“Suppressor Factor” of Neutrophils: A Short Story of a Long-Term Misconception
I. A. Linge1*, E. V. Kondratieva1, T. K. Kondratieva1, V. A. Makarov2, V. I. Polshakov3, O. Yu. Savelyev3, and A. S. Apt1*
1Central Research Institute for Tuberculosis, 107564 Moscow, Russia; E-mail: iralinge@gmail.com, asapt@aha.ru2Sechenov First Moscow State Medical University, Institute of Molecular Medicine, 119991 Moscow, Russia
3Lomonosov Moscow State University, Faculty of Fundamental Medicine, Center for Magnetic Tomography and Spectroscopy, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received May 19, 2016; Revision received August 4, 2016
A large body of evidence obtained during the last decade has demonstrated that neutrophils suppress T cell proliferation in different models of inflammation and cell interaction. The commonly used method for assessing cell proliferation and proliferation inhibition is measuring [3H]thymidine incorporation into cells. Earlier, we observed inhibition of [3H]thymidine uptake in experiments on neutrophil-mediated regulation of T cell response in tuberculosis immunity. Here, we used different types of proliferating cells to analyze the nature of the soluble “neutrophil factor” by a variety of methods (dialysis, HPLC, mass spectrometry, and NMR) and unambiguously demonstrated that neutrophils do not synthesize a specific factor inhibiting cell proliferation, but secrete high concentrations of extracellular thymidine that competitively inhibit [3H]thymidine incorporation. Although the physiological significance of thymidine secretion by neutrophils remains unknown, this phenomenon should be carefully considered when designing test systems for studying cell–cell interactions.
KEY WORDS: neutrophils, thymidine, proliferation inhibitionDOI: 10.1134/S0006297916110067
Abbreviations: APCs, antigen-presenting cells; CFSE, carboxyfluorescein succinimidyl ester; 13C-1H-HSQC, heteronuclear single quantum correlation spectroscopy; DQF-COSY, double-quantum filtered correlation spectroscopy; MHC, major histocompatibility complex; PBS, phosphate buffered saline.
Until recently, neutrophils were not viewed as participants in the
complex interactions between elements of innate and adaptive immunity.
This was due to their very short lifetime, quick replenishment of the
cell pool from the bone marrow, and ability to nonspecifically engulf
and destroy all types of bacteria without noticeable interactions with
other immune cells. Neutrophils were believed to primarily play an
important role in antibacterial defense, which sometimes can be unsafe
because of the damaging effect on host tissue. The role of neutrophils
in the intricate processes of immune response during chronic
inflammation has not been considered important [1-3]. The lack of data required
for detailed interpretation of neutrophil physiology has been mostly
related to technical difficulties of the experiments involving these
short-lived cells. Nevertheless, even this oversimplified view on the
nature of neutrophils has drastically changed during the last
decade.
The understanding of the role of neutrophils in immune response was greatly changed by two groups of publications. The first group of published data suggested an active role of neutrophils in long-term inflammatory processes such as chronic obstructive pulmonary disease (COPD) [4], lung tuberculosis [5, 6], arthritis [7], and Crohn disease [8]. The second, and probably more important pool of recent publications, directly demonstrated that neutrophils are involved in interactions with virtually all types of immune cells. In both mice and humans, neutrophils regulate the inflow of mononuclear cells into inflammation loci [9, 10] as well as the development and functioning of natural killer (NK) cells [11]. We showed that mouse B lymphocytes suppress migration of neutrophils to the site of Bacillus Calmette–Guérin (BCG) vaccine injection required for development of antituberculosis immunity [12]. Later, it was shown that IL-17A is also involved in this regulatory interaction [13]. In a noninfectious experimental system, neutrophils, on the contrary, inhibited B cell response to the protein antigen [14]. The effect of neutrophils on the immune response in experimental chronic infection caused by infecting mice with Brucella abortus was even broader and included regulation of B and T lymphocyte activation, infiltration of monocytes and dendritic cells, and a balance between types 1 and 2 immune response [15].
Numerous types of interactions between neutrophils and T lymphocytes are described in the review by Muller et al. [16] that discusses, in particular, whether neutrophils can act as antigen-presenting cells (APCs) for CD4+ T lymphocytes by implicating class II MHC molecules after acquiring them from “professional” APCs. In addition, an interesting study by D’Avila et al. [17] showed that after engulfment of BCG bacteria by neutrophils and subsequent phagocytosis of neutrophils by macrophages, the latter secrete PGE2 and TGF-β, both compounds being strong suppressors of T lymphocyte activation. These issues are closely related to our studies on the role of neutrophils in immune response in tuberculosis [5, 12, 18]. In this work, we investigated the nature of the proliferation-suppressing factor excreted by neutrophils. The results of this study are quite surprising.
MATERIALS AND METHODS
Animals. CBA mice (2-4-month-old) of both sexes were used in the study. The animals were kept under standard conditions with ad libitum access to food and water at the Animal Facility of the Central Research Institute for Tuberculosis.
Peritoneal macrophages, neutrophils, and cell supernatants were obtained as described previously [19]. To isolate peritoneal macrophages and neutrophils, mice were injected intraperitoneally with 2 ml of 3% peptone (Sigma, USA) 5 days and 16-18 h, respectively, prior to the experiment. Peritoneal exudate cells were obtained by rinsing the peritoneal cavity with 5 ml of Hanks’ balanced salt solution (Institute for Poliomyelitis and Viral Encephalitides, Russia) containing 2% fetal calf serum (FCS) and 10 U/ml heparin (Moscow Endocrine Plant, Russia). To remove heparin, the cells were then centrifuged and resuspended in complete RPMI 1640 medium (HyClone, USA) supplemented with 5% FCS, 5 mM HEPES, and 2 mM glutamine (further referred to as complete medium unless indicated otherwise). Macrophages were separated from other types of cells by adhesion to cell culture dishes in RPMI 1640 containing 10% FCS in a CO2 incubator for 60 min. The unabsorbed cells were washed off the monolayer with Versene solution. To obtain neutrophils, macrophages were removed from the peritoneal exudate by adsorption on the cell culture dish. Unabsorbed neutrophils were collected and used in experiments. The viability of cells in the suspension was estimated by Trypan Blue staining (HyClone) and comprised over 95%.
To obtain cell supernatants, isolated macrophages and/or neutrophils were incubated in 24-well plates (8·106 cells/ml per well) for 6, 24, or 48 h in complete RPMI 1640 medium containing 5% FCS, 5 mM HEPES, 2 mM glutamine, 1 mM pyruvate, 1% nonessential amino acids, 50 µM 2-mercaptoethanol, 100 µg/ml streptomycin, and 100 U/ml penicillin at 37°C in 5% CO2.
Naive lymphocytes and specific T cell lines. Naive lymphocytes were isolated from popliteal, axillary, and mesenteric lymph nodes of intact animals by the homogenization method. The cells were washed by centrifugation, transferred into complete medium, and used in the experiments on nonspecific activation of lymphocyte proliferation.
Mycobacterial antigen-specific T cell lines were obtained as described in [20]. Immune cells from popliteal lymph nodes of CBA mice were obtained 14 days after subcutaneous footpad injection of M. tuberculosis H37Rv sonicate (20 µg/mouse) in incomplete Freund’s adjuvant. The immune cells were washed with the medium, transferred to complete medium (2·106 cells/ml), and incubated in the presence of sonicated M. tuberculosis H37Rv bacteria (10 µg/ml) in a 24-well flat-bottom plate for 10-14 days at 37°C in 5% CO2. Transition of cells from proliferation to resting phase (usually, on days 10-12) was monitored visually from the cell shape using an inverted microscope. In the next stimulation cycle, the cells (2·105 cells/ml) were cultured for 9-11 days in the presence of spleen APCs (1.5·106 cells/ml) treated with mitomycin C (Kyowa, Japan) and mycobacterial sonicate (10 µg/ml). The stimulation/resting cycles were repeated until the cells either died or formed stable T cell line.
Inducers of T lymphocyte proliferation. Mycobacterial sonicate (10 µg/ml) was used for proliferation induction in specific T cell lines (2·104 cells/well); 2.5 µg/ml concanavalin A (Sigma-Aldrich, USA) or anti-CD3 antibodies (5 µg/ml, preabsorbed overnight at 4°C; R&D Systems, USA) were used for naive lymphocytes from lymph nodes (2.5·105 cells/well). All reactions were performed in 96-well flat-bottom plates in complete medium at 37°C in 5% CO2.
SP2/0 mouse myeloma cells were used as spontaneously proliferating cells.
Suppressors of major proliferation inhibitors. Each of the suppressors of major proliferation inhibitors was added to the culture of proliferating cells simultaneously with the neutrophil supernatant for subsequent estimation of [3H]thymidine incorporation. The following suppressors were used: anti-IL-10 antibodies (0.1 µg/ml; R&D Systems) to neutralize IL-10; anti-TGF-β antibodies (3 µg/ml; R&D Systems) to neutralize transforming growth factor-β; nor-NOHA (100 µM; Cayman Chemical, USA) to neutralize arginase; and indomethacin (2 µg/ml; Sigma-Aldrich) to neutralize prostaglandins.
Transwell experiments. To determine the necessity for cell–cell contacts and to estimate the effect of medium depletion on [3H]thymidine incorporation, we used a Transwell unit with a pore diameter of 0.4 µm. The lower chamber of the 24-well plate contained APCs (mitomycin C-treated splenocytes, 6·105 cells/well), specific T cell lines (5·104 cells/well), and mycobacterial sonicate (10 µg/ml). The upper chamber contained either macrophages, neutrophils, or splenocytes (all types of cells, 4·105 cells/well). After 24 h, methyl-[3H]thymidine (0.5 µCi) was added to the lower chamber, and thymidine incorporation into T cell lines was determined as described below.
[3H]Thymidine incorporation was tested in 96-well flat-bottom plates. Twenty-four hours after plating the cells, methyl-[3H]thymidine (0.5 µCi) was added. The cells were incubated for another 18 h; the contents of the wells were then transferred to glass filters using a semiautomatic cell harvester (Scarton, Norway). Label incorporation was measured with a liquid scintillation counter (Wallac, Finland).
Proliferation assay based on CFSE incorporation. Myeloma cells (108) were incubated in 1 ml of PBS containing 5 µM carboxyfluorescein succinimidyl ester (CFSE) for 8 min in the dark at room temperature. The reaction was stopped by addition of 10 ml of cold PBS. Labeled cells were washed two times by centrifugation in PBS and used for proliferation assay. The fluorescence of a cell aliquot was recorded immediately (time, 0); the rest of the cells were plated in a 24-well plate (3·105 cells/ml per well) in complete medium with or without addition of the neutrophil supernatant. Cell proliferation was estimated after 24 and 40 h of incubation from a decrease in fluorescence using a FACS Calibur flow cytometer (BD Bioscience, USA).
HPLC. Neutrophil supernatants were filtered on an Amicon Ultra-4 centrifuge filter (membrane cut-off, 10 kDa). The filtrate was loaded on a Symmetry 300C4 5 µm column (4.6 × 250 mm) using a 1-ml loop and fractionated in a 0-60% linear gradient of acetonitrile (gradient grade for HPLC; Merck, Germany) in 0.1% TFA (HPLC grade; Sigma) in water (Milli-Q purified; resistance, >18 MΩ/cm) for 15 min at a flow rate of 1 ml/min. Fractions were monitored at 280 and 214 nm at 20°C and collected manually every 2 min. Each fraction was evaporated on a centrifuge concentrator at 30°C, dissolved in complete medium, and tested for biological activity (inhibition of thymidine incorporation). The fraction with biological activity was rechromatographed on the same column in a 10-30% gradient of acetonitrile in 0.1% TFA for 15 min. Fractions were collected every 60 s and tested for biological activity. The fraction with biological activity was purified in isocratic regime (4% acetonitrile in 0.1% TFA) on a Symmetry C18 5 µm column (4.6 × 250 mm). The purity of the preparations was tested by HPLC on a Symmetry C18 5 µm column (4.6 × 250 mm) in the same isocratic regime. The collected active fraction was analyzed by electrospray ionization time-of-flight (ESI-TOF) mass spectrometry.
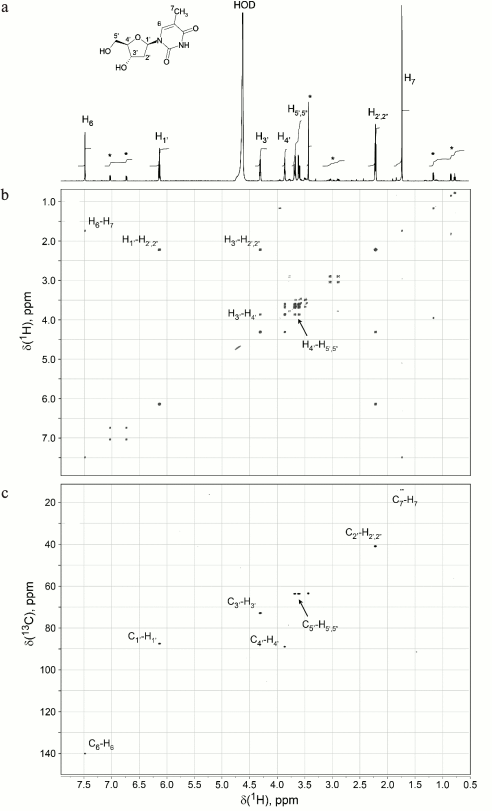
NMR spectra were registered with a Bruker Avance 600 MHz NMR spectrometer (Bruker, Germany) equipped with a 5-mm TXI Cryoprobe and a system of magnetic field pulse gradients. The 3.1.1 fraction containing the “neutrophil factor” was frozen and lyophilized to remove water and acetonitrile. Dry residue (~15 µg) was dissolved in 320 µl of D2O with a high deuterium content (99.99%) (Cambridge Isotope Laboratories, Inc., USA) and transferred to a Shigemi ampule with a magnetic susceptibility matched to D2O. To determine the structure of the identified compound, double-quantum filtered correlation (DQF-COSY) and heteronuclear single quantum correlation (13C-1H-HSQC) spectra were recorded.
RESULTS AND DISCUSSION
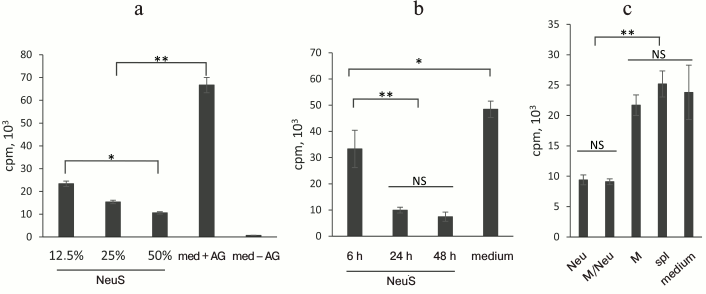
Neutrophils secrete soluble inhibitor of [3H]thymidine incorporation by T lymphocytes. First, we attempted to reproduce the results of D’Avila et al. [17] and, indeed, found that cocultivation of peritoneal exudate macrophages with immune T lymphocytes in the presence of the antigen induced a pronounced response and [3H]thymidine incorporation into T lymphocytes. Addition of neutrophils to the culture considerably reduced label incorporation (data not shown). We checked whether this inhibitory effect resulted from direct cell–cell contacts or was mediated by a soluble factor. For this purpose, the cultural medium in which neutrophils had been incubated was added to a coculture of macrophages and T lymphocytes, and the label incorporation was assayed. We found that the after incubation of neutrophils for 48 h, their cultural fluid suppressed label incorporation into T lymphocytes in a dose-dependent manner (Fig. 1a). Since the lifetime of neutrophils in a culture is only 24-48 h [5], we studied whether the inhibitory factor is actively secreted by the cells or is released into the medium after neutrophil death at the second day of culturing. Figure 1b shows that even after 6 h of neutrophil culturing, their cultural fluid significantly decreased label incorporation. The number of dead cells has not changed within 6 to 24 h of culturing and comprised less than 8% of total number of cells (data not shown). At the same time, the concentration of the “neutrophil factor” in the medium rapidly increased during this period, as follows from the inhibition of label incorporation, due probably to the activation of the “neutrophil factor” secretion by neutrophils.
Fig. 1. Neutrophil-secreted factor suppresses label incorporation by T lymphocytes. a) Addition of neutrophil supernatant (NeuS) to the cell culture of specific T lymphocytes proliferating in the presence of an antigen and APCs decreases [3H]thymidine incorporation; the effect is dose-dependent (12.5, 25, or 50% of NeuS in the medium); control, T lymphocyte proliferation in complete medium in the presence (med + AG) or absence (med – AG) of antigen. b) “Neutrophil factor” appears in the medium after 6 h of cultivation, its concentration increasing with the increase in the culturing time. Proliferation of T lymphocytes in the presence of NeuS collected after 6, 24, and 48 h of neutrophil culturing. c) Suppression of label incorporation is observed due to diffusion of the “neutrophil factor” through the membrane in the Transwell plate, but not due to the media depletion. Cells in the upper chamber: Neu, neutrophils. M/Neu, a mixture of neutrophils and macrophages; M, macrophages; spl, intact splenocytes. Data were obtained in 2-6 independent experiments; the results are presented as mean ± SD; * p < 0.05; ** p < 0.001; NS, no significant difference.
One of the reasons for the decreased T cell proliferation (as estimated from the inhibition of [3H]thymidine incorporation) in the presence of the cultural fluid might be partial substitution of complete medium by the medium depleted as a result of neutrophil culturing. To test this suggestion, antigen-responding cells were placed in a lower chamber of a Transwell unit that was separated from the upper chamber by a cell-impermeable membrane. Cells producing the studied factor were placed in the upper chamber. Figure 1c shows that neutrophils, but not macrophages or nonfractionated splenocytes, produced the inhibitory factor, although medium depletion took place for all types of the tested cells.
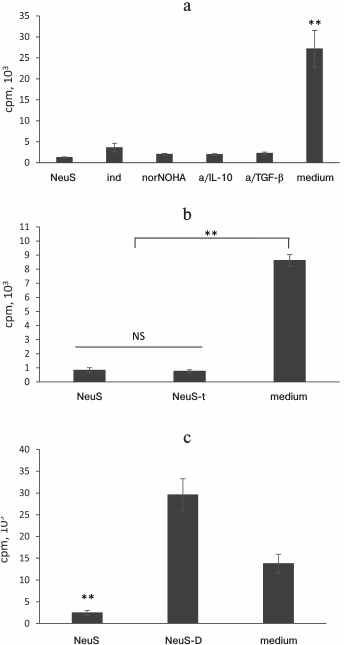
Standard approaches did not eliminate the inhibitory effect of the “neutrophil factor”. Prostaglandin E2, arginase, IL-10, and TGF-β are major soluble mediators that suppress activation of T cells according to numerous studies on the interactions (including suppression) between neutrophils and T cells [16, 21]. The cultural fluid of neutrophils contained physiologically significant concentrations of arginase, IL-10, and TGF-β (data not shown). We conducted a series of experiments in which the activity of these molecules was inhibited to elucidate whether suppression of T cell proliferation resulted from the activity of these compounds. We found (Fig. 2a) that selective inhibition of any of these factors did not abolish suppression of label incorporation into T lymphocytes. Moreover, the inhibitory effect of the cultural fluid remained even after its boiling (Fig. 2b), which usually eliminates the effects of most proteins and relatively large nucleic acid fragments. Dialysis of the cultural fluid revealed that the molecular mass of the active factor is below 10 kDa (Fig. 2c).
Fig. 2. The factor that suppresses label incorporation is a heat-resistant low molecular weight compound. a) Effect of “neutrophil factor” (neutrophil supernatant, NeuS) is not abolished by suppression of major inhibitors of T cell proliferation, such as prostaglandin E2 (indomethacin, ind), arginase (norNOHA), IL-10 (anti-IL-10 antibodies, a/IL-10), and TGF-β (anti-TGF-β antibodies, a/TGF-β). b) The “neutrophil factor” does not lose its effect after boiling: NeuS-t, boiled NeuS. c) Molecular mass of the “neutrophil factor” does not exceed 10 kDa (according to dialysis). NeuS-D, dialyzed NeuS. Data were obtained in 2-3 independent experiments; the results are presented as mean ± SD; ** p < 0.001; NS, no significant difference.
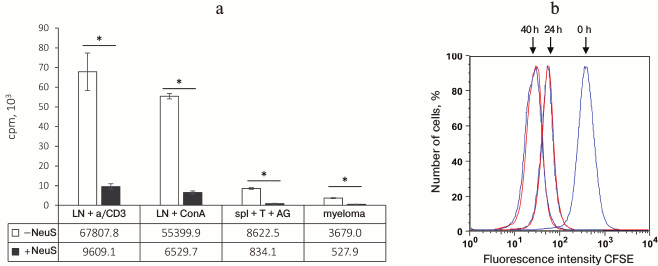
In the next series of experiments, we studied whether inhibition of label incorporation by proliferating cells depends on the character of proliferation-inducing stimulus, since all previous experiments were conducted using a classical system of specific antigen recognition by CD4+ T cells in the context of type II MHC. The low molecular weight “neutrophil factor” inhibited label incorporation not only after T cell stimulation by the antigen, but also in response to concanavalin A (stimulation via lectin receptor) or plastic-attached anti-CD3 antibodies (nonspecific activation via T cell receptors) (Fig. 3). The “neutrophil factor” also suppressed label incorporation into dividing myeloma cells (Fig. 3) (tumor growth in vitro was confirmed by visual monitoring under a microscope). These results led us to the conclusion that the observed phenomenon was not suppression of proliferation, but rather inhibition of [3H]thymidine incorporation during DNA biosynthesis. This suggestion was also confirmed by the fact that no proliferation suppression was observed when proliferation was estimated from CFSE incorporation by the flow cytometry method (Fig. 3b) and also by the absence of any influence of the “neutrophil factor” on the antigen-induced IFN-γ production by T cells (effector phase) (data not shown).
Fig. 3. “Neutrophil factor” suppresses [3H]thymidine incorporation, but not cell proliferation. a) Suppression of [3H]thymidine incorporation in various types of dividing cells after addition of neutrophil supernatant (NeuS): LN + a/CD3, naive lymph node cells stimulated with anti-CD3 antibodies; LN + ConA, naive lymph node cells stimulated with concanavalin A; spl + T + AG, response of specific T cells to antigen; myeloma, tumor spontaneous growth. b) NeuS addition to the myeloma cell culture (red) does not suppress proliferation 24 and 40 h after addition as compared to proliferation of control cells (blue) in complete cell culture medium. Cell proliferation was estimated from a decrease in CFSE fluorescence using a flow cytometer; 0 h, fluorescence measured immediately after myeloma labeling with CFSE. The data were obtained in 2-3 independent experiments; the results are presented as mean ± SD, * p < 0.0005.
The facts that boiling did not affect the activity of the “neutrophil factor” and that the factor displayed its activity in a simple proliferation system in response to concanavalin A allowed us to considerably simplify the test system. Samples were also analyzed by HPLC, mass spectrometry, and NMR methods.
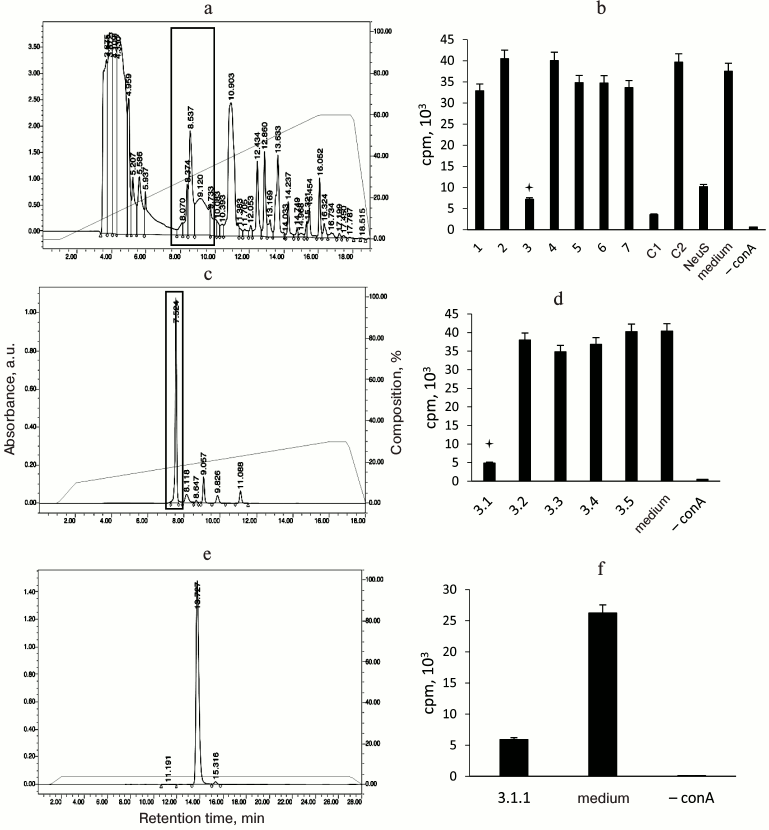
Thymidine is a factor that suppresses [3H]thymidine incorporation. Figure 4 shows subsequent fractionation of the dialyzed “neutrophil factor” by HPLC (left panel) and the effect of the resulting fractions on [3H]thymidine incorporation by stimulated T cells (right panel). The fraction with biological activity was 3.1.1. Its main peak corresponded to 242 Da, as determined by mass spectrometry. The fraction was then analyzed by the NMR method to identify the chemical structure of its major compound. The proton resonance spectrum (Fig. 5a) and two-dimensional DQF-COSY (Fig. 5b) and 13C-1H-HSQC (Fig. 5c) spectra indicated that this compound was thymidine. Its molecular mass and spectral parameters coincided with those described earlier [22]. Addition of the “neutrophil factor” to the culture of mouse EL4 thymoma caused rapid cell death (data not shown), since thymidine is highly toxic for this type of tumor cells [23].
Fig. 4. Purification of the “neutrophil factor” by HPLC (left panel) and analysis of biological activity of the resulting fractions (right panel): a) “neutrophil factor”-containing supernatant (after ultrafiltration on a 10-kDa membrane) was fractionated on an HPLC column into seven fractions; b) the fractions were tested for their ability to suppress [3H]thymidine incorporation into concanavalin A (conA)-stimulated proliferating T lymphocytes; the fraction with the inhibitory activity (marked with an asterisk and framed in the HPLC chromatogram, here and below) was (c) further fractionated by HPLC and (d) tested for activity. This resulted in the isolation of a single fraction 3.1.1 (e) with the inhibitory activity (f). NeuS, original neutrophil supernatant; C1, filtrate before column fractionation (components with molecular mass <10 kDa); C2, concentrate (components with molecular mass >10 kDa).
Fig. 5. The “neutrophil factor” suppressing thymidine incorporation is thymidine itself. NMR spectra of the “neutrophil factor” were used for deciphering its molecular structure: a) 1D proton resonance spectrum; b) DQF-COSY 2D spectrum; c) 13C-1H-HSQC 2D spectrum. The thymidine structural formula is shown on the top. The spectra show the assignment of 1H and 13C signals of thymidine. Asterisk, signals from minor unidentified impurity in the 1D spectrum.
Therefore, the factor that suppresses incorporation of labeled thymidine by dividing cell turned out to be excessive amounts of unlabeled thymidine spontaneously secreted into the cultural fluid by neutrophils. This discovery, which required almost three years of research efforts and considerable expenses, although disappointing, still is of considerable importance. First, it illustrates the necessity for using more than one commonly accepted method when studying biological phenomena (most immunologists a priori associate thymidine incorporation with proliferation), especially when numerous “hinting” publications on the topic exist. Besides, estimation of proliferation from [3H]thymidine incorporation into DNA still remains the most widely used approach, although new methods have been developed that include estimation of CFSE fluorescence, staining of nuclear markers of proliferation (ki67, PCNA, p53), and various colorimetry methods [24-26]. In our case, production by phagocytes of factors suppressing activation of T cells is a topic that has been widely studied during the last decade in different areas of immunology. Second, the biological meaning and the mechanistic explanation of the phenomenon of thymidine secretion by neutrophils remain obscure.
Similar phenomena are very poorly described in the literature. For the first time, thymidine secretion was found in studies of macrophages, but no reasonable biological explanation for this fact has been suggested [27, 28]. Thirty years later, active secretion of thymidine was demonstrated for some (but not all) hybridomas and myelomas [29], and it was emphasized that such secretion is uncharacteristic for non-tumor cells. Again, no hypothesis on the biological significance of this phenomenon has been proposed. After another 10 years, we added neutrophils to the list of cells actively secreting thymidine, but, similarly to our predecessors, we can only suggest a very broad explanation on the importance of this activity for cell functioning. In addition to the earlier suggestions that neutrophils secrete thymidine to suppress the growth of neighboring cells (indeed, extracellular thymidine is cytotoxic in relatively low concentrations), we propose a new hypothesis based on the specific properties of neutrophil physiology. Namely, neutrophils demonstrate a high level of DNA usage outside of the cell – they form the famous neutrophil extracellular traps (NETs) [30]. It remains unknown whether neutrophils synthesize the three other nitrogenous bases that form DNA, but extracellular secretion of at least one of the four bases gives reasons to consider possible relation between this process and formation and/or degradation of DNA traps.
Acknowledgements
We are grateful to A. Osterman (Sanford Burnham Medical Research Institute) and A. Richardson (Scripps Research Institute) for mass spectrometry analysis.
This work was supported by the Russian Science Foundation (A. S. Apt, project No. 14-15-00029; V. I. Polshakov, project No. 14-14-00598, registration and analysis of NMR spectra).
REFERENCES
1.Witko-Sarsat, V., Rieu, P., Descamps-Latscha, B.,
Lesavre, P., and Halbwachs-Mecarelli, L. (2000) Neutrophils: molecules,
functions and pathophysiological aspects, Lab. Invest.,
80, 617-653.
2.Nathan, C. (2006) Neutrophils and immunity:
challenges and opportunities, Nat. Rev. Immunol., 6,
173-182.
3.Kuijpers, T. W., Van den Berg, T. K., and Roos, D.
(2009) Phagocyte–Pathogen Interactions (Russel, D. G., and
Gordon S., eds.) ASM Press, Washington DC, pp. 3-26.
4.Quint, J. K., and Wedzicha, J. A. (2007) The
neutrophil in chronic obstructive pulmonary disease, J. Allergy
Clin. Immunol., 119, 1065-1071.
5.Eruslanov, E. B., Lyadova, I. V., Kondratieva, T.
K., Majorov, K. B., Scheglov, I. V., Orlova, M. O., and Apt, A. S.
(2005) Neutrophil responses to Mycobacterium tuberculosis
infection in genetically susceptible and resistant mice, Infect.
Immun., 73, 1744-1753.
6.Eum, S. Y., Kong, J. H., Hong, M. S., Lee, Y. J.,
Kim, J. H., Hwang, S. H., Cho, S. N., Via, L. E., and Barry, C. E.,
3rd. (2010) Neutrophils are the predominant infected phagocytic cells
in the airways of patients with active pulmonary TB, Chest,
137, 122-128.
7.Tanaka, D. (2006) Essential role of neutrophils in
anti-type II collagen antibody and lipopolysaccharide-induced
arthritis, Immunology, 119, 195-202.
8.Chin, A. C., and Parkos, C. A. (2006) Neutrophil
transepithelial migration and epithelial barrier function in IBD:
potential targets for inhibiting neutrophil trafficking, Ann. N. Y.
Acad. Sci., 1072, 276-287.
9.Soehnlein, O., Weber, C., and Lindbom, L. (2009)
Neutrophil granule proteins tune monocytic cell function, Trends
Immunol., 30, 538-546.
10.Yang, C. W., Strong, B. S. I., Miller, M. J., and
Unanue, E. R. (2010) Neutrophils influence the level of antigen
presentation during the immune response to protein antigens in
adjuvants, J. Immunol., 185, 2927-2934.
11.Jaeger, B. N., Donadieu, J., Cognet, C., Bernat,
C., Ordonez-Rueda, D., Barlogis, V., Mahlaoui, N., Fenis, A.,
Narni-Mancinelli, E., Beaupain, B., Bellanne-Chantelot, C., Bajenoff,
M., Malissen, B., Malissen, M., Vivier, E., and Ugolini, S. (2012)
Neutrophil depletion impairs natural killer cell maturation, function,
and homeostasis, J. Exp. Med., 209, 565-580.
12.Kondratieva, T. K., Rubakova, E. I., Linge, I.
A., Evstifeev, V. V., Majorov, K. B., and Apt, A. S. (2010) B cells
delay neutrophil migration toward the site of stimulus: tardiness
critical for effective bacillus Calmette–Guerin vaccination
against tuberculosis infection in mice, J. Immunol., 184,
1227-1234.
13.Kozakiewicz, L., Chen, Y., Xu, J., Wang, Y.,
Dunussi-Jannopopulos, K., Ou, Q., Flynn, J. L., Porcelli, S. A.,
Jacobs, W. R., Jr., and Chan, J. (2013) B cells regulate neutrophilia
during Mycobacterium tuberculosis infection and BCG vaccination
by modulating the interleukin-17 response, PLoS Pathog.,
9, e1003472.
14.Yang, D., De la Rosa, G., Tewary, P., and
Oppenheim, J. J. (2009) Alarmins link neutrophils and dendritic cells,
Trends Immunol., 30, 531-537.
15.Barquero-Calvo, E., Martirosyan, A.,
Ordonez-Rueda, D., Arce-Gorvel, V., Alfaro-Alarcon, A., Lepidi, H.,
Malissen, B., Malissen, M., Gorvel, J. P., and Moreno, E. (2013)
Neutrophils exert a suppressive effect on Th1 responses to
intracellular pathogen Brucella abortus, PLoS
Pathog., 9, e1003167.
16.Muller, I., Munder, M., Kropf, P., and Hansch, G.
M. (2009) Polymorphonuclear neutrophils and T-lymphocytes: strange
bedfellows or brothers in arms? Trends Immunol., 30,
522-530.
17.D’Avila, H., Roque, N. R., Cardoso, R. M.,
Castro-Faria-Neto, H. C., Melo, R. C., and Bozza, P. T. (2008)
Neutrophils recruited to the site of Mycobacterium bovis BCG
infection undergo apoptosis and modulate lipid body biogenesis and
prostaglandin E production by macrophages, Cell. Microbiol.,
10, 2589-2604.
18.Yeremeev, V., Linge, I., Kondratieva, T., and
Apt, A. (2015) Neutrophils exacerbate tuberculosis infection in
genetically susceptible mice, Tuberculosis (Edinb.), 95,
447-451.
19.Majorov, K. B., Lyadova, I. V., Kondratieva, T.
K., Eruslanov, E. B., Rubakova, E. I., Orlova, M. O., Mischenko, V. V.,
and Apt, A. S. (2003) Different innate ability of I/St and A/Sn mice to
combat virulent Mycobacterium tuberculosis: phenotypes expressed
in lung and extrapulmonary macrophages, Infect. Immun.,
71, 697-707.
20.Pichugin, A. V., Khaidukov, S. V., Moroz, A. M.,
and Apt, A. S. (1998) Capacity of murine T cells to retain long-term
responsiveness to mycobacterial antigens is controlled by the H-2
complex, Clin. Exp. Immunol., 111, 316-324.
21.Nagarai, S., Youn, J. I., and Gabrilovich, D. I.
(2013) Reciprocal relationship between myeloid-derived suppressor cells
and T-cells, J. Immunol., 191, 17-23.
22.Biological Magnetic Resonance Data Bank.
Thymidine
(http://bmrb.wisc.edu/metabolomics/mol_summary/show_data.php?molName=thymidine&id=bmse000244).
23.Reiter, H. (1979) Effect of thymidine on the
survival of mice with EL4 tumors, Cancer Res., 39,
4856-4860.
24.Lyons, A. B., and Parish, C. R. (1994)
Determination of lymphocyte division by flow cytometry, J. Immunol.
Methods, 171, 131-137.
25.Iatropoulos, M. J., and Williams, G. M. (1996)
Proliferation markers, Exp. Toxicol. Pathol., 48,
175-181.
26.Vega-Avila, E., and Pugsley, M. K. (2011) An
overview of colorimetric assay methods used to assess survival or
proliferation of mammalian cells, Proc. West Pharmacol. Soc.,
54, 10-14.
27.Opitz, H. G., Niethammer, D., Lemke, H., Flad, H.
D., and Huget, R. (1975) Inhibition of 3H-thymidine
incorporation of lymphocytes by a soluble factor from macrophages,
Cell. Immunol., 16, 379-388.
28.Stadecker, M. J., Calderon, J., Karnovsky, M. L.,
and Unanue, E. R. (1977) Synthesis and release of thymidine by
macrophages, J. Immunol., 119, 1738-1743.
29.Spilsberg, B., Rise, F., Petersen, D., and
Nissen-Meyer, J. (2006) Thymidine secretion by hybridoma and myeloma
cells, Biochem. Biophys. Res. Commun., 342, 221-226.
30.Vorobjeva, N. V., and Pinegin, B. V. (2014)
Neutrophil extracellular traps: mechanisms of formation and role in
health and disease, Biochemistry (Moscow), 79,
1286-1296.