REVIEW: Vitamin D Deficiency in Europeans Today and in Viking Settlers of Greenland
H. Göring* and S. Koshuchowa
Göring Consulting, Mahlsdorfer Strasse 91, De 12555, Berlin, Germany; E-mail: horst-goering@online.de* To whom correspondence should be addressed.
Received September 15, 2016
The vast majority of the Earth’s population lives between the 20th and 40th parallel north and south. It seems that right here humans have found the best living conditions relating not only to temperature and food recourses, but also to UV radiation necessary for the production of vitamin D by human skin. An exception to this general rule is Europe. Nearly half a billion people live between the 40th and 60th parallel north of the equator despite the fact that the amounts of UV radiation there are much lower. Moreover, since the time of the Vikings, there has always been a part of the European population that lived even further north than the 60th parallel (the northern parts of Europe, including Greenland). In this work, we present the potential role that vitamin D deficiency might have played in the extinction of the Vikings of Greenland. We analyze factors that contribute to the discrepancy between the theoretical distribution of areas with vitamin D deficiency and today’s reality, like the impact of civilization, religious traditions, as well as vitamin D supplementation in food products and as a biologically active dietary additive. The global migration of people on a scale and speed never seen before is now even more important for this discrepancy.
KEY WORDS: vitamin D (deficiency), UV radiation, Europe, Greenland, Vikings, Scottish paradoxDOI: 10.1134/S0006297916120117
In our previous review on vitamin D [1], we examined the biological background of the role of vitamin D in animal and human life. It seems appropriate to analyze the epidemiological side of the role of vitamin D in human life to better understand its function and possible mechanism of its action in the organism.
Nowhere else on Earth do so many people live as far away from the equator as in Europe. What does it mean for their lives?
EUROPE: IS IT THE BEST PLACE FOR HUMAN EXISTENCE?
It has been established that modern humans (as a species) emerged close to the equator, in the highlands of East Africa. Their predecessors, the first representatives of the Homo genus, lived there already 2.8 million years ago. The H. sapiens species is believed to have appeared only 200,000 years ago [2]. In this area, “the naked newcomer” was in the optimal conditions: (i) sufficient amount of suitable food; (ii) suitable temperature despite the proximity of the equator; (iii) high intensity of solar radiation (including UV-B-radiation), which stimulates the formation of cholecalciferol known as vitamin D [1].
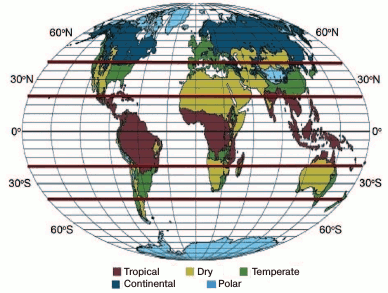
Obviously, over time, the population was growing and living conditions were deteriorating, which stimulated the beginning of human migration around 120,000 years ago. Humans migrated in search for food sources and suitable climatic conditions. It turned out that the optimal environmental conditions for human existence were located almost exclusively between 20° and 40° latitude N and S (Fig. 1) [3]. Small regions of moderate climate can be found also up to 50°N and S, especially in coastal areas. Nevertheless, there is one huge exception – Europe. Moderate climate in Europe extends to 60°N, i.e. up to the southern regions of Greenland due to the favorable effect of the warm Gulf Stream. Hunters-gatherers could find food in abundance in European forests, and temperature conditions were no longer as significant, as H. sapiens was already at such a developmental stage that they could resist the winter cold.
Fig. 1. Climatic zones of the Earth (after Köppen [39]) are shown in the form presented by [3]. All the regions of temperate and dry climate are located almost entirely at the same distance from the equator between 20° and 40°N and S. Note the only exception: Europe, which is between 35° and 70°N.
However, European people moved too far away from the equator. In Europe, the sun rises high enough for UV-B-radiation to reach the Earth’s surface for only a short time period. In winter, the daily dose of UV-B-radiation at 20° latitude is about 50% of its maximal value at the equator, while at 40° latitude it is already only 10% [4]. According to the results obtained by Jablonski and Chaplin [5], humans can exist under natural conditions without extra vitamin D supplement only up to 42° latitude [6]. At 55° latitude (Newfoundland, Newcastle, Copenhagen, Smolensk, Moscow, Novosibirsk, Kamchatka), the daily dose of UV-B radiation during four winter months is already so low that no vitamin D can be synthesized in human skin [4].
On the other hand, the length of a summer day gets longer when moving further away from the equator. That is why the summer daily dose of UV-B radiation at 40° latitude is hardly different from the equatorial one. Thus, the conditions at 40° latitude (Denver, Philadelphia, Madrid, Ankara, Beijing) are quite suitable for providing humans with vitamin D throughout the year given that we spend enough time in the air under the sun. The situation changes significantly when people move to the regions of higher latitude.
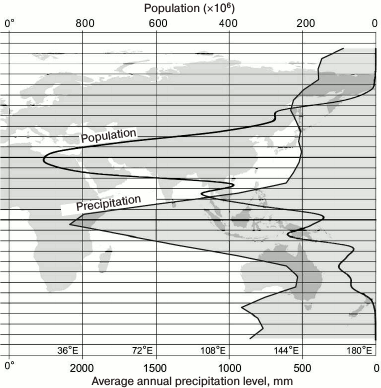
According to data published by Kummu and Varis [3], the majority of people living geographically above the 40° latitude are European (Fig. 2). The greater part of Europe is located between 40° and 60° latitude, i.e. exactly in the region where a markedly pronounced shoulder is observed on the population curve. Currently about 700 million people reside in Europe. Europeans constitute 53 and 86% of the population living above 40°N and 50°N latitude, respectively [3]. (If we could estimate the number of indigenous people who had lived in these latitudes several hundred years ago, the dominance of Europeans over the rest of the inhabitants would have probably been even more significant.) The remaining 16% are mostly Asians and people of North America, and they constitute only a tiny part of this population. In absolute terms, very small numbers of people living around 80°N latitude are residents of North America and Greenland (Inuit).
Fig. 2. Distribution of the total population depending on the geographical latitude [3] based on data from [40]. Distribution of the human population around the globe is shown within 5° of geographical latitude. Note the shoulder at 40-60°N latitude.
Holick et al. [7] showed that in the regions above 35° latitude vitamin D synthesis in human skin is virtually absent in winter months (data were obtained in Boston located at 42.21° latitude). In model experiments conducted by Holick et al., 7-dihydroxycholesterol dissolved in ethanol (in ampoules), was exposed for 1 h to the sun at noon in June and in October. In October, the efficiency of solar radiation in regard to vitamin D formation was five times lower than in June [8]. Similar results were obtained in 1988 in Edmonton (53.33°) [9].
Jablonski and Chaplin [10] used data from specific measurements of the intensity of UV-B radiation incident directly on the Earth’s surface (it became possible only based on long-range measurements from space). They clearly showed a strong correlation between the degree of human skin pigmentation, absolute value of geographical latitude, and the level of UV-B radiation. While a constant high intensity of UV-B radiation with two intensity peaks in spring and fall is observed throughout the year in the equator region, moving further away from the equator results in a pronounced reduction of the radiation intensity with one peak in summer [5]. Ability for temporal increase of skin pigmentation (summer tan) has become an important result of human adaptation to the increase in the intensity of UV radiation in summer in the geographical area within 23-46° latitude. In addition, given sufficient amount of food, people in these latitudes are well provided with vitamin D. This might be the reason for the extremely dense population in these regions of the globe.
SCOTTISH PARADOX?
Over half a billion people live in Europe on a relatively small area far away from the equator. How can such a life in the far north provide people with enough vitamin D? At some point, Gillie drew attention to this special position of Europeans using the example of Scotland [11]. Compared to neighboring England, Scotland faces significantly higher mortality in all segments of the population. At the same time, the risk of serious diseases is also much higher in Scotland. The author suggested that the northern position of the country (55-61°N) and very cloudy maritime climate resulting in insufficient duration of solar radiation are the reasons for this situation. Whereas in the south of England the sun shines for 1500-1900 h per year, in Scotland this parameter is less than 1000 h. This surely affects the level of vitamin D supply for the residents of Scotland.
Gillie et al. discuss in detail the situation in Scotland located at latitude above 50°N [12, 13]. Even in Spain (~40° latitude), the winter season when vitamin D synthesis in human skin is completely absent due to weak UV radiation lasts for about three months. And in Scotland, such “solar starvation” lasts six months. In addition, high mortality rate observed in Scotland cannot be explained only by such factors as smoking, alcohol abuse, poor nutrition, or poverty [14].
Chaplin and Jablonski [15] described the situation in Scotland as “the vitamin D compromise”. When people migrated to Scotland at the end of the last Ice Age (~14,000 years ago), two factors became crucial: maximally depigmented skin (which contributed to a better vitamin D synthesis) and high-calorie diet rich in vitamin D (especially fatty fish). Then the living conditions changed. The Scots were engaged in farming, which dramatically reduced vitamin D supply. Over the last 200 years, living conditions have deteriorated even further due to urbanization and industrialization.
The position of Scotland is extremely unfavorable from the perspective of ensuring direct vitamin D supply by UV-B radiation. However, compared to certain other European regions, its geographical position is not extreme. Scandinavian countries are mostly located further north. Even St. Petersburg in Russia is located at 60° latitude, i.e. practically at the same level as the southern regions of Sweden and Norway, the northern border of Scotland, and the southern part of Greenland. Taking into account the climatic conditions, the situation in Scotland is extremely unfavorable for human life, but everything which has been said about this country applies to a lesser or greater degree to almost all of Europe [16, 17].
WAS VITAMIN D SUPPLY SUFFICIENT FOR VIKINGS IN GREENLAND?
How could the Vikings live for almost 500 years so far in the north when even in Scotland living conditions were so unfavorable for humans due to insufficient vitamin D supply? Many people sailed to Greenland from Scandinavia and the northern islands of Scotland. What do we know about their way of life?
Systematic archeological research started in Greenland in 1921. Paul Norlund, the first supervisor of this work, put together a detailed description of all the data obtained by that time [18]. Careful study of his book reveals a fairly clear picture of the life of the Vikings in Greenland.
Erik the Red (Eirikr enn raudi) gathered in Iceland ~700 people; they sailed to Greenland on 25 ships. Of these, 14 ships with 400 settlers and livestock on board reached the destination in 985 or 986. Norlund believed that a total of about 3000 people lived in Greenland (other sources say up to 6000). In the early days of colonization, the population mainly increased due to resettlement of people from Iceland, Norway, and northern Scottish islands.
This small population of Vikings lived mainly in two centers, Eystribygd and Vestribygd (western and eastern settlements, the first at ~60°N, and the second at ~64°N). The distance between them was almost 300 km. Within the settlement, the distance between the outer houses was measured in tens of kilometers. Even the yards located close to each other were often separated by high ridges, i.e. they were accessible only through the fjords cut deep into the island. Up to 20-30 people lived in each household. Vikings lived in Greenland for ~450 years. About 15 generations changed during that time. They were marrying mainly within the same household or, in the best case, within the settlement. Over the years, inbreeding became inevitable. Let us recall the attributes that appear in the offspring due to inbreeding. In the case of birds and mammals, significant reduction of offspring weight, survival rate, reproduction, and resistance to diseases and stressful environmental conditions are observed [19-22]. Negative effects on survival rate and fecundity are also observed in plants [23-25].
What was happening to the Vikings during the change of their generations in Greenland? The estate of Erik the Red, Brattahlíð, was the most significant in the eastern settlement. Even the remains of a chapel were found there. One hundred forty four skeletons, among them 65 men, were unearthed in the area around the chapel. These men had lived for at least 40-60 years. Many of them were taller than 1.80 m. This discovery helped to understand why such tall men had called the people from the Dorset Culture II, and later also Inuit Skraelings (i.e. scallywag). According to genetic rules, the Vikings were hardly changing during the first few generations. However, later the effects of inbreeding began to manifest with increasing rate.
Skeletons unearthed at the cemetery in Herjolfsnes date back to the late XIV-early XV century. They belong to the people from the last generations of Vikings in Greenland. It was found that men were 1.55-1.70 m tall, and the average height of women was only 1.40 m. Judging from the number of graves, high infant mortality and short lifespan were characteristic of that time. Moreover, the study of skulls showed that the brains of these last Vikings were surprisingly small. Thus, Paul Norlund stated: “Powerless handful of individuals debilitated by diseases, scrubby and ugly, that’s all that was left of them”.
Another biological factor was also essential to the disappearance of the Vikings in Greenland. The Vikings were mainly farmers, and unlike the aboriginal inhabitants of the extreme northern regions, they did not have a sufficient amount of marine animal meat or oily fish (salmon, cod, etc.) in their diet. Relatively small amounts of vitamin D, which they obtained from food, could not compensate for the lack of UV-radiation at northern latitudes. Although it is no longer possible to calculate the degree of vitamin D deficiency, their unearthed skeletons provide compelling evidence of the significant lack of this compound. Spine curvature, deformation of arms and legs, narrowed pelvis, rickets, and osteomalacia that is manifested in adults in the form of bone deformation, pathological fractures, hypotension, and muscle hypotrophy were found in many skeletons. Such bone softening develops in women in the pelvis, lumbar spine, and upper femur. Women with such pelvis pathology were destined to die in childbirth. Paul Norlund provides numerous data in support of those changes in his book [18]. Other studies also discuss the wide spread of rickets among the Vikings in Greenland [26, 27]. Thus, we can assume that the lack of vitamin D as well as inbreeding played an important role in the reduction of the Viking population.
ENSURING PEOPLE WITH VITAMIN D IN NATURAL CONDITIONS AND EFFECT
OF CIVILIZATIONAL FACTORS ON IT
Homo sapiens are known to have originated near the equator, that is, under high intensity of UV-radiation. Strong melatonin pigmentation of skin protected them against the harmful effects of high doses of UV-radiation. However, radiation reaching the skin epidermis was more than enough to provide them with vitamin D [28]. It was proven that the higher the latitude of human habitat, the lower is skin pigmentation [5, 10]. This adaptation to the low intensity of UV-radiation resulted from the long time of human migration to new regions from the equator to northern Europe. Many studies have shown the dependency between the vitamin D level in human blood and latitude of their habitat [29-31]. However, recently published results indicate that data expected based on such ideas do not correlate with the results obtained by measuring vitamin D level in the regions of various latitudes [4]. What are the reasons for this?
Geographical features. Intensity of UV-radiation depends not only on geographical latitude, but also on the altitude above the sea level [8]. In addition, the composition of the atmosphere (e.g. clouds, fog, pollution) also affect the absorption of UV-radiation [11].
Civilization and religion. This includes primarily the presence of clothes. Humans started to cover their bodies with clothes for a substantial part of the year. They began to build protective facilities for greater comfort, and it resulted in a significant body deprivation of solar radiation [1]. On the other hand, some people should avoid solar radiation due to certain diseases [32] or due to poor physical condition [33]. However, even in case of good solar radiation, for example, in Arab countries, many people, and especially women, cover their entire bodies with clothing because of religious canons, which leads to the acute lack of vitamin D [34]. The same applies to ultra-orthodox Jewish culture [35].
Migration of people. In the course of evolution, people migrated to new regions. This was happening over relatively long periods. To some extent, people had enough time to adapt. This applies in particular to the sufficient vitamin D supply due to a sharp decrease in skin pigmentation (see above). But the development of effective transportation systems has enabled the fast relocation of people (even large masses), which greatly facilitates migration. As a result, the number of people living in environmental mismatch dramatically increases. Many people from the southern regions move (or were forcibly relocated in the past) to the northern regions: black Africans – to America, Mexicans – to the United States, black Africans and people of Mediterranean countries – to more northern European countries, people from Asia, Central Asia, and Middle East – to the central and northern European countries including Caucasians and Central Asians migrating to the center of European Russia. On the other hand, residents of European countries and North America move temporarily (vacation, temporary work) or even permanently to countries with abundant solar radiation.
Martin et al. [36] analyzed data published in 34 medical research reports. It turned out that almost 70% of examined people had vitamin D deficiency (9562 out of 13,974 people). Vitamin D deficiency was particularly common among people with a dark skin type, in particular, in immigrants from the regions of the enlarged Middle East. Another study has shown that in the case of the European population, vitamin D deficiency was found in 37% of people, whereas among the residents of elderly homes and immigrants of non-European origin, this figure rises to 80% [37]. The authors studied the spread of tuberculosis and acute respiratory infections among Europeans. Based on data obtained in randomized controlled studies, they provided evidence of the protective role of vitamin D in both tuberculosis and acute respiratory infections. Therefore, they recommend daily intake of 1000 IU of vitamin D for high-risk people as an inexpensive way to provide its required level in blood.
The above-listed features are all affecting the measured vitamin D levels and can account for significant deviations from the values that we expect based on geographical position [4]. However, vitamin D deficiency in the population is a global problem [7, 38]. High-risk groups may vary in different regions. For example, Europe with its relatively large population and significant influx of refugees from sunnier countries is obviously different from all the other regions, but the official health authorities of the European Union and individual countries currently do not consider this in their policies.
REFERENCES
1.Göring, H., and Koshuchowa, S. (2015) Vitamin
D – the sun hormone. Life in environmental mismatch,
Biochemistry (Moscow), 80, 14-28.
2.Villmoare, B., Kimbel, W. H., Seyoum, C.,
Campisano, C. J., DiMaggio, E. N., Rowan, J., Braun, D. R., Arrowsmith,
J. R., and Reed, K. E. (2015) Early homo at 2.8 Ma from Ledi-Geraru,
Afar, Ethiopia, Science, 347, 1352-1355.
3.Kummu, M., and Varis, O. (2011) The world by
latitudes: A global analysis of human population, development level and
environment across the north-south axis over the past half century,
Appl. Geogr., 31, 495-507.
4.Grigalavicius, M., Juzeniene, A., Baturaite, Z.,
Dahlback, A., and Moan, J. (2013) Biologically efficient solar
radiation: vitamin D production and induction of cutaneous malignant
melanoma, Dermatoendocrinology, 5, 150-158.
5.Jablonski, N. G., and Chaplin, G. (2010) Human skin
pigmentation as an adaption to UV radiation, Proc. Natl. Acad. Sci.
USA, 107, 8962-8968.
6.Jablonski, N. G., and Chaplin, G. (2012) Human skin
pigmentation, migration and disease susceptibility, Philos. Trans. R
Soc. Lond. B Biol. Sci., 367, 785-792.
7.Holick, M. F. (2013) Vitamin D and health:
evolution, biologic functions, and recommendeddietary intakes for
vitamin D, in Vitamin D Physiology, Molecular Biology,
and Clinical Applications (Holick, M. F., ed.) Humana Press Inc,
New York, pp. 3-33.
8.Holick, M. F., Chen, T. C., Lu, Z., and Sauter, E.
(2007) Vitamin D and skin physiology: a D-lightful story, J. Bone
Miner Res., 22, Suppl. 2, V28-33.
9.Webb, A. R., Kline, L., and Holick, M. F. (1988)
Influence of saison and latitudeon the cutaneous synthesis of vitamin
D₃: exposure to winter sunlight in Boston and Edmonton will not
promote vitamin D₃ synthesis in human skin, J. Clin.
Endocrinol. Metab., 67, 373-378.
10.Jablonski, N. G., and Chaplin, G. (2000) The
evolution of human skin coloration, J. Hum. Evol., 39,
57-106.
11.Gillie, O. (2012) The Scot’s paradox: can
sun exposure, or lack of it, explain major paradoxes in epidemiology?
Anticancer Res., 32, 237-248.
12.Gillie, O. (2010) Sunlight robbery: A critique of
public health policy on vitamin D in the UK, Mol. Nutr. Food
Res., 54, 1148-1163.
13.Gillie, O. (2016) Controlled trials of vitamin D,
causality and type 2 statistical error, Public Health Nutr.,
19, 409-414.
14.Hanlon, P., Lawder, R., and Buchanan, D. (2005)
Why is mortality higher in Scotland than in England and Wales?
Decreasing influence of socioeconomic deprivation between 1981 and 2001
supports the existence of the “Scottish effect”, J.
Public Health (Oxford), 27, 199-204.
15.Chaplin, G., and Jablonski, N. G. (2013) The
human environment and the vitamin D compromise: Scotland as a case
study in human biocultural adaptation and disease susceptibility,
Hum. Biol., 85, 529-552.
16.Pludowski, P., Karczmarewicz, E., Bayer, M.,
Carter, G., Chlebna-Sokol, D., Czech-Kowalska, J., Debski, R., Decsi,
T., Dobrzanska, A., Franek, E., Gluszko, P., Grant, W. B., Holick, M.
F., Yankovskaya, L., Konstantynowicz, J., Ksiazyk, J. B.,
Ksiezopolska-Orlowska, K., Lewinski, A., Litwin, M., Lohner, S.,
Lorenc, R. S., Lukaszkiewicz, J., Marcinowska-Suchowierska, E.,
Milewicz, A., Misiorowski, W., Nowicki, M., Povoroznyuk, V., Rozentryt,
P., Rudenka, E., Shoenfeld, Y., Socha, P., Solnica, B., Szalecki, M.,
Talalaj, M., Varbiro, S., and Zmijewski, M. A. (2013) Practical
guidelines for the supplementation of vitamin D and the treatment of
deficits in Central Europe – recommended vitamin D intakes
in the general population and groups at risk of vitamin D deficiency,
Endokrynol. Pol., 64, 319-327.
17.Pludowski, P., Grant, W. B., Bhattoa, H. P.,
Bayer, M., Povoroznyuk, V., Rudenka, E., Ramanau, H., Varbiro, S.,
Rudenka, A., Karczmarewicz, E., Lorenc, R., Czech-Kowalska, J., and
Konstantynowicz, J. (2014) Vitamin D status in Central Europe, Int.
J. Endocrinol., 589587.
18.Norlund, P. (1937) Wikingersiedlungen in
Grönland. Ihre Entstehung und Ihr Schicksal, Curt Kabitzsch
Verlag, Leipzig.
19.Keller, L., and Waller, D. (2002) Inbreeding
effects in wild population, Trends Ecol. Evol., 17,
230-241.
20.Acevedo-Whitehouse, K., Gulland, F., Greig, D.,
and Amos, W. (2003) Inbreeding: disease susceptibility in California
sea lions, Nature, 422, 35.
21.Mc Parland, S., Kearney, J. F., Rath, M., and
Berry, D. P. (2007) Inbreeding effects on milk production, calving
performance, fertility, and conformation in Irish Holstein-Friesians,
J. Dairy Sci., 90, 4411-4419.
22.Huisman, J., Kruuk, L., Ellis, P., Clutton-Brock,
T. H., and Pemberton, J. M. (2016) Inbreeding depression across the
lifespan in a wild mammal population, Proc. Natl. Acad. Sci.
USA, 113, 3585-3590.
23.Göring, H., and Mitchenkova, T. A. (1961)
Einige Fragen der Physiologie von Maispflanzen, die eine
unterschiedliche Vitalität besitzen [in Russian], Agrobiologiya
(Moscow), 383-389.
24.Charlesworth, D., and Willis, J. (2009) The
genetics of inbreding depression, Nat. Rev. Genet., 10,
783-796.
25.Göring, H. (1963) Zellteilungsstörungen
nach Selbstung bei Zea mays L., Biol. Rundschau,
1, 41-42.
26.Yuen, A. W. C., and Jablonski, N. G. (2010)
Vitamin D: in the evolution of human skin colour, Med.
Hypotheses, 74, 39-44.
27.Murray, F. G. (1934) Pigmentation, sunlight, and
nutritional disease, Am. Anthropol., 36, 438-445.
28.Chen, T. C. Lu, Z., and Holick, M. F. (2013)
Photobiology of vitamin D, in Vitamin D Physiology, Molecular
Biology, and Clinical Applications (Holick, M. F., ed.) Human Press
Inc., New York, pp. 35-60.
29.Lu, Z., Chen, T. C., and Holick, M. F. (1992)
Influence of saison and timeof day on the synthesis of vitamin
D₃, in Biological Effects of Light (Holick, M. F., and
Kligman, A. M., eds.) Walter de Gruyter, Berlin-New York, pp.
57-61.
30.Mithal, A., Wahl, D. A., Bonjour, J. P.,
Burckhardt, P., Dawson-Hughes, B., Eisman, J. A., Fulican, G. E., Joss,
R. G., Lips, P., and Morales-Torres, J. (2009) Global vitamin D status
anbd determinants of hypovitaminosis D, Osteoporos Int.,
20, 1807-1820.
31.Brickley, M. B., Moffat, T., and Watamaniuk, L.
(2014) Biocultural perspectives of vitamin D deficiency in the past,
J. Anthropol. Archaeol., 36, 48-59.
32.Bogaczewicz, J., Karczmarewicz, E., Pludowski,
P., Zabek, J., and Wozniacka, A. (2016) Requirement for vitamin D
supplementation in patients using photoprotection: variations in
vitamin D levels and bone formation markers, Int. J. Dermatol.,
55, 176-183.
33.Grant, W. B., Wimalawansa, S. J., Holick, M. F.,
Cannell, J. J., Pludowski, P., Lappe, J. M., Pittaway, M., and May, P.
(2015) Emphasizing the health benefits of vitamin D for those with
neurodevelopmental disorders and intellectual disabilities,
Nutrients, 7, 1538-1564.
34.Buyukuslu, N., Esin, K., Hizli, H., Sunal, N.,
Yigit, P., and Garipagaoglu, M. (2014) Clothing preference affects
vitamin D status of young women, Nutr. Res., 34,
688-693.
35.Tsur, A., Metzger, M., and Dresner-Pollak, R.
(2011) Effect of different dress style on vitamin D level in healthy
young Orthodox and ultra-Orthodox students in Israel, Osteoporos.
Int., 22, 2895-2898.
36.Martin, C. A., Gowda, U., and Renzaho, A. M.
(2016) The prevalence of vitamin D deficiency among dark-skinned
populations according to their stage of migration and region of birth:
a meta-analysis, Nutrition, 32, 21-32.
37.Zittermann, A., Pilz, S., Hoffmann, H., and Marz,
W. (2016) Vitamin D and airway infections: a European perspective,
Eur. J. Med. Res., 21, 14.
38.Rautiainen, S., Manson, J. E., Lichtenstein, A.
H., and Sesso, H. D. (2016) Dietary supplements and disease
prevention – a global overview, Nat. Rev.
Endocrinol., 12, 407-420.
39.Rubel, F., and Kottek, M. (2010) Observed and
projected climate shifts 1901-2100 depicted by world maps of the
Köppen–Geiger climate classification, Meteorol.
Zeit., 19, 135-141.
40.Klein Goldewijk, K., Beusen, A., and Janssen, P.
(2010) Long-term dynamic modeling of global population and built-up
area in a spatially explicit way: HYDE 3.1, Holocene, 20,
565-573.