Study of Age-Dependent Structural and Functional Changes of Mitochondria in Skeletal Muscles and Heart of Naked Mole Rats (Heterocephalus glaber)
S. Holtze1*, C. M. Eldarov2, V. B. Vays2, I. M. Vangeli2, M. Yu. Vysokikh2*, L. E. Bakeeva2*, V. P. Skulachev2, and T. B. Hildebrandt1
1Leibniz Institute for Zoo and Wildlife Research, Department of Reproduction Management, 10315 Berlin, Germany; E-mail: holtze@izw-berlin.de2Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, 119991 Moscow, Russia; E-mail: bakeeva@belozersky.msu.rui, mike@genebee.msu.ru
* To whom correspondence should be addressed.
Received July 8, 2016
Morphometric analysis of mitochondria in skeletal muscles and heart of 6- and 60-month-old naked mole rats (Heterocephalus glaber) revealed a significant age-dependent increase in the total area of mitochondrial cross-sections in studied muscle fibers. For 6- and 60-month-old animals, these values were 4.8 ± 0.4 and 12.7 ± 1.8%, respectively. This effect is mainly based on an increase in the number of mitochondria. In 6-month-old naked mole rats, there were 0.23 ± 0.02 mitochondrial cross-sections per µm2 of muscle fiber, while in 60-month-old animals this value was 0.47 ± 0.03. The average area of a single mitochondrial cross-section also increased with age in skeletal muscles – from 0.21 ± 0.01 to 0.29 ± 0.03 µm2. Thus, naked mole rats show a drastic enlargement of the mitochondrial apparatus in skeletal muscles with age due to an increase in the number of mitochondria and their size. They possess a neotenic type of chondriome accompanied by specific features of mitochondrial functioning in the state of oxidative phosphorylation and a significant decrease in the level of matrix adenine nucleotides.
KEY WORDS: naked mole rat, morphometry, respiration, aging, ultrastructure, mitochondria, neotenyDOI: 10.1134/S000629791612004X
Mechanisms of physiological aging of an organism and development of age-related pathologies remain key problems in biology. Therefore, studying species with a naturally reduced rate of aging is of particular interest. Such studies provide new information on the mechanisms of aging and, as a result, allow finding a way to control these processes. The naked mole rat (Heterocephalus glaber) belongs to this group of species. This miniature (average body mass ~ 35 g) rodent lives in underground burrows in arid and semi-arid areas of Kenya, Somalia, and Ethiopia. Naked mole rat burrows are dark and poorly ventilated with presumed low oxygen and high carbon dioxide levels [1-7]. Heterocephalus glaber is a naked rodent with a wide head, long incisors adapted for digging, and short but flexible legs which allow quick movements both forward and backward [8]. This species is characterized by some features unique for mammals: a complex social organization of colonies, slow metabolism, low sensitivity to certain pain types (thermal burns and chemical burns caused by acids), and high resistance to brain hypoxia [9-12]. Life expectancy is one of the most interesting features of the naked mole rat. Currently, the longevity record for naked mole rats living in captivity is over 31 years [13]. It has also been shown that up to 24 years of age, death rate rises only slightly across all age groups [13]. The highest mortality rates are observed during the first two months of life; adult animals rarely get sick and die.
Currently, scientists actively search for an explanation of the phenomenon of H. glaber’s longevity. The free radical theory remains one of the most popular theories of aging [14]. According to this theory, an aging organism is damaged by reactive oxygen species (ROS) formed in mitochondria [15-27]. Therefore, it seems very important to study mitochondria from naked mole rat tissues. According to certain data, the amounts of ROS produced by tissues of the naked mole rat and short-lived species (for example, mice) are comparable [28, 29]. The ultrastructure of the naked mole rat mitochondria has not been studied. There is evidence of the immutability of the mitochondrial mass during the entire lifespan of the naked mole rat [30], but these data are based on the assessment of the level of porin expression, and the research was performed only for the heart muscle of animals not younger than 2 years. The described results were not illustrated by imaging studies, even though the method of electron microscopy remains the most effective in evaluation of the chondriome in the tissues of organisms: it allows not only to see the structure of mitochondria, but also to perform morphometric evaluation of several chondriome features based on the obtained images [31].
It should be noted that there is one characteristic sign of aging, sarcopenia, present in virtually all animals. Sarcopenia is the age-dependent change in the structural and functional state of skeletal muscles associated with both decrease in muscle mass and frequency of contractions. It has been repeatedly noted that the condition of the mitochondrial apparatus plays the leading role in muscle aging. In addition, the significance of the age-dependent mitochondrial dysfunction as well as the depletion of chondriome structure is widely recognized (for reviews, see [32, 33]).
However, there are no data available on ultrastructural studies of naked mole rat tissues except for the work by Onyango et al. performed on the testicular tissue of the naked mole rat [34]. Therefore, we decided to study the chondriome structure in the skeletal and cardiac naked mole rat muscular tissues. We used the method of morphometric analysis to identify age-dependent changes in the ultrastructure of the mitochondrial apparatus in naked mole rat muscles compared to the same parameters in short-lived rodents. Earlier we had already described age-dependent changes in the ultrastructure of mitochondria from skeletal muscle fibers of short-lived animals such as Wistar rats [31]. Data obtained earlier and in this study are analyzed and presented together in this article.
MATERIALS AND METHODS
Animals. The study was carried out on two groups of naked mole rats (6- and 60-month-old). Each group contained four male workers. Naked mole rat colonies are kept at the Leibniz-Institute for Zoo and Wildlife Research (Berlin) in artificial plexiglass labyrinths. The temperature in the system was maintained at 26-29°C, and relative humidity was rather high, 60-80%. The boxes contained wooden litter, small twigs, and pieces of paper. Fresh food was available daily without restrictions and included sweet potatoes, carrots, apples, fennel, groats with vitamins and minerals, and oat flakes. Experiments were approved by the Ethics Committee of Landesamt für Gesundheit und Soziales, Berlin, Germany (#ZH 156; G 0221/12; T 0073/15).
Isolation of heart mitochondria. Mitochondrial fractionation was performed by differential centrifugation with our modifications. After anesthesia of the animal by CO2 or isoflurane and cervical dislocation, hearts of fifteen 18.5-day-old mouse embryos or five adult 2.5-month-old female mice of the C57BL/6 line or five 60-month-old naked mole rats were collected. They were placed on a concave surface of a frosted glass previously cooled to 0°C, weighed, and transferred under an MBS10 binocular microscope (Lomo, Russia). The heart was crushed by a cooled Teflon pestle and ground to a homogenous state. The suspension was scraped by an ocular scalpel blade and was quantitatively transferred to a 1.5-ml plastic tube. Isolation medium was added to the tube: 10 mM Tris, 0.25 M sucrose, 0.5 mM EDTA, 0.5 mM EGTA, pH 7.5, with 0.1% bovine serum albumin at a ratio of 10 ml/g of original heart weight. The suspension was stirred by a single inversion, and the precipitate was collected by centrifugation for 10 s at 10,000g on a high-speed tabletop centrifuge (ScientificPlastic, USA). Then it was homogenized twice in a microhomogenizer (glass/Teflon) in 0.5 ml of the isolation medium at 4°C and centrifuged for 10 min at 1000g and 4°C to precipitate heavy fractions such as nuclei, Golgi apparatus, unbroken cells, etc. The supernatant was collected and centrifuged for 10 min at 10,000g and 4°C. The pellet was resuspended in the isolation medium without BSA with the same ratio between the original tissue weight and volume. Then it was centrifuged under the same conditions, the supernatant was removed, and the pellet was resuspended in a minimal volume of the isolation medium without BSA, but with 5 µM EGTA. Mitochondrial protein concentration was determined using bicinchoninic acid in accordance with the manufacturer’s recommendations (Pierce, Waltham, USA).
Polarography. Respiration rate of isolated mitochondria in various metabolic states was determined by measuring the decrease in oxygen content in a closed-type cell using a Clark electrode on a two-channel polarograph (Oroboros, Innsbruck, Austria) at 25°C. Mitochondria were energized by adding glutamate and malate (ratio 4 mM/1 mM) to the incubation medium (10 mM Tris, 0.25 M sucrose, 5 µM EGTA, pH 7.5). ADP (0.01-0.5 mM) was used to stimulate respiration and to determine its rate in the phosphorylating state 3. Maximal rate of coupled respiration was determined in the presence of 0.5 mM ADP, and in state 4 – in the presence of 1 µM oligomycin. Maximal rate of uncoupled respiration was determined during titration by aliquots of 1 µl FCCP (10 µM) with a discrete increment of the final uncoupler concentration of 10 nM in the cuvette.
Adenine nucleotides were determined in tissue samples by measuring luminescence in the luciferin–luciferase system and the system of coupled enzymes by Ugarova’s method on a chemiluminometer (Protego, Russia) [35].
Electron microscopy. Tissues from thigh muscles (musculus vastus lateralis, -medius, -intermedius) were taken for the experiment. Material was fixed by 3% glutaraldehyde solution in phosphate buffer, pH 7.4, for 2 h at 4°C; then it was additionally fixed in 1% osmium tetroxide solution in the buffer for 1.5 h and was dehydrated in alcohol solutions with increasing ethanol concentrations (70% ethanol was saturated with uranyl acetate). The material was put into Epon-812 epoxy resin. Serial ultrathin sections were cut using a Leica ultramicrotome and stained with lead (Reynolds method [36]). The preparations were viewed and photographed on a JEOL JEM-1400 (JEOL, Tokyo, Japan) electron microscope with a QUEMESA digital camera.
Morphometry and statistical analysis. Twenty electron microscopic photographs (magnification ×1500) for each group of animals were selected for morphometric analysis. In these photos, mitochondria of muscle fibers were marked using the Adobe Photoshop (Adobe®, San Jose, USA) graphical editor, and the number of mitochondrial cross-sections was estimated using the Count tool. The data and the photograph scaling allowed calculation of the following parameters using the Photoshop analysis package: (i) the number of mitochondrial cross-sections per square micrometer of muscle fibers; (ii) the average area of mitochondrial cross-sections; (iii) the ratio of the total area of mitochondrial cross-sections in one cut to the total area of the muscle fiber, which determines the volume fraction of mitochondria in the fiber volume.
STATISTICA 8.0 (StatSoft Inc., Tulsa, USA) software was used for statistical analysis of the data. The Student and Mann–Whitney criteria were used to evaluate the reliability of the results.
RESULTS
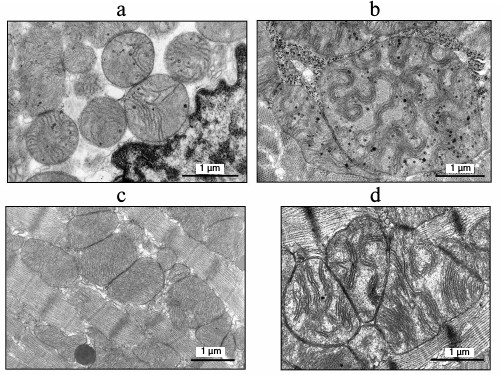
Figure 1a shows a cross section of a skeletal muscle of a 6-month-old naked mole rat. The ultrastructure of the muscle fibers corresponds to the norm. The chondriome in the muscle fibers is represented by individual mitochondria located mainly in isotropic areas (Fig. 1a; mitochondria are shown by arrows).
Fig. 1. Ultrastructure of the cross section of a skeletal muscle fiber: a) of a 6-month-old naked mole rat; b) of a 60-month-old naked mole rat; c) of a 2-day-old Wistar rat; d) of a 30-month-old Wistar rat ((c) and (d) taken from the article by Bakeeva et al. [37]). Arrows indicate mitochondria.
Figure 1b shows an electron micrograph of a skeletal muscle cross section of a 60-month-old naked mole rat. Numerous individual mitochondria seen in the picture do not form the structure of a mitochondrial reticulum. To compare the data on the age-dependent chondriome changes with previously published results on studies of mitochondria of short-lived rodents, Fig. 1c shows an electron micrograph of a cross section of a diaphragm muscle fiber of a 2-day-old Wistar rat. The distribution of individual mitochondria located in the isotropic zone is similar to the image seen in Fig. 1a for a 6-month-old naked mole rat. However, the cross section of the skeletal muscle of a 30-month-old Wistar rat clearly shows a complex system of mitochondrial reticulum (Fig. 1d). However, mitochondria remain individual organelles and do not form reticular structures in the skeletal muscle of an adult naked mole rat (Fig. 1b).
Figure 2a shows the ultrastructure of cardiomyocyte mitochondria from a 6-month-old naked mole rat. We see an abundantly expressed matrix and few randomly located cristae. At the same time, in the case of adult naked mole rats, the electron micrographs of cardiomyocytes clearly show that mitochondrial cristae are convoluted, twisted and assembled in stacks (Fig. 2b). Bakeeva et al. [38] showed earlier that cardiomyocyte cristae of 2.5-month-old Wistar rats have a classic ultrastructure with tightly packed parallel cristae (Fig. 2c), while in the case of 30-month-old Wistar rat cardiomyocyte mitochondria (Fig. 2d) the cristae display a mild wave shape and are assembled in stacks [38], similar in their morphology to those of a 60-month-old naked mole rat (Fig. 2b).
Fig. 2. Inner ultrastructure of cardiomyocyte mitochondria: a) of a 6-month-old naked mole rat; b) of a 60-month-old naked mole rat; c) of a 2.5-month-old Wistar rat; d) of a 30-month-old Wistar rat.
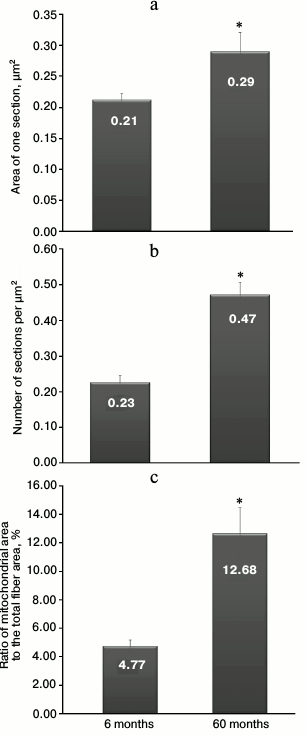
Morphometric analysis of electron micrographs of longitudinal sections was carried out to evaluate age-dependent dynamics of the chondriome of skeletal muscle fibers. Figure 3a shows the results of the measurements of the average sectional area of muscle fiber mitochondria. This parameter was 0.21 ± 0.01 µm2 for 6-month-old naked mole rats. With age, the average area of a single mitochondrion increases, reaching 0.29 ± 0.03 µm2 in 60-month-old animals. These results are significant at the level of p < 0.05.
Fig. 3. a) Average values of a sectional area of muscle fiber mitochondria in 6- and 60-month-old naked mole rats. b) Average values of the number of mitochondria per 1 µm2 of muscle fiber in 6- and 60-month-old naked mole rats. c) Average values of the ratio of chondriome area to the total area of muscle fiber in 6- and 60-month-old naked mole rats (in %). * The difference is significant at p < 0.05. Error bars on all the graphs correspond to the standard error of the mean.
The number of mitochondrial sections per 1 µm2 of a muscle fiber is the second parameter characterizing the changes in the mitochondrial apparatus of the naked mole rat with age (Fig. 3b). The average number of mitochondria in a skeletal muscle was shown to be 0.23 ± 0.02 per µm2 in 6-month-old animals. With age, we observed a more than twofold increase in the number of skeletal muscle mitochondria, reaching up to 0.47 ± 0.03 per µm2 in 60-month-old animals. This difference is significant at the level of p < 0.05.
Furthermore, we calculated the ratio of the chondriome area to the total area of the muscle fiber (in %; Fig. 3c). At the age of 6 months, the share of mitochondria was 4.8 ± 0.4%. With age, it significantly increased; for 60-month-old animals, the parameter was almost three times as high: 12.7 ± 1.8%, p < 0.05.
To compare age-related changes in the functional state of the chondriome of naked mole rats and short-lived rodents, we determined parameters of respiration and content of adenine nucleotides in mitochondria isolated from cardiac tissue. We found the content of adenine nucleotides in cardiac muscle mitochondria of a 60-month-old naked mole rat to be two times below that of adult mice; its value was closer to that of a mouse embryo (table).
Content of adenine nucleotides and respiration rates in cardiac
mitochondria of naked mole rats and of mice
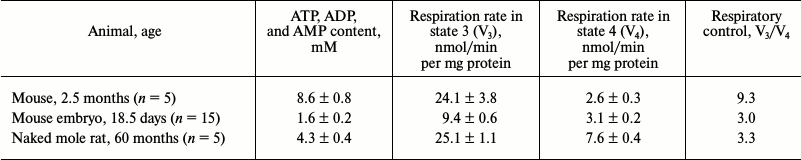
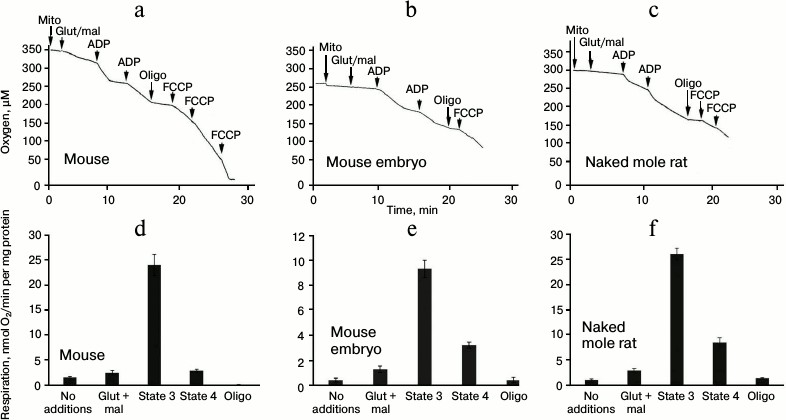
In addition, measurements of respiration rate in isolated mitochondria in different states demonstrated that there is no significant difference between naked mole rats and adult mice in terms of absolute values of respiration rate in heart mitochondria energized by a glutamate/malate mixture in the absence of added ADP (2.3 ± 0.5 and 2.5 ± 0.7 nmol O2/min per mg protein, respectively). However, the respiration rate of cardiac mitochondria of a 18.5-day-old mouse embryo was substantially lower (1.4 nmol O2/min per mg protein; Fig. 4). Addition of 0.1 mM ADP to the incubation medium caused a sharp acceleration of respiration. After the depletion of added ADP, the respiration rate significantly dropped in adult mouse mitochondria (transition to state 4; Fig. 4, a and d). In contrast, in cardiac mitochondria from the naked mole rat (Fig. 4, c and f) and mouse embryos (Fig. 4, b and e) the decrease in respiration rate during transition from state 3 to state 4 is not as significant, which leads to a considerable reduction of respiratory control values (table).
Fig. 4. Respiration in mitochondria of C57BL/6 mice and naked mole rats. a, d) 3-month-old adult mouse; b, e) 18.5-day-old embryo of C57BL/6 mouse; c, f) 60-month-old naked mole rat. a-c) Typical polarograms (dependence of oxygen consumption on time); d-f) rate of oxygen consumption by mitochondria in various metabolic conditions. All additions are as described in the “Materials and Methods” section. State 3 on the charts corresponds to the rate of oxygen consumption after the first addition of 0.1 mM ADP, and state 4 – after the depletion of added ADP.
DISCUSSION
Studying morphological changes in muscle tissues requires, along with electron microscopy methods, morphometric evaluation of various tissue parameters that supplement visual electron microscopy data with important numerical parameters characterizing the changes in the functional state of the tissue. The state of the chondriome is one of the most significant determinants for muscle tissues. Depending on the purpose of the research, its assessment can be based on parameters such as the number of mitochondria per cell unit, average size, ratio of greater diameter to smaller, circularity, area of the inner membrane per volume unit, etc. According to the literature, the so-called point count method is mainly used to measure various morphometric parameters in both muscle and other tissues. This method, originally proposed by A. A. Glagolev [39], became widespread after a series of studies by McCallister [40-42] and was later summarized by Weibel in the handbook on practical methods of morphometry [43]. With this method, a test system consisting of points or segments is applied to a section; the number of points or intersections of the segments that fall on the profiles of various cell structures are counted [43]. This method was used to obtain a large amount of data on changes in heart structure, in particular, age-related changes in the size of ventricles and mitochondrial apparatus [44, 45], hypertrophy [40], thyroxine treatment [41], and other pathologies. Many studies on the morphometry of lung tissues and blood vessels of various mammal and bird species also used the point count method, which, however, cannot provide an accurate, objective, and statistically reliable assessment of the state of mitochondrial ultrastructure [46-50].
In our work, we tried to completely abandon the conventional use of the point and segment system, choosing digital data processing with the Adobe Photoshop graphical editor. The results obtained in preliminary studies dedicated to the development of this method [31] showed the efficiency of our approach. The method used in this study significantly simplifies the process of morphometric measurements and offers good opportunities for its further optimization using image recognition technologies. For example, in this study the electron microscopic examination revealed a significant age-related increase in the mitochondrial apparatus of skeletal muscle fibers of the naked mole rat. Morphometric analysis of electron micrographs of the naked mole rat muscle tissues showed the degree of chondriome increase and the nature of the changes in mitochondrial ultrastructure. An almost three-fold increase in the ratio of chondriome area to the total area of the muscle fiber is observed in the aging skeletal muscles of the naked mole rat (between 6-60 months of age; this parameter changes from 4.8 ± 0.4 to 12.7 ± 1.8%; Fig. 3c). The increase in chondriome volume is largely due to the increase in the number of organelles – the number of mitochondrial sections per µm2 of the muscle fiber increases from 0.23 to 0.47 (Fig. 3b). At the same time, mitochondria also increase in volume: the mean sectional area of a single mitochondrion from muscle fibers increases from 0.21 to 0.29 µm2 (between 6-60 months of age; Fig. 3a).
Studying the functional state of mitochondria isolated from the cardiac tissue of 60-month-old naked mole rats and mice of various ages revealed a feature specific for mitochondria from the naked mole rat: a relatively high respiration rate was observed after the depletion of added ADP (Fig. 4). Interestingly, a similar effect was observed in mitochondria isolated from the cardiac muscle of rodent embryos [51]. This coupled respiration is sensitive to both oligomycin and carboxyatractylate, inhibitors of H+-ATP-synthase and ATP/ADP antiporter, respectively. If a high respiration rate after the depletion of ADP leads to the reduction of mitochondrial membrane potential, it can be interpreted as a mechanism of reducing ROS production during reverse electron transfer in the mitochondrial respiratory chain. This phenomenon was found in both, adult naked mole rats, as well as in embryos and newborns of short-lived rodents. This observation is in good agreement with the previously demonstrated fact of a strict dependence of ROS generation on mitochondrial membrane potential [52]. Furthermore, we can assume that embryos of most mammals and of naked mole rats of all ages possess an ATPase that hydrolyzes the ATP synthesized by the H+-ATP-synthase in the mitochondrial matrix and transferred into mitochondrial intermembrane space by the adenine nucleotide translocator. Such an enzyme may be responsible for the “mild uncoupling” of phosphorylating respiration.
The hypothetical ATPase responsible for this “mild uncoupling” will be inhibited by increasing ADP levels, thus ATP hydrolysis by this ATPase will cease in metabolic state 3 in case of ADP excess. Consequently, this ATPase does not compete with other ATPases, for example, actomyosin ATPase, which is active during muscle contraction. However, this ATPase will be activated in the state of rest, when the ADP level is close to depletion.
Thus, by comparing the levels of adenine nucleotides in cardiac mitochondria of 60-month-old naked mole rats, adult mice, and 18.5-day-old mouse embryos, we found unusually low levels in the naked mole rat. It should be noted that Aprille and Asimakis [51] reported in 1980 that mitochondria from neonatal rat liver contained significantly less adenine nucleotides than mitochondria from the liver of adult animals, and that this difference quickly disappeared after birth.
All data – those from visual observations, morphometric ultrastructural analysis, as well as that on the functional state of mitochondria, show that the chondriome, being very poorly developed in 6-month-old naked mole rats, powerfully increases in ones that have reached the age of 60 months. We can also observe functional neoteny of the most important mitochondrial parameter – the rate of phosphorylating respiration.
This result is of special interest since it contrasts with data on age-related changes in the ultrastructure of the mitochondrial apparatus in skeletal muscles of short-lived rodents. It has already been noted that the commonly accepted view is that the degree of sarcopenia and its consequences for the state of skeletal muscles increase with age [53]. Our studies [31] have shown that 24-30-month-old rats demonstrate degradation of a unified system of mitochondrial reticulum in skeletal muscles, and the ratio of the total mitochondrial area to the total area of the muscle fiber isotropic zone is reduced. However, in the case of naked mole rats, 60-month-old animals not only lack pathological changes in the mitochondrial apparatus of skeletal muscles, but on the contrary, their chondriome undergoes significant growth and development, which corresponds to the phenotype of a young animal. Mitochondria do not join to form a reticulum, which is yet another feature of neoteny in the naked mole rat.
Acknowledgements
The authors express their gratitude for help in experiments to the department of Prof. Alex Greenwood, to Dagmar Viertel from the laboratory of electronic microscopy, and her supervisor Dr. Gudrun Wibbelt, as well as to Michaela Morhart.
This study was supported by the Russian Science Foundation (projects Nos. 14-24-00107, isolation and biochemical experiments on mitochondria and analysis of their results; 14-50-00029, maintenance of animals and electron microscopy).
REFERENCES
1.Bennett, N. C., and Faulkes, C. G. (2000) The
Evolution of Sociality in African Mole Rats, Cambridge University
Press, Cambridge, UK.
2.Bennett, N. C., and Faulkes, C. G. (2000) Social
Organization in African Mole Rats, Cambridge University Press,
Cambridge, UK.
3.Brett, R. A. (1991) The Population Structure of
Naked Mole Rat Colonies, Princeton University Press, Princeton,
NJ.
4.Buffenstein, R., Park, R., Hanes, M., and Antwohl,
J. E. (2012) in The Laboratory Rabbit, Guinea Pig, Hamster, and
Other Rodents (Suckow, M. A., Stevens, K. A., and Wilson, R. P.,
eds.) Elsevier, London, pp. 1055-1074.
5.Jarvis, J. U. (1981) Eusociality in a mammal:
cooperative breeding in naked mole-rat colonies, Science,
212, 571-573.
6.Sherman, P. W., Jarvis, J. U., and Alexander, R. D.
(1991) The Biology of the Naked Mole-Rat, Princeton University
Press, Princeton, NJ.
7.Lacey, E. A., Patton, J. L., and Cameron, G. N.
(2000) Life Underground: The Biology of Subterranean Rodents,
University of Chicago Press, Chicago, IL.
8.Delaney, M. A., Nagy, L., Kinsel, M. J., and
Treuting, P. M. (2013) Spontaneous histologic lesions of the adult
naked mole rat (Heterocephalus glaber): a retrospective survey
of lesions in a zoo population, Vet. Pathol., 50,
607-621.
9.Buffenstein, R. (2000) Ecological and
Physiological Responses to Underground Habitats, University of
Chicago Press, Chicago, IL.
10.Larson, J., and Park, T. J. (2009) Extreme
hypoxia tolerance of naked mole rat brain, Neuroreport,
20, 1634-1637.
11.Maina, J. N., Gebreegziabher, Y., Woodley, R.,
and Buffenstein, R. (2001) Effects of change in environmental
temperature and natural shifts in carbon dioxide and oxygen
concentrations on the lungs of captive naked mole rats
(Heterocephalus glaber): a morphological and morphometric study,
J. Zool., 253, 371-382.
12.Park, T. J., Lu, Y., Juttner, R., Smith, E. S.,
Hu, J., Brand, A., Wetzel, C., Milenkovic, N., Erdmann, B.,
Heppenstall, P. A., Laurito, C. E., Wilson, S. P., and Lewin, G. R.
(2008) Selective inflammatory pain insensitivity in the African naked
mole rat (Heterocephalus glaber), PLoS Biol.,
6, e13.
13.Buffenstein, R. (2008) Negligible senescence in
the longest living rodent, the naked mole rat: insights from a
successfully aging species, J. Comp. Physiol. B, 178,
439-445.
14.Harman, D. (1956) Aging: a theory based on free
radical and radiation chemistry, J. Gerontol., 11,
298-300.
15.Lenaz, G. (2001) The mitochondrial production of
reactive oxygen species: mechanisms and implications in human
pathology, IUBMB Life, 52, 159-164.
16.Andreyev, A. Yu., Kushnareva, Yu. E., and
Starkov, A. A. (2005) Mitochondrial metabolism of reactive oxygen
species, Biochemistry (Moscow), 70, 200-214.
17.Honda, H. M., Korge, P., and Weiss, J. N. (2005)
Mitochondria and ischemia/reperfusion injury, Ann. N. Y. Acad.
Sci., 1047, 248-258.
18.Zweier, J. L., and Talukder, M. A. (2006) The
role of oxidants and free radicals in reperfusion injury,
Cardiovasc. Res., 70, 181-190.
19.Yellon, D. M., and Hausenloy, D. J. (2007)
Myocardial reperfusion injury, N. Engl. J. Med., 357,
1121-1135.
20.Eltzschig, H. K., and Eckle, T. (2011) Ischemia
and reperfusion – from mechanism to translation, Nat.
Med., 17, 1391-1401.
21.Borutaite, V., Toleikis, A., and Brown, G. C.
(2013) In the eye of the storm: mitochondrial damage during heart and
brain ischemia, FEBS J., 280, 4999-5014.
22.Chouchani, E. T., Pell, V. R., Gaude, E.,
Aksentijevic, D., Sundier, S. Y., Robb, E. L., Logan, A., Nadtochiy, S.
M., Ord, E. N., Smith, A. C., Eyassu, F., Shirley, R., Hu, C. H., Dare,
A. J., James, A. M., Rogatti, S., Hartley, R. C., Eaton, S., Costa, A.
S., Brookes, P. S., Davidson, S. M., Duchen, M. R., Saeb-Parsy, K.,
Shattock, M. J., Robinson, A. J., Work, L. M., Frezza, C., Krieg, T.,
and Murphy, M. P. (2014) Ischaemic accumulation of succinate controls
reperfusion injury through mitochondrial ROS, Nature,
515, 431-435.
23.Harman, D. (1972) The biologic clock: the
mitochondria? J. Am. Geriatr. Soc., 20, 145-147.
24.Miquel, J., Economos, A. C., Fleming, J., and
Johnson, J. E. (1980) Mitochondrial role in cell aging, Exp.
Gerontol., 15, 575-591.
25.Skulachev, V. P. (1997) Aging is a specific
biological function rather than the result of a disorder in complex
living systems: biochemical evidence in support of Weismann’s
hypothesis, Biochemistry (Moscow), 62, 1191-1195.
26.Skulachev, V. P. (1999) Phenoptosis: programmed
death of an organism, Biochemistry (Moscow), 64,
1418-1426.
27.Skulachev, V. P. (2001) Phenomena of programmed
death. Mitochondria, cells and organs: role of reactive oxygen species,
Soros. Obraz. Zh., 7, 4-10.
28.Labinskyy, N., Csiszar, A., Orosz, Z., Smith, K.,
Rivera, A., Buffenstein, R., and Ungvari, Z. (2006) Comparison of
endothelial function, O2– and
H2O2 production, and vascular oxidative stress
resistance between the longest-living rodent, the naked mole rat, and
mice, Am. J. Physiol. Heart. Circ. Physiol., 291,
H2698-2704.
29.Lambert, A. J., Boysen, H. M., Buckingham, J. A.,
Yang, T., Podlutsky, A., Austad, S. N., Kunz, T. H., Buffenstein, R.,
and Brand, M. D. (2007) Low rates of hydrogen peroxide production by
isolated heart mitochondria associate with long maximum lifespan in
vertebrate homeotherms, Aging Cell, 65, 607-618.
30.Csiszar, A., Labinskyy, N., Orosz, Z., Xiangmin,
Z., Buffenstein, R., and Ungvari, Z. (2007) Vascular aging in the
longest-living rodent, the naked mole rat, Am. J. Physiol. Heart.
Circ. Physiol., 293, H919-927.
31.Vays, V. B., Eldarov, C. M., Vangely, I. M.,
Kolosova, N. G., Bakeeva, L. E., and Skulachev, V. P. (2014)
Antioxidant SkQ1 delays sarcopenia-associated damage of mitochondrial
ultrastructure, Aging (Albany, NY), 6, 140-148.
32.Carter, H. N., Chen, C. C. W., and Hood, D. A.
(2015) Mitochondria, muscle health, and exercise with advancing age,
Physiology, 30, 208-223.
33.Del Campo, A., Jaimovich, E., and Tevy, M. F.
(2016) Mitochondria in the aging muscles of flies and mice: new
perspectives for old characters, Oxid. Med. Cell. Longev.,
2016, 9057593.
34.Onyango, D. W., and Oduorokelo, D. (1993)
Ultrastructural study of the testis of non-breeding naked mole-rat
(Heterocephalus glaber Ruppell), Ann. Anat., 175,
447-452.
35.Brovko, L., Romanova, N. A., and Ugarova, N. N.
(1994) Bioluminescent assay of bacterial intracellular AMP, ADP, and
ATP with the use of a coimmobilized three-enzyme reagent (adenylate
kinase, pyruvate kinase, and firefly luciferase), Anal.
Biochem., 220, 410-414.
36.Reynolds, E. S. (1963) The use of lead citrate at
high pH as an electron-opaque stain in electron microscopy, J. Cell
Biol., 17, 208-212.
37.Bakeeva, L. E., Chentsov, Y. S., and Skulachev,
V. P. (1981) Ontogenesis of mitochondrial reticulum in rat diaphragm
muscle, Eur. J. Cell. Biol., 25, 175-181.
38.Bakeeva, L. E. (2015) Age-related changes in
ultrastructure of mitochondria. Effect of SkQ1, Biochemistry
(Moscow), 80, 1582-1588.
39.Glagolev, A. A. (1941) Geometric Methods of
Quantitative Analysis of Aggregates under the Microscope [in
Russian], Gosgeolizdat, Moscow.
40.McCallister, B. D., and Brown, A. L. (1965) A
quantitative study of myocardial mitochondria in experimental cardiac
hypertrophy, Lab. Invest., 14, 692-700.
41.McCallister, L. P., and Page, E. (1973) Effects
of thyroxin on ultrastructure of rat myocardial cells: a stereological
study, J. Ultrastruct. Res., 42, 136-55.
42.McCallister, L. P., Page, E., and Power, B.
(1971) Stereological measurements of cardiac ultrastructures implicated
in excitation–contraction coupling, Proc. Natl. Acad.
Sci. USA, 68, 1465-1466.
43.Weibel, E. R. (1979) Stereological Methods.
Vol. 1. Practical Methods for Biological Morphometry, Academic
Press, London.
44.Sachs, H. G., Colgan, J. A., and Lazarus, M. L.
(1977) Ultrastructure of the aging myocardium: a morphometric approach,
Am. J. Anat., 150, 63-71.
45.Frenzel, H., and Feimann, J. (1984) Age-dependent
structural changes in the myocardium of rats. A quantitative light- and
electron-microscopic study on the right and left chamber wall, Mech.
Ageing Dev., 27, 29-41.
46.Maina, J. N. (1988) Morphology and morphometry of
the normal lung of the adult vervet monkey (Cercopithecus
aethiops), Am. J. Anat., 183, 258-267.
47.Maina, J. N. (2002) Some recent advances on the
study and understanding of the functional design of the avian lung:
morphological and morphometric perspectives, Biol. Rev. Camb.
Philos. Soc., 77, 97-152.
48.Maina, J. N., and King, A. S. (1987) A
morphometric study of the lung of a Humboldt penguin (Sphenicus
humboldti), Anat. Histol. Embryol., 16, 293-297.
49.Maina, J. N., and Nathaniel, C. (2001) A
qualitative and quantitative study of the lung of an ostrich
Struthio camelus, J. Exp. Biol., 204,
2313-2330.
50.Maina, J. N., and Van Gils, P. (2001)
Morphometric characterization of the airway and vascular systems of the
lung of the domestic pig Sus scrofa: comparison of the airway,
arterial and venous systems, Comp. Biochem. Physiol. A Mol. Integr.
Physiol., 130, 781-798.
51.Aprille, J. R., and Asimakis, G. K. (1980)
Postnatal development of rat liver mitochondria state 3 respiration,
adenine nucleotide translocase activity, and the net accumulation of
adenine nucleotides, Arch. Biochem. Biophys., 201,
564-575.
52.Korshunov, S. S., Skulachev, V. P., and Starkov,
A. A. (1997) High protonic potential actuates a mechanism of production
of reactive oxygen species in mitochondria, FEBS Lett.,
416, 15-18.
53.Marzetti, E., Hwang, J. C. Y., Lees, H. A.,
Wohlgemuth, S. E., Dupont-Versteegden, E. E., Carter, C. S., Bernabei,
R., and Leeuwenburgha, C. (2010) Mitochondrial death effectors:
relevance to sarcopenia and disuse muscle atrophy, Biochim. Biophys.
Acta, 1800, 235-244.