presence of Hashimoto thyroiditis was shown to increase the risk of developing thyroid cancer [106]. Increased apoptosis and proliferation of thyrocytes during thyroiditis can increase risk of DNA damage and, subsequently, results in tumor development [107]. Arif et al. reported 21 cases of Hashimoto thyroiditis harboring RET/PTC oncogenic rearrangements, whereas only six of these cases demonstrated histological evidence of papillary microcarcinoma [108]. Thus, one may suggest that Hashimoto thyroiditis precedes development of thyroid cancer. On the other hand, Mechler et al. performed a similar study analyzing six cases of familial papillary thyroid cancer coincident with Hashimoto thyroiditis [109]; in this study, RET/PTC rearrangements were found only in tumor but not in surrounding healthy tissues.
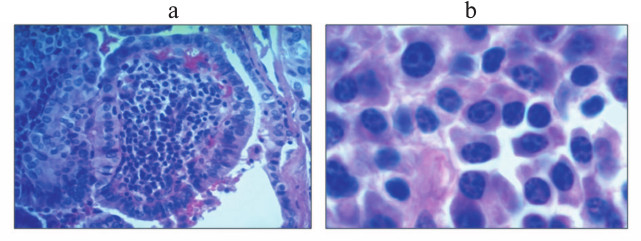
Warthin-like papillary thyroid carcinoma, named for its histological similarity with the Warthin’s tumor of salivary gland, is a relatively rare tumor characterized by especially abundant immune infiltration of tumor stroma [110]; immune cells in this tumor may account for up to 85% of total tumor cell count (Bogolyubova et al., unpublished data; see hematoxylin and eosin staining in Fig. 2). Interestingly, Warthin’s tumor and Warthin-like papillary thyroid carcinoma, as well as medullary breast cancer mentioned hereinbefore are the only neoplasms when abundance of immune cells infiltrating tumor stroma serves as a diagnostic criterion. An in-depth analysis of subset composition of lymphocytic infiltrate of this tumor had not been studied in sufficient details, particularly due to limited number of cases published to date (~200 cases). The potential role of the immune infiltrate in pathogenesis of this tumor remains controversial, although there are reasons to believe that these cells may be specific to tumor antigens and might take part in restraining tumor progression (Bogolyubova et al., unpublished data).
Fig. 2. Section of Warthin-like papillary thyroid carcinoma (stained with hematoxylin and eosin): a) low magnification (100×); b) high magnification (400×).
IMMUNOGENICITY OF COLORECTAL CANCER AND ITS PROGNOSTIC
SIGNIFICANCE
The immunogenicity of a subset of colorectal carcinomas demonstrating microsatellite instability (MSI+) was first examined in the mid-to-late 1990s [111-113]. Both hereditary- (developing in MLH1, MSH2, MSH6, or PMS2 germline mutation carriers, Lynch syndrome) and sporadic-type (primarily demonstrating promoter hypermethylation in MLH1 gene) MSI+ colorectal carcinomas are usually represented by high-grade tumors; despite of this fact, MSI+ tumors are characterized by markedly more favorable prognosis compared to MSI– tumors. A marked inflammatory reaction occurring in tumor stroma [112, 113] being absent in the majority of MSI– tumors is believed to account for atypically favorable prognosis in these histologically highly malignant tumors. Intra-epithelial lymphocytes in MSI+ vs. MSI– tumors demonstrate phenotype of activated cytotoxic effector T cells (CD3+CD8+TIA-1/GMP-17+); their numbers are several fold higher than the total number of CD3+, CD8+, and GrB+ cells, as well as the percentage of GrB+ cells among CD3+ and CD8+ T cell subsets in MSI– tumors [111, 114].
Mechanistically, the phenomenon observed in MSI+ tumors may be explained by defects in the mismatch repair system that result in dramatically increased rate of somatic mutations (single nucleotide substitutions, small insertions and deletions). This in turn results in a synthesis of a large number of frameshift peptides, the peptides bearing unique C-terminal sequences never found in vivo except in MSI+ tumors, which predisposes its recognition by the adaptive immune system. In particular, generation of both T [115-118] and B cell [119, 120] responses against such peptides was demonstrated in many studies. It seems that these “patient-specific” frameshift peptides comprise a major pool of antigens recognized by effector immune cells in patients with MSI+ tumors.
Nonetheless, in order for a tumor to grow and progress, such an extensive immune response should be counter-balanced by immunosuppressive factors produced by tumor cells and their microenvironment. Llosa et al. [121] demonstrated specific overexpression of immunological checkpoint molecules such as PD-1, PD-L1, CTLA-4, LAG-3, and IDO, lacking in MSI– tumors, thus rendering patients with MSI+ tumors as excellent candidates for checkpoint blockade immunotherapy successfully applied in patients with melanoma [122, 123]. Based on phase II clinical trial of pembrolizumab (a PD-1 blocker) in patients with MSI+ tumors, this approach is highly promising [124].
Collectively, MSI+-colorectal carcinomas comprise not more than 15% of all colorectal cancers generally considered as poorly immunogenic tumors. In 2006, Galon et al. [125] studied density, type, and location of tumor-infiltrating lymphocytes in a large cohort of patients with colorectal cancer regardless of MSI. The quite predictable results on improved survival of patients with abundant vs. modest CD3+ T lymphocytic infiltrate turned out to be rather intriguing upon considering location of tumor-infiltrating lymphocytes (intra- and peritumoral), additional specific detection of memory CD45RO+ T cells, and comparison of prognostic value of these parameters with tumor staging according to UICC TNM. In particular, it was shown that patients with stage I-III tumors abundantly infiltrated with CD3+ T cells in the central and peripheral zones demonstrate significantly better survival rates than the stage-matched patients with tumors showing poor lymphocytic infiltration of the same areas; importantly, the survival rate within each group (abundant/poor infiltration) showed no correlation with TNM stage.
Even more striking were results obtaining using the same approach after additional analysis of density of memory CD45RO+ T cells. It was found that 10-year survival of patients with high abundance of both CD3+ and CD45RO+ cells within tumor stroma appeared to be more than 80% irrespectively of TNM stage (I, II or III), whereas all patients with poor
CD3+ and CD45RO+ infiltration died within 1 year, including patients with TNM stage I tumors, generally considered to be quite indolent and potentially completely curable. Thus, for the first time this study demonstrated high and, to some extent, TNM-staging independent prognostic value of phenotype of intratumoral lymphocytic infiltration. Later, Galon et al. formalized the density of immune infiltrate by introducing an ImmunoScore parameter varying from 0 to 4 and verified previous data demonstrating its high prognostic value that was superior to the TNM system in predicting tumor recurrences and distant metastasis [126-128]. In 2011, an international task force was organized aimed at multi-center evaluation of prognostic value of ImmunoScore and potentially groundbreaking introduction of the relevant additional parameter into UICC TNM staging (TNM-I) [129].
Finally, in 2016 Mlecnik et al. [130] reported that ImmunoScore provided better prognostic results compared to microsatellite instability analysis, which might be used to select patients for checkpoint blockade immunotherapy.
Here we discussed three types of malignant neoplasms in humans (melanoma, medullary breast cancer and papillary thyroid cancer) that were shown to display an association between abundant lymphocytic infiltrate and favorable prognosis. The subset composition of immune infiltrate varies over a wide range depending on multiple factors including yet unknown parameters; depending on activation stage and cytokine milieu, each immune cell subset may exert either suppressive or activating effects on tumor growth and progression. The in-depth characterization of immune infiltrate (ImmunoScore) developed for patients with colorectal cancer could allow a new look at assessing prognosis and selection of treatment strategy for other tumor types.
Acknowledgements
We are thankful to Prof. D. V. Kuprash for valuable suggestions and productive discussions during preparation of our manuscript.
This study was conducted with financial support by the Russian Science Foundation (project No. 16-15-10423).
REFERENCES
1.Zhang, Q., Liu, L., Gong, C., Shi, H., Zeng, Y.,
Wang, X., Zhao, Y., and Wei, Y. (2012) Prognostic significance of
tumor-associated macrophages in solid tumor: a meta-analysis of the
literature, PLoS One, 7, e50946.
2.Fridman, W. H., Galon, J., Dieu-Nosjean, M.-C.,
Cremer, I., Fisson, S., Damotte, D., Pages, F., Tartour, E., and
Sautes-Fridman, C. (2011) Immune infiltration in human cancer:
prognostic significance and disease control, Curr. Top. Microbiol.
Immunol., 344, 1-24.
3.Dunn, G. P., Bruce, A. T., Ikeda, H., Old, L. J.,
and Schreiber, R. D. (2002) Cancer immunoediting: from
immunosurveillance to tumor escape, Nat. Immunol., 3,
991-998.
4.Dunn, G. P., Old, L. J., and Schreiber, R. D.
(2004) The three Es of cancer immunoediting, Annu. Rev.
Immunol., 22, 329-360.
5.Schreiber, R. D., Old, L. J., and Smyth, M. J.
(2011) Cancer immunoediting: integrating immunity’s roles in
cancer suppression and promotion, Science, 331,
1565-1570.
6.Schiavoni, G., Gabriele, L., and Mattei, F. (2013)
The tumor microenvironment: a pitch for multiple players, Front.
Oncol., 3, 90.
7.Hao, N.-B., Lu, M.-H., Fan, Y.-H., Cao, Y.-L.,
Zhang, Z.-R., and Yang, S.-M. (2012) Macrophages in tumor
microenvironments and the progression of tumors, Clin. Dev.
Immunol., 948098.
8.Edin, S., Wikberg, M. L., Dahlin, A. M., Rutegard,
J., Oberg, A., Oldenborg, P.-A., and Palmqvist, R. (2012) The
distribution of macrophages with a M1 or M2 phenotype in relation to
prognosis and the molecular characteristics of colorectal cancer,
PLoS One, 7, e47045.
9.Zhang, M., He, Y., Sun, X., Li, Q., Wang, W., Zhao,
A., and Di, W. (2014) A high M1/M2 ratio of tumor-associated
macrophages is associated with extended survival in ovarian cancer
patients, J. Ovarian Res., 7, 19.
10.Edin, S., Wikberg, M. L., Oldenborg, P.-A., and
Palmqvist, R. (2013) Macrophages: good guys in colorectal cancer,
Oncoimmunology, 2, e23038.
11.Martinez, F. O., Helming, L., and Gordon, S.
(2009) Alternative activation of macrophages: an immunologic functional
perspective, Annu. Rev. Immunol., 27, 451-483.
12.Solinas, G., Germano, G., Mantovani, A., and
Allavena, P. (2009) Tumor-associated macrophages (TAM) as major players
of the cancer-related inflammation, J. Leukoc. Biol., 86,
1065-1073.
13.Daurkin, I., Eruslanov, E., Stoffs, T., Perrin,
G. Q., Algood, C., Gilbert, S. M., Rosser, C. J., Su, L.-M., Vieweg,
J., and Kusmartsev, S. (2011) Tumor-associated macrophages mediate
immunosuppression in the renal cancer microenvironment by activating
the 15-lipoxygenase-2 pathway, Cancer Res., 71,
6400-6409.
14.Chen, P., Huang, Y., Bong, R., Ding, Y., Song,
N., Wang, X., Song, X., and Luo, Y. (2011) Tumor-associated macrophages
promote angiogenesis and melanoma growth via adrenomedullin in a
paracrine and autocrine manner, Clin. Cancer Res., 17,
7230-7239.
15.Liu, J., Zhang, N., Li, Q., Zhang, W., Ke, F.,
Leng, Q., Wang, H., Chen, J., and Wang, H. (2011) Tumor-associated
macrophages recruit CCR6+ regulatory T-cells and promote the
development of colorectal cancer via enhancing CCL20 production in
mice, PLoS One, 6, e19495.
16.Zijlmans, H. J., Fleuren, G. J., Baelde, H. J.,
Eilers, P. H. C., Kenter, G. G., and Gorter, A. (2006) The absence of
CCL2 expression in cervical carcinoma is associated with increased
survival and loss of heterozygosity at 17q11.2, J. Pathol.,
208, 507-517.
17.Koelzer, V. H., Canonica, K., Dawson, H., Sokol,
L., Karamitopoulou-Diamantis, E., Lugli, A., and Zlobec, I. (2016)
Phenotyping of tumor-associated macrophages in colorectal cancer:
impact on single cell invasion (tumor budding) and clinicopathological
outcome, Oncoimmunology, 5, e1106677.
18.Gerlini, G., Di Gennaro, P., and Borgognoni, L.
(2012) Enhancing anti-melanoma immunity by electrochemotherapy and
in vivo dendritic-cell activation, Oncoimmunology,
1, 1655-1657.
19.Michielsen, A. J., Noonan, S., Martin, P.,
Tosetto, M., Marry, J., Biniecka, M., Maguire, A. A., Hyland, J. M.,
Sheahan, K. D., O’Donoghue, D. P., Mulcahy, H. E., Fennelly, D.,
Ryan, E. J., and O’Sullivan, J. N. (2012) Inhibition of dendritic
cell maturation by the tumor microenvironment correlates with the
survival of colorectal cancer patients following bevacizumab treatment,
Mol. Cancer Ther., 11, 1829-1837.
20.Yu, H., Huang, X., Liu, X., Jin, H., Zhang, G.,
Zhang, Q., and Yu, J. (2013) Regulatory T-cells and plasmacytoid
dendritic cells contribute to the immune escape of papillary thyroid
cancer coexisting with multinodular non-toxic goiter, Endocrine,
44, 172-181.
21.Preynat-Seauve, O., Contassot, E., Schuler, P.,
French, L. E., and Huard, B. (2007) Melanoma-infiltrating dendritic
cells induce protective antitumor responses mediated by T cells,
Melanoma Res., 17, 169-176.
22.Scouten, W. T., and Francis, G. L. (2014) Thyroid
cancer and the immune system: a model for effective immune
surveillance, Exp. Rev. Endocrinol. Metab., 1,
353-366.
23.Perrot, I., Blanchard, D., Freymond, N., Isaac,
S., Guibert, B., Pacheco, Y., and Lebecque, S. (2007) Dendritic cells
infiltrating human non-small cell lung cancer are blocked at immature
stage, J. Immunol., 178, 2763-2769.
24.Marvel, D., and Gabrilovich, D. I. (2015)
Myeloid-derived suppressor cells in the tumor microenvironment: expect
the unexpected, J. Clin. Invest., 125, 3356-3364.
25.Eruslanov, E. B., Bhojnagarwala, P. S.,
Quatromoni, J. G., Stephen, T. L., Ranganathan, A., Deshpande, C.,
Akimova, T., Vachani, A., Litzky, L., Hancock, W. W., Conejo-Garcia, J.
R., Feldman, M., Albelda, S. M., and Singhal, S. (2014)
Tumor-associated neutrophils stimulate T-cell responses in early-stage
human lung cancer, J. Clin. Invest., 124, 5466-5480.
26.Solito, S., Marigo, I., Pinton, L., Damuzzo, V.,
Mandruzzato, S., and Bronte, V. (2014) Myeloid-derived suppressor cell
heterogeneity in human cancers, Ann. N. Y. Acad. Sci.,
1319, 47-65.
27.Senovilla, L., Vacchelli, E., Galon, J.,
Adjemian, S., Eggermont, A., Fridman, W. H., Sautes-Fridman, C., Ma,
Y., Tartour, E., Zitvogel, L., Kroemer, G., and Galluzzi, L. (2012)
Trial watch: prognostic and predictive value of the immune infiltrate
in cancer, Oncoimmunology, 1, 1323-1343.
28.Vesely, M. D., Kershaw, M. H., Schreiber, R. D.,
and Smyth, M. J. (2011) Natural innate and adaptive immunity to cancer,
Annu. Rev. Immunol., 29, 235-271.
29.Hamai, A., Benlalam, H., Meslin, F., Hasmim, M.,
Carre, T., Akalay, I., Janji, B., Berchem, G., Noman, M. Z., and
Chouaib, S. (2010) Immune surveillance of human cancer: if the
cytotoxic T-lymphocytes play the music, does the tumoral system call
the tune? Tissue Antigens, 75, 1-8.
30.Sakuishi, K., Apetoh, L., Sullivan, J. M.,
Blazar, B. R., Kuchroo, V. K., and Anderson, A. C. (2010) Targeting
Tim-3 and PD-1 pathways to reverse T-cell exhaustion and restore
anti-tumor immunity, J. Exp. Med., 207, 2187-2194.
31.Park, H. J., Kusnadi, A., Lee, E.-J., Kim, W. W.,
Cho, B. C., Lee, I. J., Seong, J., and Ha, S.-J. (2019)
Tumor-infiltrating regulatory T-cells delineated by upregulation of
PD-1 and inhibitory receptors, Cell. Immunol., 278,
76-83.
32.Wang, Y., Ma, Y., Fang, Y., Wu, S., Liu, L., Fu,
D., and Shen, X. (2012) Regulatory T-cell: a protection for tumor
cells, J. Cell. Mol. Med., 16, 425-436.
33.Fridman, W. H., Pages, F., Sautes-Fridman, C.,
and Galon, J. (2012) The immune contexture in human tumors: impact on
clinical outcome, Nat. Rev. Cancer, 12, 298-306.
34.Badoual, C., Hans, S., Merillon, N., Van Ryswick,
C., Ravel, P., Benhamouda, N., Levionnois, E., Nizard, M., Si-Mohamed,
A., Besnier, N., Gey, A., Rotem-Yehudar, R., Pere, H., Tran, T.,
Guerin, C. L., Chauvat, A., Dransart, E., Alanio, C., Albert, S.,
Barry, B., Sandoval, F., Quintin-Colonna, F., Bruneval, P., Fridman, W.
H., Lemoine, F. M., Oudard, S., Johannes, L., Olive, D., Brasnu, D.,
and Tartour, E. (2013) PD-1-expressing tumor-infiltrating T-cells are a
favorable prognostic biomarker in HPV-associated head and neck cancer,
Cancer Res., 73, 128-138.
35.Badoual, C., Hans, S., Fridman, W. H., Brasnu,
D., Erdman, S., and Tartour, E. (2009) Revisiting the prognostic value
of regulatory T-cells in patients with cancer, J. Clin. Oncol.,
27, e5-6.
36.Germain, C., Gnjatic, S., and Dieu-Nosjean, M.-C.
(2015) Tertiary lymphoid structure-associated B-cells are key players
in anti-tumor immunity, Front. Immunol., 6, 67.
37.De Visser, K. E., Korets, L. V., and Coussens, L.
M. (2005) De novo carcinogenesis promoted by chronic
inflammation is B-lymphocyte dependent, Cancer Cell, 7,
411-423.
38.DeNardo, D. G., Andreu, P., and Coussens, L. M.
(2010) Interactions between lymphocytes and myeloid cells regulate pro-
versus anti-tumor immunity, Cancer Metastasis Rev., 29,
309-316.
39.Olkhanud, P. B., Damdinsuren, B., Bodogai, M.,
Gress, R. E., Sen, R., Wejksza, K., Malchinkhuu, E., Wersto, R. P., and
Biragyn, A. (2011) Tumor-evoked regulatory B cells promote breast
cancer metastasis by converting resting CD4+ T-cells to
T-regulatory cells, Cancer Res., 71, 3505-3515.
40.Horikawa, M., Minard-Colin, V., Matsushita, T.,
and Tedder, T. F. (2011) Regulatory B-cell production of IL-10 inhibits
lymphoma depletion during CD20 immunotherapy in mice, J. Clin.
Invest., 121, 4268-4280.
41.Cipponi, A., Mercier, M., Seremet, T., Baurain,
J.-F., Theate, I., Van den Oord, J., Stas, M., Boon, T., Coulie, P. G.,
and Van Baren, N. (2012) Neogenesis of lymphoid structures and antibody
responses occur in human melanoma metastases, Cancer Res.,
72, 3997-4007.
42.Coughlin, C. M., Vance, B. A., Grupp, S. A., and
Vonderheide, R. H. (2004) RNA-transfected CD40-activated B-cells induce
functional T-cell responses against viral and tumor antigen targets:
implications for pediatric immunotherapy, Blood, 103,
2046-2054.
43.Ronchese, F., and Hausmann, B. (1993)
B-lymphocytes in vivo fail to prime naive T-cells but can
stimulate antigen-experienced T-lymphocytes, J. Exp. Med.,
177, 679-690.
44.Shi, J.-Y., Gao, Q., Wang, Z.-C., Zhou, J., Wang,
X.-Y., Min, Z.-H., Shi, Y.-H., Shi, G.-M., Ding, Z.-B., Ke, A.-W., Dai,
Z., Qiu, S. J., Song, K., and Fan, J. (2013) Margin-infiltrating
CD20+ B cells display an atypical memory phenotype and
correlate with favorable prognosis in hepatocellular carcinoma,
Clin. Cancer Res., 19, 5994-6005.
45.Dieu-Nosjean, M.-C., Antoine, M., Danel, C.,
Heudes, D., Wislez, M., Poulot, V., Rabbe, N., Laurans, L., Tartour,
E., De Chaisemartin, L., Lebecque, S., Fridman, W. H., and Cadranel, J.
(2008) Long-term survival for patients with non-small-cell lung cancer
with intratumoral lymphoid structures, J. Clin. Oncol.,
26, 4410-4417.
46.Germain, C., Gnjatic, S., Tamzalit, F.,
Knockaert, S., Remark, R., Goc, J., Lepelley, A., Becht, E., Katsahian,
S., Bizouard, G., Validire, P., Damotte, D., Alifano, M., Magdeleinat,
P., Cremer, I., Teillaud, J. L., Fridman, W. H., Sautes-Fridman, C.,
and Dieu-Nosjean, M. C. (2014) Presence of B-cells in tertiary lymphoid
structures is associated with a protective immunity in patients with
lung cancer, Am. J. Respir. Crit. Care Med., 189,
832-844.
47.Miyasaka, M., and Tanaka, T. (2004) Lymphocyte
trafficking across high endothelial venules: dogmas and enigmas,
Nat. Rev. Immunol., 4, 360-370.
48.Dieu-Nosjean, M.-C., Giraldo, N. A., Kaplon, H.,
Germain, C., Fridman, W. H., and Sautes-Fridman, C. (2016) Tertiary
lymphoid structures, drivers of the anti-tumor responses in human
cancers, Immunol. Rev., 271, 260-275.
49.Carrega, P., Loiacono, F., Di Carlo, E.,
Scaramuccia, A., Mora, M., Conte, R., Benelli, R., Spaggiari, G. M.,
Cantoni, C., Campana, S., Bonaccorsi, I., Morandi, B., Truini, M.,
Mingari, M. C., Moretta, L., and Ferlazzo, G. (2015) NCR+
ILC3 concentrate in human lung cancer and associate with intratumoral
lymphoid structures, Nat. Commun., 6, 8280.
50.Coca, S., Perez-Piqueras, J., Martinez, D.,
Colmenarejo, A., Saez, M. A., Vallejo, C., Martos, J. A., and Moreno,
M. (1997) The prognostic significance of intratumoral natural killer
cells in patients with colorectal carcinoma, Cancer, 79,
2320-2328.
51.Villegas, F. R., Coca, S., Villarrubia, V. G.,
Jimenez, R., Chillon, M. J., Jareno, J., Zuil, M., and Callol, L.
(2002) Prognostic significance of tumor infiltrating natural killer
cells subset CD57 in patients with squamous cell lung cancer, Lung
Cancer, 35, 23-28.
52.Clark, W. H., From, L., Bernardino, E. A., and
Mihm, M. C. (1969) The histogenesis and biologic behavior of primary
human malignant melanomas of the skin, Cancer Res., 29,
705-727.
53.Elder, D. E., Guerry, D., Vanhorn, M., Hurwitz,
S., Zehngebot, L., Goldman, L. I., Larossa, D., Hamilton, R., Bondi, E.
E., and Clark, W. H. (1985) The role of lymph node dissection for
clinical stage I malignant melanoma of intermediate thickness
(1.51-3.99 mm), Cancer, 56, 413-418.
54.Busam, K. J., Antonescu, C. R., Marghoob, A. A.,
Nehal, K. S., Sachs, D. L., Shia, J., and Berwick, M. (2001) Histologic
classification of tumor-infiltrating lymphocytes in primary cutaneous
malignant melanoma. A study of interobserver agreement, Am. J. Clin.
Pathol., 115, 856-860.
55.Schatton, T., Scolyer, R. A., Thompson, J. F.,
and Mihm, M. C. (2014) Tumor-infiltrating lymphocytes and their
significance in melanoma prognosis, Methods Mol. Biol.,
1102, 287-324.
56.Tuthill, R. J., Unger, J. M., Liu, P. Y.,
Flaherty, L. E., and Sondak, V. K. (2002) Risk assessment in localized
primary cutaneous melanoma: a Southwest Oncology Group study evaluating
nine factors and a test of the Clark logistic regression prediction
model, Am. J. Clin. Pathol., 118, 504-511.
57.Clemente, C. G., Mihm, M. C., Bufalino, R.,
Zurrida, S., Collini, P., and Cascinelli, N. (1996) Prognostic value of
tumor infiltrating lymphocytes in the vertical growth phase of primary
cutaneous melanoma, Cancer, 77, 1303-1310.
58.Mihm, M. C., Clemente, C. G., and Cascinelli, N.
(1996) Tumor infiltrating lymphocytes in lymph node melanoma
metastases: a histopathologic prognostic indicator and an expression of
local immune response, Lab. Invest., 74, 43-47.
59.Cohen, P. J., Lotze, M. T., Roberts, J. R.,
Rosenberg, S. A., and Jaffe, E. S. (1987) The immunopathology of
sequential tumor biopsies in patients treated with interleukin-2.
Correlation of response with T-cell infiltration and HLA-DR expression,
Am. J. Pathol., 129, 208-216.
60.Romero, P., Valmori, D., Pittet, M. J.,
Zippelius, A., Rimoldi, D., Levy, F., Dutoit, V., Ayyoub, M.,
Rubio-Godoy, V., Michielin, O., Guillaume, P., Batard, P., Luescher, I.
F., Lejeune, F., Lienard, D., Rufer, N., Dietrich, P. Y., Speiser, D.
E., and Cerottini, J. C. (2002) Antigenicity and immunogenicity of
Melan-A/MART-1 derived peptides as targets for tumor reactive CTL in
human melanoma, Immunol. Rev., 188, 81-96.
61.Kawakami, Y., Robbins, P. F., Wang, R. F.,
Parkhurst, M., Kang, X., and Rosenberg, S. A. (1998) The use of
melanosomal proteins in the immunotherapy of melanoma, J.
Immunother., 21, 237-246.
62.Kawakami, Y. (2000) New cancer therapy by
immunomanipulation: development of immunotherapy for human melanoma as
a model system, Cornea, 19, S2-6.
63.Chauvin, J.-M., Pagliano, O., Fourcade, J., Sun,
Z., Wang, H., Sander, C., Kirkwood, J. M., Chen, T. T., Maurer, M.,
Korman, A. J., and Zarour, H. M. (2015) TIGIT and PD-1 impair tumor
antigen-specific CD8+ T-cells in melanoma patients, J.
Clin. Invest., 125, 2046-2058.
64.Harlin, H., Kuna, T. V., Peterson, A. C., Meng,
Y., and Gajewski, T. F. (2006) Tumor progression despite massive influx
of activated CD8+ T-cells in a patient with malignant
melanoma ascites, Cancer Immunol. Immunother., 55,
1185-1197.
65.Boon, T., Coulie, P. G., Van den Eynde, B. J.,
and Van der Bruggen, P. (2006) Human T-cell responses against melanoma,
Annu. Rev. Immunol., 24, 175-208.
66.Sharma, M. D., Hou, D.-Y., Liu, Y., Koni, P. A.,
Metz, R., Chandler, P., Mellor, A. L., He, Y., and Munn, D. H. (2009)
Indoleamine 2,3-dioxygenase controls conversion of Foxp3+
Tregs to TH17-like cells in tumor-draining lymph nodes, Blood,
113, 6102-6111.
67.Vukmanovic-Stejic, M., Zhang, Y., Cook, J. E.,
Fletcher, J. M., McQuaid, A., Masters, J. E., Rustin, M. H. A., Taams,
L. S., Beverley, P. C. L., Macallan, D. C., and Akbar, A. N. (2006)
Human CD4+ CD25hi Foxp3+ regulatory T cells are
derived by rapid turnover of memory populations in vivo, J.
Clin. Invest., 116, 2423-2433.
68.Lowes, M. A., Alex Bishop, G., Crotty, K.,
Barnetson, R. S. C., and Halliday, G. M. (1997) T-helper 1 cytokine
mRNA is increased in spontaneously regressing primary melanomas, J.
Invest. Dermatol., 108, 914-919.
69.Conrad, C. T., Ernst, N. R., Dummer, W., Brocker,
E. B., and Becker, J. C. (1999) Differential expression of transforming
growth factor beta 1 and interleukin 10 in progressing and regressing
areas of primary melanoma, J. Exp. Clin. Cancer Res., 18,
225-232.
70.Somasundaram, R., Robbins, P., Moonka, D., Loh,
E., Marincola, F., Patel, A., Guerry, D., and Herlyn, D. (2000)
CD4+, HLA class I-restricted, cytolytic T-lymphocyte clone
against primary malignant melanoma cells, Int. J. Cancer,
85, 253-259.
71.Dudley, M. E., Wunderlich, J. R., Robbins, P. F.,
Yang, J. C., Hwu, P., Schwartzentruber, D. J., Topalian, S. L., Sherry,
R., Restifo, N. P., Hubicki, A. M., Robinson, M. R., Raffeld, M.,
Duray, P., Seipp, C. A., Rogers-Freezer, L., Morton, K. E.,
Mavroukakis, S. A., White, D. E., and Rosenberg, S. A. (2002) Cancer
regression and autoimmunity in patients after clonal repopulation with
antitumor lymphocytes, Science, 298, 850-854.
72.Hunder, N. N., Wallen, H., Cao, J., Hendricks, D.
W., Reilly, J. Z., Rodmyre, R., Jungbluth, A., Gnjatic, S., Thompson,
J. A., and Yee, C. (2008) Treatment of metastatic melanoma with
autologous CD4+ T-cells against NY-ESO-1, N. Engl. J.
Med., 358, 2698-2703.
73.Ladanyi, A., Kiss, J., Mohos, A., Somlai, B.,
Liszkay, G., Gilde, K., Fejos, Z., Gaudi, I., Dobos, J., and Timar, J.
(2011) Prognostic impact of B-cell density in cutaneous melanoma,
Cancer Immunol. Immunother., 60, 1729-1738.
74.Jager, E., Nagata, Y., Gnjatic, S., Wada, H.,
Stockert, E., Karbach, J., Dunbar, P. R., Lee, S. Y., Jungbluth, A.,
Jager, D., Arand, M., Ritter, G., Cerundolo, V., Dupont, B., Chen,
Y.-T., Old, L. J., and Knuth, A. (2000) Monitoring CD8 T-cell responses
to NY-ESO-1: correlation of humoral and cellular immune responses,
Proc. Natl. Acad. Sci. USA, 97, 4760-4765.
75.Ferlay, J., Shin, H.-R., Bray, F., Forman, D.,
Mathers, C., and Parkin, D. M. (2010) Estimates of worldwide burden of
cancer in 2008: GLOBOCAN 2008, Int. J. Cancer, 127,
2893-2917.
76.Loi, S., Michiels, S., Salgado, R., Sirtaine, N.,
Jose, V., Fumagalli, D., Kellokumpu-Lehtinen, P.-L., Bono, P., Kataja,
V., Desmedt, C., Piccart, M. J., Loibl, S., Denkert, C., Smyth, M. J.,
Joensuu, H., and Sotiriou, C. (2014) Tumor infiltrating lymphocytes are
prognostic in triple negative breast cancer and predictive for
trastuzumab benefit in early breast cancer: results from the FinHER
trial, Ann. Oncol., 25, 1544-1550.
77.Adams, S., Gray, R. J., Demaria, S., Goldstein,
L., Perez, E. A., Shulman, L. N., Martino, S., Wang, M., Jones, V. E.,
Saphner, T. J., Wolff, A. C., Wood, W. C., Davidson, N. E., Sledge, G.
W., Sparano, J. A., and Badve, S. S. (2014) Prognostic value of
tumor-infiltrating lymphocytes in triple-negative breast cancers from
two phase III randomized adjuvant breast cancer trials: ECOG 2197 and
ECOG 1199, J. Clin. Oncol., 32, 2959-2966.
78.Loi, S., Sirtaine, N., Piette, F., Salgado, R.,
Viale, G., Van Eenoo, F., Rouas, G., Francis, P., Crown, J. P. A.,
Hitre, E., De Azambuja, E., Quinaux, E., Di Leo, A., Michiels, S.,
Piccart, M. J., and Sotiriou, C. (2013) Prognostic and predictive value
of tumor-infiltrating lymphocytes in a phase III randomized adjuvant
breast cancer trial in node-positive breast cancer comparing the
addition of docetaxel to doxorubicin with doxorubicin-based
chemotherapy: BIG 02-98, J. Clin. Oncol., 31,
860-867.
79.Loi, S., Michiels, S., Salgado, R., Sirtaine, N.,
Jose, V., Fumagalli, D., Brown, D., Kellokumpu-Lehtinen, P.-L., Bono,
P., Kataja, V., Desmedt, C., Piccart-Gebhart, M. J., Loibl, S., Untch,
M., Denkert, C., Smyth, M. J., Joensuu, H., and Sotiriou, C. (2013)
Abstract S1-05: tumor infiltrating lymphocytes (TILs) indicate
trastuzumab benefit in early-stage HER2-positive breast cancer
(HER2+BC), Cancer Res., 73, S1-05.
80.Seo, A. N., Lee, H. J., Kim, E. J., Kim, H. J.,
Jang, M. H., Lee, H. E., Kim, Y. J., Kim, J. H., and Park, S. Y. (2013)
Tumor-infiltrating CD8+ lymphocytes as an independent
predictive factor for pathological complete response to primary
systemic therapy in breast cancer, Br. J. Cancer, 109,
2705-2713.
81.Brown, J. R., Wimberly, H., Lannin, D. R., Nixon,
C., Rimm, D. L., and Bossuyt, V. (2014) Multiplexed quantitative
analysis of CD3, CD8, and CD20 predicts response to neoadjuvant
chemotherapy in breast cancer, Clin. Cancer Res., 20,
5995-6005.
82.Lee, H. J., Seo, J.-Y., Ahn, J.-H., Ahn, S.-H.,
and Gong, G. (2013) Tumor-associated lymphocytes predict response to
neoadjuvant chemotherapy in breast cancer patients, J. Breast
Cancer, 16, 32-39.
83.Ma, C., Zhang, Q., Ye, J., Wang, F., Zhang, Y.,
Wevers, E., Schwartz, T., Hunborg, P., Varvares, M. A., Hoft, D. F.,
Hsueh, E. C., and Peng, G. (2012) Tumor-infiltrating γδ T
lymphocytes predict clinical outcome in human breast cancer, J.
Immunol., 189, 5029-5036.
84.Gu-Trantien, C., Loi, S., Garaud, S., Equeter,
C., Libin, M., De Wind, A., Ravoet, M., Le Buanec, H., Sibille, C.,
Manfouo-Foutsop, G., Veys, I., Haibe-Kains, B., Singhal, S. K.,
Michiels, S., Rothe, F., Saldago, R., Duvillier, H., Ignatiadis, M.,
Desmedt, C., Bron, D., Larsimont, D., Piccart, M., Sotiriou, C., and
Willard-Gallo, K. (2013) CD4+ follicular helper T-cell infiltration
predicts breast cancer survival, J. Clin. Invest., 123, 2873-2892.
85.Gjerstorff, M. F., Benoit, V. M., Laenkholm,
A.-V., Nielsen, O., Johansen, L. E., and Ditzel, H. J. (2006)
Identification of genes with altered expression in medullary breast
cancer vs. ductal breast cancer and normal breast epithelia, Int. J.
Oncol., 28, 1327-1335.
86.Pedersen, L., Zedeler, K., Holck, S., Schiodt,
T., and Mouridsen, H. T. (1995) Medullary carcinoma of the breast.
Prevalence and prognostic importance of classical risk factors in
breast cancer, Eur. J. Cancer, 31A, 2289-2295.
87.Yakirevich, E., Maroun, L., Cohen, O., Izhak, O.
B., Rennert, G., and Resnick, M. B. (2000) Apoptosis, proliferation,
and Fas (APO-1, CD95)/Fas ligand expression in medullary carcinoma of
the breast, J. Pathol., 192, 166-173.
88.Jensen, V., Jensen, M. L., Kiaer, H., Andersen,
J., and Melsen, F. (1997) MIB-1 expression in breast carcinomas with
medullary features. An immunohistological study including correlations
with p53 and bcl-2, Virchow’s Arch., 431,
125-130.
89.Bacus, S. S., Zelnick, C. R., Chin, D. M.,
Yarden, Y., Kaminsky, D. B., Bennington, J., Wen, D., Marcus, J. N.,
and Page, D. L. (1994) Medullary carcinoma is associated with
expression of intercellular adhesion molecule-1. Implication to its
morphology and its clinical behavior, Am. J. Pathol.,
145, 1337-1348.
90.Ridolfi, R. L., Rosen, P. P., Port, A., Kinne,
D., and Mike, V. (1977) Medullary carcinoma of the breast. A
clinicopathologic study with 10-year follow-up, Cancer,
40, 1365-1385.
91.Hansen, M. H., Nielsen, H. V., and Ditzel, H. J.
(2002) Translocation of an intracellular antigen to the surface of
medullary breast cancer cells early in apoptosis allows for an
antigen-driven antibody response elicited by tumor-infiltrating
B-cells, J. Immunol., 169, 2701-2711.
92.Kotlan, B., Simsa, P., Teillaud, J.-L., Fridman,
W. H., Toth, J., McKnight, M., and Glassy, M. C. (2005) Novel
ganglioside antigen identified by B cells in human medullary breast
carcinomas: the proof of principle concerning the tumor-infiltrating
B-lymphocytes, J. Immunol., 175, 2278-2285.
93.Hansen, M. H., Nielsen, H., and Ditzel, H. J.
(2001) The tumor-infiltrating B cell response in medullary breast
cancer is oligoclonal and directed against the autoantigen actin
exposed on the surface of apoptotic cancer cells, Proc. Natl. Acad.
Sci. USA, 98, 12659-12664.
94.Matsubayashi, S., Kawai, K., Matsumoto, Y.,
Mukuta, T., Morita, T., Hirai, K., Matsuzuka, F., Kakudoh, K., Kuma,
K., and Tamai, H. (1995) The correlation between papillary thyroid
carcinoma and lymphocytic infiltration in the thyroid gland, J.
Clin. Endocrinol. Metab., 80, 3421-3424.
95.Gupta, S., Patel, A., Folstad, A., Fenton, C.,
Dinauer, C. A., Tuttle, R. M., Conran, R., and Francis, G. L. (2001)
Infiltration of differentiated thyroid carcinoma by proliferating
lymphocytes is associated with improved disease-free survival for
children and young adults, J. Clin. Endocrinol. Metab.,
86, 1346-1354.
96.Fiumara, A., Belfiore, A., Russo, G., Salomone,
E., Santonocito, G. M., Ippolito, O., Vigneri, R., and Gangemi, P.
(1997) In situ evidence of neoplastic cell phagocytosis by
macrophages in papillary thyroid cancer, J. Clin. Endocrinol.
Metab., 82, 1615-1620.
97.Bagnasco, M., Venuti, D., Paolieri, F., Torre,
G., Ferrini, S., and Canonica, G. W. (1989) Phenotypic and functional
analysis at the clonal level of infiltrating T-lymphocytes in papillary
carcinoma of the thyroid: prevalence of cytolytic T-cells with natural
killer-like or lymphokine-activated killer activity, J. Clin.
Endocrinol. Metab., 69, 832-836.
98.Salmaso, C., Olive, D., Pesce, G., and Bagnasco,
M. (2002) Costimulatory molecules and autoimmune thyroid diseases,
Autoimmunity, 35, 159-167.
99.Shah, R., Banks, K., Patel, A., Dogra, S.,
Terrell, R., Powers, P. A., Fenton, C., Dinauer, C. A., Tuttle, R. M.,
and Francis, G. L. (2002) Intense expression of the b7-2 antigen
presentation coactivator is an unfavorable prognostic indicator for
differentiated thyroid carcinoma of children and adolescents, J.
Clin. Endocrinol. Metab., 87, 4391-4397.
100.Wilders-Truschnig, M. M., Kabel, P. J.,
Drexhage, H. A., Beham, A., Leb, G., Eber, O., Hebenstreit, J.,
Loidolt, D., Dohr, G., and Lanzer, G. (1989) Intrathyroidal dendritic
cells, epitheloid cells, and giant cells in iodine deficient goiter,
Am. J. Pathol., 135, 219-225.
101.Pinzon-Charry, A., Maxwell, T., and Lopez, J.
A. (2005) Dendritic cell dysfunction in cancer: a mechanism for
immunosuppression, Immunol. Cell Biol., 83, 451-461.
102.Kusmartsev, S., Nefedova, Y., Yoder, D., and
Gabrilovich, D. I. (2004) Antigen-specific inhibition of
CD8+ T-cell response by immature myeloid cells in cancer is
mediated by reactive oxygen species, J. Immunol., 172,
989-999.
103.Dailey, M. E. (1955) Relation of thyroid
neoplasms to hashimoto disease of the thyroid gland, Arch.
Surg., 70, 291.
104.Kashima, K., Yokoyama, S., Noguchi, S.,
Murakami, N., Yamashita, H., Watanabe, S., Uchino, S., Toda, M.,
Sasaki, A., Daa, T., and Nakayama, I. (1998) Chronic thyroiditis as a
favorable prognostic factor in papillary thyroid carcinoma,
Thyroid, 8, 197-202.
105.Souza, S. L., Montalli Da Assumpcao, L. V., and
Ward, L. S. (2003) Impact of previous thyroid autoimmune diseases on
prognosis of patients with well-differentiated thyroid cancer,
Thyroid, 13, 491-495.
106.Loh, K. C., Greenspan, F. S., Dong, F., Miller,
T. R., and Yeo, P. P. (1999) Influence of lymphocytic thyroiditis on
the prognostic outcome of patients with papillary thyroid carcinoma,
J. Clin. Endocrinol. Metab., 84, 458-463.
107.Okayasu, I., Fujiwara, M., Hara, Y., Tanaka,
Y., and Rose, N. R. (1995) Association of chronic lymphocytic
thyroiditis and thyroid papillary carcinoma. A study of surgical cases
among Japanese, and white and African Americans, Cancer,
76, 2312-2318.
108.Arif, S., Blanes, A., and Diaz-Cano, S. J.
(2002) Hashimoto’s thyroiditis shares features with early
papillary thyroid carcinoma, Histopathology, 41,
357-362.
109.Mechler, C., Bounacer, A., Suarez, H., Saint
Frison, M., Magois, C., Aillet, G., and Gaulier, A. (2001) Papillary
thyroid carcinoma: 6 cases from 2 families with associated lymphocytic
thyroiditis harbouring RET/PTC rearrangements, Br. J. Cancer,
85, 1831-1837.
110.Apel, R. L., Asa, S. L., and LiVolsi, V. A.
(1995) Papillary Hurthle cell carcinoma with lymphocytic stroma.
“Warthin-like tumor” of the thyroid, Am. J. Surg.
Pathol., 19, 810-814.
111.Dolcetti, R., Viel, A., Doglioni, C., Russo,
A., Guidoboni, M., Capozzi, E., Vecchiato, N., Macri, E., Fornasarig,
M., and Boiocchi, M. (1999) High prevalence of activated
intraepithelial cytotoxic T-lymphocytes and increased neoplastic cell
apoptosis in colorectal carcinomas with microsatellite instability,
Am. J. Pathol., 154, 1805-1813.
112.Kim, H., Jen, J., Vogelstein, B., and Hamilton,
S. R. (1994) Clinical and pathological characteristics of sporadic
colorectal carcinomas with DNA replication errors in microsatellite
sequences, Am. J. Pathol., 145, 148-156.
113.Risio, M., Reato, G., Di Celle, P. F.,
Fizzotti, M., Rossini, F. P., and Foa, R. (1996) Microsatellite
instability is associated with the histological features of the tumor
in nonfamilial colorectal cancer, Cancer Res., 56,
5470-5474.
114.Phillips, S. M., Banerjea, A., Feakins, R., Li,
S. R., Bustin, S. A., and Dorudi, S. (2004) Tumour-infiltrating
lymphocytes in colorectal cancer with microsatellite instability are
activated and cytotoxic, Br. J. Surg., 91, 469-475.
115.Linnebacher, M., Gebert, J., Rudy, W., Woerner,
S., Yuan, Y. P., Bork, P., and Von Knebel Doeberitz, M. (2001)
Frameshift peptide-derived T-cell epitopes: a source of novel
tumor-specific antigens, Int. J. Cancer, 93, 6-11.
116.Linnebacher, M., Wienck, A., Boeck, I., and
Klar, E. (2010) Identification of an MSI-H tumor-specific cytotoxic
T-cell epitope generated by the (–1) frame of U79260(FTO),
J. Biomed. Biotechnol., 2010, 841451.
117.Schwitalle, Y., Kloor, M., Eiermann, S.,
Linnebacher, M., Kienle, P., Knaebel, H. P., Tariverdian, M., Benner,
A., and Von Knebel Doeberitz, M. (2008) Immune response against
frameshift-induced neopeptides in HNPCC patients and healthy HNPCC
mutation carriers, Gastroenterology, 134, 988-997.
118.Bauer, K., Nelius, N., Reuschenbach, M., Koch,
M., Weitz, J., Steinert, G., Kopitz, J., Beckhove, P., Tariverdian, M.,
Von Knebel Doeberitz, M., and Kloor, M. (2013) T-cell responses against
microsatellite instability-induced frameshift peptides and influence of
regulatory T-cells in colorectal cancer, Cancer Immunol.
Immunother., 62, 27-37.
119.Ishikawa, T., Fujita, T., Suzuki, Y., Okabe,
S., Yuasa, Y., Iwai, T., and Kawakami, Y. (2003) Tumor-specific
immunological recognition of frameshift-mutated peptides in colon
cancer with microsatellite instability, Cancer Res., 63,
5564-5572.
120.Reuschenbach, M., Kloor, M., Morak, M.,
Wentzensen, N., Germann, A., Garbe, Y., Tariverdian, M., Findeisen, P.,
Neumaier, M., Holinski-Feder, E., and Von Knebel Doeberitz, M. (2010)
Serum antibodies against frameshift peptides in microsatellite unstable
colorectal cancer patients with Lynch syndrome, Fam. Cancer,
9, 173-179.
121.Llosa, N. J., Cruise, M., Tam, A., Wicks, E.
C., Hechenbleikner, E. M., Taube, J. M., Blosser, R. L., Fan, H., Wang,
H., Luber, B. S., Zhang, M., Papadopoulos, N., Kinzler, K. W.,
Vogelstein, B., Sears, C. L., Anders, R. A., Pardoll, D. M., and
Housseau, F. (2015) The vigorous immune microenvironment of
microsatellite instable colon cancer is balanced by multiple
counter-inhibitory checkpoints, Cancer Discov., 5,
43-51.
122.Xiao, Y., and Freeman, G. J. (2015) The
microsatellite instable subset of colorectal cancer is a particularly
good candidate for checkpoint blockade immunotherapy, Cancer
Discov., 5, 16-18.
123.Kroemer, G., Galluzzi, L., Zitvogel, L., and
Fridman, W. H. (2015) Colorectal cancer: the first neoplasia found to
be under immunosurveillance and the last one to respond to
immunotherapy? Oncoimmunology, 4, e1058597.
124.Le, D. T., Uram, J. N., Wang, H., Bartlett, B.
R., Kemberling, H., Eyring, A. D., Skora, A. D., Luber, B. S., Azad, N.
S., Laheru, D., Biedrzycki, B., Donehower, R. C., Zaheer, A., Fisher,
G. A., Crocenzi, T. S., Lee, J. J., Duffy, S. M., Goldberg, R. M., de
la Chapelle, A., Koshiji, M., Bhaijee, F., Huebner, T., Hruban, R. H.,
Wood, L. D., Cuka, N., Pardoll, D. M., Papadopoulos, N., Kinzler, K.
W., Zhou, S., Cornish, T. C., Taube, J. M., Anders, R. A., Eshleman, J.
R., Vogelstein, B., and Diaz, L. A., Jr. (2015) PD-1 blockade in tumors
with mismatch-repair deficiency, N. Engl. J. Med., 372,
2509-2520.
125.Galon, J., Costes, A., Sanchez-Cabo, F.,
Kirilovsky, A., Mlecnik, B., Lagorce-Pages, C., Tosolini, M., Camus,
M., Berger, A., Wind, P., Zinzindohoue, F., Bruneval, P., Cugnenc, P.
H., Trajanoski, Z., Fridman, W. H., and Pages, F. (2006) Type, density,
and location of immune cells within human colorectal tumors predict
clinical outcome, Science, 313, 1960-1964.
126.Mlecnik, B., Tosolini, M., Kirilovsky, A.,
Berger, A., Bindea, G., Meatchi, T., Bruneval, P., Trajanoski, Z.,
Fridman, W.-H., Pages, F., and Galon, J. (2011) Histopathologic-based
prognostic factors of colorectal cancers are associated with the state
of the local immune reaction, J. Clin. Oncol., 29,
610-618.
127.Pages, F., Kirilovsky, A., Mlecnik, B.,
Asslaber, M., Tosolini, M., Bindea, G., Lagorce, C., Wind, P., Marliot,
F., Bruneval, P., Zatloukal, K., Trajanoski, Z., Berger, A., Fridman,
W. H., and Galon, J. (2009) In situ cytotoxic and memory T-cells
predict outcome in patients with early-stage colorectal cancer, J.
Clin. Oncol., 27, 5944-5951.
128.Anitei, M.-G., Zeitoun, G., Mlecnik, B.,
Marliot, F., Haicheur, N., Todosi, A.-M., Kirilovsky, A., Lagorce, C.,
Bindea, G., Ferariu, D., Danciu, M., Bruneval, P., Scripcariu, V.,
Chevallier, J. M., Zinzindohoue, F., Berger, A., Galon, J., and Pages,
F. (2014) Prognostic and predictive values of the immunoscore in
patients with rectal cancer, Clin. Cancer Res., 20,
1891-1899.
129.Galon, J., Pages, F., Marincola, F. M., Angell,
H. K., Thurin, M., Lugli, A., Zlobec, I., Berger, A., Bifulco, C.,
Botti, G., Tatangelo, F., Britten, C. M., Kreiter, S., Chouchane, L.,
Delrio, P., Arndt, H., Asslaber, M., Maio, M., Masucci, G. V., Mihm,
M., Vidal-Vanaclocha, F., Allison, J. P., Gnjatic, S., Hakansson, L.,
Huber, C., Singh-Jasuja, H., Ottensmeier, C., Zwierzina, H., Laghi, L.,
Grizzi, F., Ohashi, P. S., Shaw, P. A., Clarke, B. A., Wouters, B. G.,
Kawakami, Y., Hazama, S., Okuno, K., Wang, E., O’Donnell-Tormey,
J., Lagorce, C., Pawelec, G., Nishimura, M. I., Hawkins, R., Lapointe,
R., Lundqvist, A., Khleif, S. N., Ogino, S., Gibbs, P., Waring, P.,
Sato, N., Torigoe, T., Itoh, K., Patel, P. S., Shukla, S. N.,
Palmqvist, R., Nagtegaal, I. D., Wang, Y., D’Arrigo, C., Kopetz,
S., Sinicrope, F. A., Trinchieri, G., Gajewski, T. F., Ascierto, P. A.,
and Fox, B. A. (2012) Cancer classification using the Immunoscore: a
worldwide task force, J. Transl. Med., 10, 205.
130.Mlecnik, B., Bindea, G., Angell, H. K., Maby,
P., Angelova, M., Tougeron, D., Church, S. E., Lafontaine, L., Fischer,
M., Fredriksen, T., Sasso, M., Bilocq, A. M., Kirilovsky, A., Obenauf,
A. C., Hamieh, M., Berger, A., Bruneval, P., Tuech, J. J., Sabourin, J.
C., Le Pessot, F., Mauillon, J., Rafii, A., Laurent-Puig, P., Speicher,
M. R., Trajanoski, Z., Michel, P., Sesboue, R., Frebourg, T., Pages,
F., Valge-Archer, V., Latouche, J. B., and Galon, J. (2016) Integrative
analyses of colorectal cancer show immunoscore is a stronger predictor
of patient survival than microsatellite instability, Immunity,
44, 698-711.