Alterations in Tear Biochemistry Associated with Postanesthetic Chronic Dry Eye Syndrome
E. Yu. Zernii1*, M. O. Golovastova1, V. E. Baksheeva1, E. I. Kabanova2, I. E. Ishutina2, O. S. Gancharova1,3, A. E. Gusev4, M. S. Savchenko1, A. P. Loboda1, L. F. Sotnikova2, A. A. Zamyatnin, Jr.1,5, P. P. Philippov1, and I. I. Senin1*
1Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, 119991 Moscow, Russia; E-mail: zerni@belozersky.msu.ru, senin@belozersky.msu.ru, zerni@mail.ru2Skryabin Moscow State Academy of Veterinary Medicine and Biotechnology, Department of Biology and Pathology Domestic, Laboratory and Exotic Animals, 109472 Moscow, Russia
3Lomonosov Moscow State University, Institute of Mitoengineering, Pathology Department, 119991 Moscow, Russia
4Principle Clinical Military Hospital of the Russian Federation, 143040 Golitsyno, Moscow Region, Russia
5Sechenov First Moscow State Medical University, Institute of Molecular Medicine, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received July 27, 2016; Revision received September 1, 2016
Perioperative dry eye syndrome (DES) is a common ocular complication of long-term general anesthesia. Chronic DES can lead to permanent damage to the cornea and disturbance of visual function, up to total loss of vision. Here, a relationship between the duration of general anesthesia and the risk of chronic DES in patients was demonstrated. Using an experimental model of perioperative corneal abrasions in rabbits, it was found that introduction of animals to 3-h general anesthesia resulted in clinically significant chronic damage to the cornea in 50% of cases. The development of the complication was not associated with irreversible or long-term impairment of tear secretion, but it was accompanied by a decrease in tear film stability and growth of the total protein content as well as decrease in total antioxidant activity of the tear induced by low molecular weight antioxidants. In addition, anesthesia-induced changes in activity of tear antioxidant enzymes including superoxide dismutase and enzymes providing homeostasis of reduced glutathione (glutathione peroxidase, glutathione-S-transferase, glutathione reductase) were observed. All these alterations were protracted (up to 1-2 weeks) and therefore might account for transition of the perioperative DES into the chronic form. These findings can be useful in the development of novel approaches for the prevention and treatment of chronic forms of DES in the postanesthetic period.
KEY WORDS: general anesthesia, dry eye syndrome, corneal abrasion, tear secretion, tear film stability, tear proteins, tear antioxidant activity, superoxide dismutase, glutathione peroxidase, glutathione-S-transferase, glutathione reductaseDOI: 10.1134/S0006297916120166
Abbreviations: AOA, antioxidant activity; DES, dry eye syndrome; GPO, glutathione peroxidase; GR, glutathione reductase; GST, glutathione-SH-transferase; ROS, reactive oxygen species; SOD, superoxide dismutase.
Perioperative dry eye syndrome (DES or keratoconjunctivitis
sicca) is a common ophthalmological complication of long-term
general anesthesia performed for diminishing pain syndrome during
invasive procedures [1-6]. DES
is generally defined as a complex of traits typical for clinically
apparent or latent corneo-conjunctival xerosis associated with
destabilization of the tear film that normally protects the corneal
surface [7, 8]. As the cornea
is the most densely innervated body tissue, its injury is associated
with onset of a whole spectrum of symptoms: from foreign body sensation
to ophthalmalgia, photophobia, and blurred vision. So far, decreased
tear production caused by suppression of parasympathetic innervation of
the lacrimal gland in the presence of anesthetics was considered as the
main pathogenic factor of perioperative DES [9-11]. This disorder can result in weakening of tear
film protective qualities, manifesting in development of focal lesions
(abrasions) of the cornea. Yet, recent studies involving an animal
perioperative DES model demonstrated that pathological processes within
the course of anesthesia include not only lowered tear secretion, but
also alterations in tear biochemistry [12].
Indeed, during anesthesia tear film stability declines, possibly due to
the depressed production of phospholipids, necessary for maintaining
the integrity of its outer layer and prevention of tear evaporation [7]. Furthermore, under these conditions, the proteomic
composition of tears also changes, and its antioxidant activity becomes
depressed, which may lead to local inflammatory response (Zernii et
al., unpublished data).
Since regeneration of the corneal epithelium normally takes 24-72 h, small perioperative abrasions generally do not require treatment. Nevertheless, some patients develop progressing DES with associated symptoms becoming apparent in a few days or even weeks post-surgery, and these are not uniformly interpreted as being due to general anesthesia. Degenerative changes in the cornea were reported to display chronicity in approximately 16% of cases [13]. Chronic DES mechanisms, along with contribution of different perioperative factors (time of anesthesia, patient positioning, surgery type, etc.) to the risk of its development, remain unclear. In the meantime, of all variants of DES, the chronic form may result in irreversible damage to the cornea, deterioration of visual function, and ultimately complete vision loss. Patients are now subjected to long-term and costly rehabilitation courses to avoid these complications [13]. This situation calls for the development of more effective approaches to prevention and/or timely correction of perioperative corneal abrasions.
In this study, we assessed the incidence of chronic DES in a representative group of surgery patients and characterized the correlation between duration of anesthesia and risks of DES. Using experimental animals (rabbits), we developed a model of corneal abrasions induced by anesthesia the duration of which corresponded to elevated DES risks in patients. We monitored clinical status of the cornea, tear secretion, as well as stability and biochemical properties of the tear film during a long-term (up to 30 days) postanesthetic period. Our data are the first to assess the contributions of different pathogenic factors to chronic anesthesia-induced corneal injury. The results of this study could aid in designing novel approaches to prevention and treatment of chronic DES in the postanesthetic period.
MATERIALS AND METHODS
To determine relationships between duration of general anesthesia and risk of chronic DES, the clinical status of the cornea was surveyed in a group of 20 surgery patients consisting of 10 male (age 30-59) and 10 female (age 35-55) individuals. The survey was conducted consistently with Good Clinical Practice standards, Helsinki declaration, and regulatory protocols of the Russian Federation. Every patient provided a letter of informed consent. Patients were included into the test group because of having spent at least 100 min under general anesthesia within the course of non-ophthalmic surgery (herniotomy, cholecystectomy, thyroidectomy, salpingectomy, adnexectomy, hysterectomy, mastectomy, and lumbal discectomy). Among the exclusion criteria were symptoms of DES and tear production disorders in the preoperative period, as well as eye surgery, neurosurgery, and ENT surgery on the patients’ clinical record. All patients were subjected to ophthalmological inspection via fluorescein test [14], performed immediately prior to the surgery and on the 3rd and 5th days after the surgery. Estimation of test results was carried out using a slit lamp with blue filter. The registered corneal injuries were assigned clinical scores (CS) of 0-3 depending on the size of the affected corneal surface: <15 point lesions (CS = 1); >15 point lesions or one larger confluent lesion (CS = 2); >15 point lesions and one confluent lesion (CS = 3).
For the experimental model of chronic DES, 22 healthy pigmented rabbits (2-3 months old, weighting 2.3-3.0 kg) were purchased from a certified farm (KrolInfo, Moscow Region). Prior to experiments, the animals were housed at a 12 h light-dark cycle at temperature of 22-25°C and humidity of 55-60% with free access to food and water. The animals were treated according to the 8th edition of Guide for the Care and Use of Laboratory Animals of the National Research Council and Statement for the Use of Animals in Ophthalmic and Visual Research of the Association for Research in Vision and Ophthalmology (ARVO). The protocol was approved by the Belozersky Institute of Physico-Chemical Biology Animal Care and Use Committee (Protocol No. 1/2016).
Chronic anesthesia-associated DES was induced in the experimental group of 16 animals (group 1). The animals were placed in a constraining device and subjected to general anesthesia by intramuscular injection of anesthetic preparation containing 50 mg/ml tiletamine and 50 mg/ml zolazepam in a dose of 16-24 mg/kg of body weight. Additional injections were performed in the course of anesthesia to uphold continuous narcotic sleep for 3 h (mean duration of anesthesia in experimental groups 1 and 2 (see below) was 183 ± 12 min). This time span was chosen considering the correlations between duration of anesthesia and risk of DES development, revealed in the patients’ survey (see below). Development of corneal abrasions in experimental animals was monitored immediately prior to anesthesia and after 1, 3, and 7 days. In the course of the experiment, the animals were kept under normal conditions described above. Corneal lesions were identified via the fluorescein test [14]. To this end, the animals were given 2 µl instillations (under the lower eyelid) of 1% sodium fluorescein, which was then distributed over the cornea by 2-3 movements imitating eye blinking. Test results were assessed using a slit lamp with blue filter. The grade of corneal lesions were assigned clinical scores (CS) of 0-4 depending on the size of the stained surface (fluorescein scale [15]): no staining (CS = 0), 0-12.5% of corneal surface (CS = 1), 12.5-25% of corneal surface (CS = 2), 25-50% of corneal surface (CS = 3), more than 50% of corneal surface (CS = 4).
Tear secretion was evaluated by the standardized Schirmer’s test [16] in experimental group 1 (16 animals) immediately prior to and after the end of 3-h general anesthesia, as well as after 1, 3, 7, 14, and 30 days of the postanesthetic period. Within the specified time limits, the animals were kept under normal conditions described above. Gauged Schirmer’s test strips were placed beneath the lower eyelid for 5 min, and then the length of the moistened surface (in mm) was recorded.
Tear film stability was assessed by the tear break-up time (BUT) test [17] in experimental group 1 (16 rabbits) prior to, immediately after, and 1, 3, 7, 14, and 30 days after their exposure to general anesthesia. Fluorescein was instilled intraconjunctivally, as described above, and the time until the appearance of the first stained spot in the center of the cornea was measured. Staining was detected by a slit lamp with blue filter.
Total protein concentration was measured in tear samples of animals from experimental group 2 (six rabbits) that were subjected to the same anesthetic conditions as group 1. The samples were collected, using gauged Schirmer’s test strips, immediately prior to and after the anesthesia, and after the consecutive 1 h, 1 day, 3 days, and 7 days. During these periods, the animals were kept under normal conditions (see above). Tear samples were extracted from 15-mm test strips during 30 min with 150 µl of PBS. Protein concentration in the resulting extract was measured by the bicinchoninic acid (BCA) method using a BCA Protein Assay Kit (Thermo Fisher Scientific, USA) in accordance with the manufacturer’s instructions.
Total antioxidant activity (AOA) of tears was evaluated in the same tear samples that were used for total protein measurements (group 2, six rabbits). To this end, the hemoglobin–H2O2–luminol test system [18] was applied. A calibration curve was generated using standard solutions of Trolox (1, 2, 4, 5, 8 µM) in PBS. A 30-µl sample of the standard or tear extract (dissolved 4-fold in PBS) was added to 440 µl of reaction mixture containing 0.01 mM luminol and 0.5 mM hemoglobin in PBS. Luminol oxidation was triggered by adding H2O2 to the final concentration of 6 µM. Chemiluminescence was recorded every 1 s for 10 min by a Glomax-Multi multimode reader (Promega, USA). The data were analyzed using SigmaPlot 11 software (SYSTAT Software).
Activity of tear enzymes involved in antioxidant protection (superoxide dismutase, glutathione peroxidase, glutathione reductase, and glutathione-SH-transferase) was determined in the same tear samples that were used for total protein measurements (group 2, six rabbits) using commercially available kits (Sigma-Aldrich, USA) in accordance with the manufacturer’s instructions. Intensity of colorimetric reactions was determined with an Ultrospec 1000 spectrophotometer (Pharmacia Biotech, UK) and MR-96A plate reader (Mindray, USA). The data were analyzed using SigmaPlot 11 software (SYSTAT Software).
RESULTS AND DISCUSSION
Correlation between duration of general anesthesia and risk of chronic DES. To determine the incidence of chronic DES during the postanesthetic period, we assessed the corneal status of patients after different surgical interventions in a representative group of 20 individuals. Previously, it was demonstrated that the first signs of corneal damage become noticeable after at least 1.5-2.0 h of the exposure to general anesthesia [3, 10]. Thus, patients were included in the test group on the primary condition of spending more than 100 min under narcosis. The perioperative DES symptoms were diagnosed by means of ophthalmological survey based on the fluorescein test. DES is generally considered chronic when corneal abrasions linger for as long as 3 days after the end of anesthesia [13]. Based on this condition, ophthalmological inspection of patients was carried out prior to the operation and on the 3rd and 5th days of the postoperative period. Of 20 patients, five demonstrated positive fluorescein staining within the specified time limits (Table 1).
Table 1. Severity scores of chronic corneal
lesions in susceptible surgery patients subjected to different times of
general anesthesia
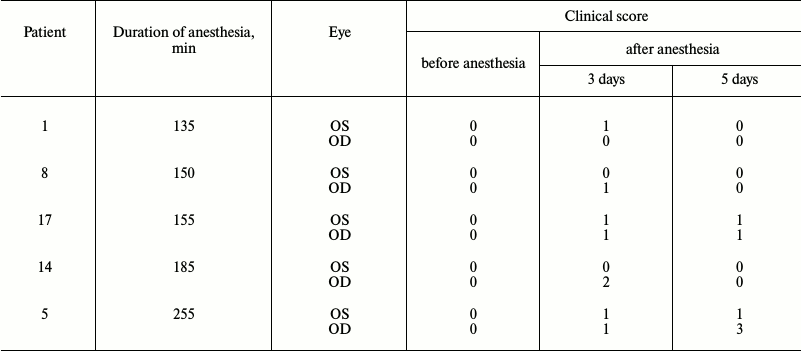
According to our results, chronic DES incidence in the test group reached 25%, in agreement with previous estimations [13]. Remarkably, anesthetic conditions associated with corneal abrasions in some patients had no visible effect on the others, indicating different levels of patient susceptibility to perioperative DES. The correlation between duration of anesthesia and severity of corneal damage was observed among patients with the aforementioned complication. Thus, patients exposed to 135-150-min-long anesthesia displayed first-degree abrasions in one eye, which completely regenerated after 5 days. However, in cases when the duration of the narcosis exceeded 150 min, corneal damage was more prominent, manifesting in both eyes, and in one case even progressing with time. Combined with existing data [3, 10], these results indicate that the risk of chronic DES development increases with prolonged duration of narcosis within the 2-4 h range. Therefore, it was decided to use 3-h anesthesia in our chronic animal DES model, and to study the alterations in biochemical properties of tears associated with these conditions.
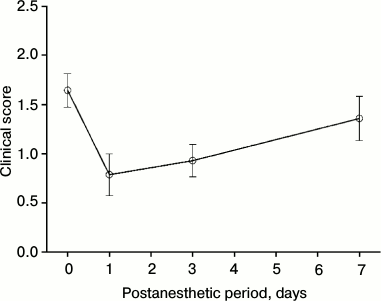
Modeling of chronic DES associated with general anesthesia in experimental animals. In previous studies, it was noted that development of corneal abrasions under effect of general anesthesia is accompanied by changes in secretion, stability, and biochemical composition of tears [1-6, 12]. Monitoring these parameters is crucial for studying chronic perioperative DES due to their potential diagnostic value. Moreover, it might promote further understanding of mechanisms of the complication, which is necessary to design novel approaches to its prevention and treatment. However, this kind of research is complicated by limited availability of patients for ophthalmological tests during prolonged postoperative period. With that in mind, studies of long-term alterations in tear film composition and stability after application of general anesthesia were performed using our previously developed DES model system [12] adapted to the chronic course of the disease. Rabbits (Oryctolagus cuniculus) were chosen as model animals for simulation of anesthesia-induced corneal abrasions. This choice was particularly supported by the fact that the species’ visual sensory system possesses many common anatomical, biomechanical, and biochemical features with the human eye [19]. Sixteen animals from experimental group 1 were subjected to general anesthesia for 3 h. Their corneal status was surveyed immediately after awakening and during the following week. As demonstrated by the fluorescein test results, 3 h anesthesia induced corneal abrasions in all subjects. In the meantime, 13 of 16 animals sustained corneal damage during the first 24 h (19 eyes, 59%), 11 animals – during first 72 h (18 eyes, 56%), 8 animals – during first week (13 eyes, 40%) after the anesthesia (Table 2). For the latter group, progression of corneal injury over time was assessed. We found that while the tissue underwent partial regeneration during the first 24 h, this remission was followed by further aggravation of the disease (Fig. 1). Indeed, in half of the subjects (40% of the eyes) DES was demonstrated to take a long-term chronic course, with third degree corneal abrasions observed as early as after one week of the postanesthetic period (Table 2). In summary, according to the data, the species of choice (rabbit) displays pronounced susceptibility to chronic anesthesia-induced corneal abrasions, and, therefore provides a convenient model for studying various pathogenic factors underlying chronic DES.
Table 2. Severity scores of acute and
chronic corneal abrasions in experimental animals (rabbits), subjected
to 3 h of general anesthesia
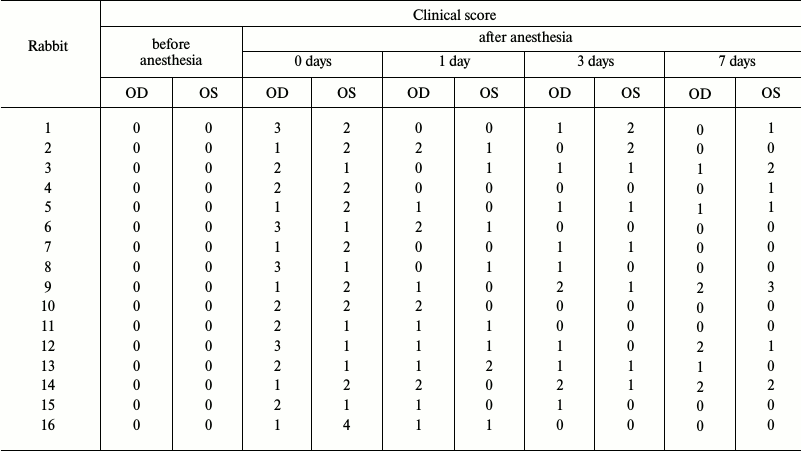
Fig. 1. Clinical course of chronic corneal abrasions in a group of eight animals (rabbits No. 1, 3, 4, 5, 9, 12, 13, and 14 from experimental group 1) during the first 7 days after their exposure to 3-h general anesthesia.
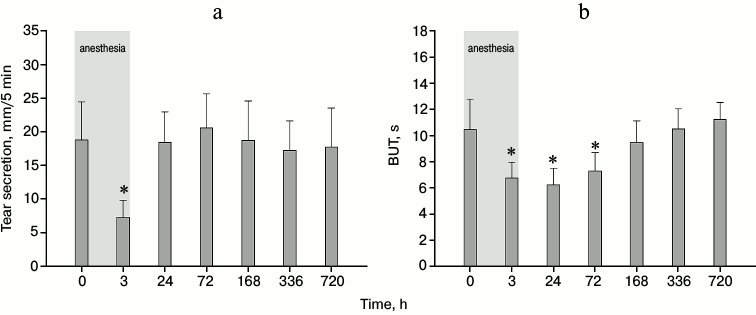
Monitoring of tear secretion and tear film stability in experimental animals with general anesthesia-induced chronic DES. As stated above, until recently, low tear secretion associated with the suppressive effect of anesthetics on lacrimal gland activity was considered the main pathogenic factor underlying perioperative DES. To estimate the contribution of this factor to the risk of development of chronic DES, we measured tear secretion in animals from experimental group 1 (subjected to 3-h general anesthesia) during 30 days of the postanesthetic period. Using the standardized Schirmer’s test, secreted tear volume was found to decrease 3-fold immediately after the recovery from narcosis (Fig. 2a). This result agrees well with existing observations that anesthesia effectively suppresses tear production [9-11]. Nevertheless, tear secretion rate completely recovered during the first 24 h and maintained normal levels during the next 29 days. Based on these observations, it can be suggested that chronic corneal abrasions detected in half of the animals after 5-7 days (Table 1) are associated with changes in quality, rather than quantity, of the secreted tears. Indeed, the tear break-up time (BUT) test performed on the same animals revealed an alternate curve of tear film stability during the same period (Fig. 2b). Namely, this parameter hit its lowest point after the first 24 h of the postanesthetic period, while the recovery was delayed, not reaching normal levels until 7 days later. In conclusion, general anesthesia in animals does not cause long-term or irreversible tear production failure, but it promotes tear film destabilization and, consequentially, decline of its protective qualities. This could be one of the major factors contributing to chronic DES development.
Fig. 2. Tear secretion (a) and tear film stability (b) in rabbits from experimental group 1 under the effect of 3-h general anesthesia (highlighted in gray) and on the 1st, 3rd, 7th, 14th, and 30th day of the postanesthetic period (*p < 0.05 in comparison with the values obtained before the anesthesia).
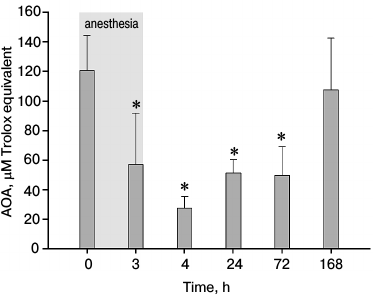
Study of total antioxidant activity of tears in experimental animals with general anesthesia-induced chronic DES. The observed negative effect of general anesthesia on tear film stability could be associated with anesthesia-induced alterations in the biochemical composition of tears. For instance, the decline of tear phospholipid content is known to result in degeneration of the outer layer of the tear film, thereby enhancing its evaporation [7]. The major biochemical component of tears is a set of low molecular weight antioxidants, including cysteine, glutathione, tyrosine, ascorbates, and urates [20, 21]. The chemical properties of these compounds make a strong contribution to total antioxidant activity (AOA) of tears, protecting corneal epithelium from negative impact of oxidative stress [21]. Since the loss of this protection followed by increased harmful activity of reactive oxygen species (ROS) is one of the confirmed pathogenic factors affecting DES, we characterized changes in tear AOA during the anesthesia-induced chronic variant of the disease. Animals from experimental group 2 (6 total) were subjected to 180 min general anesthesia, followed by 2 weeks of tear sample collecting and AOA measurements. Figure 3 shows the results of this survey during the first week. As was observed, AOA decreases 2-fold along the course of the anesthesia itself, which correlates with our previous findings ([12], Zernii et al., unpublished data). However, tear AOA of the experimental animals continues to decline for 1-h post-narcosis, whereas the first signs of recovery become visible in 2 to 3 days, and complete restoration takes up to 7 days. In summary, the decrease in concentration/impact of tear antioxidants is one on the first and most dynamic responses to the anesthetic action. This lingering condition might be one of the factors enabling DES to develop chronicity.
Fig. 3. Total antioxidant activity (AOA) in rabbit tear samples obtained from experimental group 2 within 3 h of general anesthesia (highlighted in gray), and after 1 h, 1 day, 3 days, and 7 days of the postanesthetic period (*p < 0.05 in comparison with the values obtained before the anesthesia).
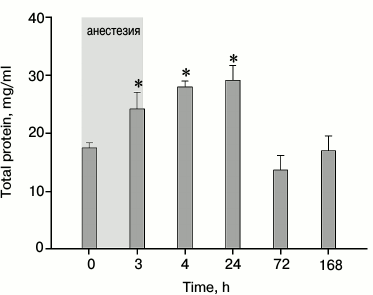
Analysis of tear proteins in experimental animals with general anesthesia-induced chronic DES. Tear proteins secreted by lacrimal and Meibomian glands, conjunctival cells, and the cornea itself are supposed to be involved in protective mechanisms providing defense of the cornea from ROS and other chemical agents, along with antibacterial and antiinflammatory action [7]. To determine the mechanisms of chronic DES and to develop diagnostic and prognosis methods for this disorder, characterization of anesthesia-induced alterations in the tear proteins is required. With this in mind, we studied the changes in total protein content of tears and, in accordance with previous AOA measurements, monitored the activity of antioxidant enzymes in tear samples from experimental group 2 (see previous section). Our data demonstrate that under the effect of anesthesia total protein concentration in tears increased, peaking by the end of the first hour, and proceeded to decline during 3 to 7 days post-narcosis (Fig. 4). The resulting curve is different from the one observed in our recent studies, where protein concentration was shown to normalize within the course of 2-h anesthesia. However, the same studies revealed a proinflammatory response in experimental animals that developed after 3 h from the start of the anesthesia (Zernii et al., unpublished data). It was characterized by upregulation of tear proinflammatory cytokines that are known to enhance permeability of conjunctival blood vessels to serum proteins [22]. As a result, the influx of these proteins from blood to tears could explain the observed increase in total protein content. This suggestion agrees with the fact that proteins such as serum albumin and serotransferrin are major components of the tear proteome ([23], Zernii et al., unpublished data).
Fig. 4. Total protein concentration in rabbit tear samples obtained from experimental group 2 within 3 h of general anesthesia (highlighted in gray), and after 1 h, 1 day, 3 days, and 7 days of the postanesthetic period (*p < 0.05 in comparison with the values obtained before the anesthesia).
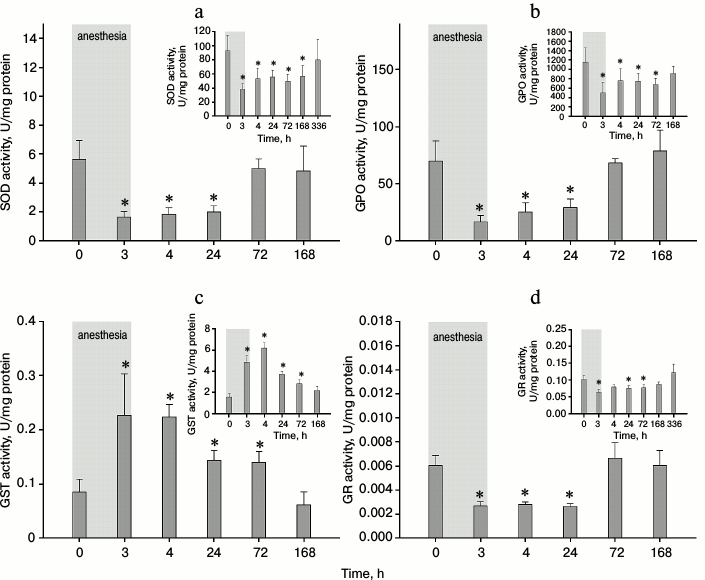
As the growth of total protein concentration in tear samples corresponded to the local AOA decline (Figs. 3 and 4), we suggested that the latter was at least partially associated with the inability of protective enzymes to neutralize ROS and to replenish the pool of low molecular weight antioxidants [24]. To test this hypothesis, we measured the activity of four major enzymes involved in antioxidant protection – superoxide dismutase (SOD), glutathione peroxidase (GPO), glutathione-SH-transferase (GST), and glutathione reductase (GR) – in animal tear samples from experimental group 2. During the experiments, the activity curve for each enzyme was traced within the post-narcosis period. As demonstrated in Fig. 5 (a, b, d), a decrease in SOD, GPO, and GR specific activity was observed within the course of anesthesia and for the next 24 h. This effect did not stem from the overall increase of protein content in tears (Fig. 4) because absolute activity of these enzymes also declined (Fig. 5, a, b, and d, the insets). In contrast with these results, both specific and absolute GST activities elevated within the specified time limits (Fig. 5c). Although the recovery dynamics slightly varied among the analyzed factors, in general all enzymes normalized within one week after anesthesia. In summary, general anesthesia has a noticeable impact on different tear components with protective antioxidant activity. For instance, exposure to general anesthesia in experimental animals hinders the function of SOD, the only antioxidant enzyme in tears [25]. Furthermore, this condition apparently suppresses the activity of reduced glutathione (which plays a major role in tear AOA [26]) and significantly alters the functioning of enzymes involved in its metabolism (GPO, GR, GST). Reduced glutathione diminishes production of hydrogen peroxide in tears, thus inhibiting oxidation of phospholipids, crucial for tear film mechanical integrity [26]. Furthermore, in the presence of increased ROS, glutathione deficit in tears inhibits proliferation of conjunctival cells – especially goblet cells, which are the main source of mucins responsible for tear film stability [27]. Based on these observations, it can be speculated that decreased concentration of reduced glutathione, along with enzymes providing its antioxidant activity (GPO), and regeneration (GR), partially underlies the prolonged depression of tear film stability under general anesthesia (Fig. 2b). As a result, this condition might result in the development of chronic DES.
Fig. 5. Specific and absolute (insets) activity of superoxide dismutase (SOD) (a), glutathione peroxidase (GPO) (b), glutathione-S-transferase (GST) (c), and glutathione reductase (GR) (d) in rabbit tear samples obtained from experimental group 2 within 3 h of general anesthesia (highlighted in gray), and after 1 h, 1 day, 3 days, and 7 days of the postanesthetic period (*p < 0.05 in comparison with the values obtained before the anesthesia).
Current approaches to perioperative DES prevention generally involve artificial tears or ointments that slow tear evaporation, thus assisting in the recovery of its protective function [28-31]. However, such methods display limited effectiveness, since pathogenesis of perioperative DES is complex, and many contributing factors remain unattended by commonly used prophylactic measures [12, 31]. In the light of this research, it seems viable to carry out complex preventive treatment to avoid chronic corneal injury associated with prolonged general anesthesia. This treatment could include administration of agents promoting restoration of tear antioxidant function, e.g. vitamins A, C, E [32-34], as well as novel targeted antioxidants [35-37].
Acknowledgements
Induction and clinical examination of corneal abrasions associated with general anesthesia in rabbits, as well as analysis of tear proteins, total antioxidant activity and activity of antioxidant defense enzymes were performed in Lomonosov Moscow State University with financial support of the Russian Science Foundation (grant No. 16-15-00255).
REFERENCES
1.Terry, H. R., Jr., Kearns, T. P., Love, J. G., and
Orwoll, G. (1965) Untoward ophthalmic and neurologic events of
anesthesia, Surg. Clin. North Am., 45, 927-938.
2.Snow, J. C., Kripke, B. J., Norton, M. L., Chandra,
P., and Woodcome, H. A. (1975) Corneal injuries during general
anesthesia, Anesth. Analg., 54, 465-467.
3.Batra, Y. K., and Bali, I. M. (1977) Corneal
abrasions during general anesthesia, Anesth. Analg., 56,
363-365.
4.Cucchiara, R. F., and Black, S. (1988) Corneal
abrasion during anesthesia and surgery, Anesthesiology,
69, 978-979.
5.Roth, S., Thisted, R. A., Erickson, J. P., Black,
S., and Schreider, B. D. (1996) Eye injuries after nonocular surgery. A
study of 60,965 anesthetics from 1988 to 1992, Anesthesiology,
85, 1020-1027.
6.White, E., and Crosse, M. M. (1998) The aetiology
and prevention of peri-operative corneal abrasions, Anaesthesia,
53, 157-161.
7.Alves, M., Fonseca, E. C., Alves, M. F., Malki, L.
T., Arruda, G. V., Reinach, P. S., and Rocha, E. M. (2013) Dry eye
disease treatment: a systematic review of published trials and a
critical appraisal of therapeutic strategies, Ocul. Surf.,
11, 181-192.
8.Dogru, M., Nakamura, M., Shimazaki, J., and
Tsubota, K. (2013) Changing trends in the treatment of dry-eye disease,
Expert. Opin. Investig. Drugs, 22, 1581-1601.
9.Cross, D. A., and Krupin, T. (1977) Implications of
the effects of general anesthesia on basal tear production, Anesth.
Analg., 56, 35-37.
10.Hrazdirova, V., Navratilova, B., and Ventrubova,
R. (1990) Use of contact lenses during general anesthesia, Cesk.
Oftalmol., 46, 223-229.
11.Krupin, T., Cross, D. A., and Becker, B. (1977)
Decreased basal tear production associated with general anesthesia,
Arch. Ophthalmol., 95, 107-108.
12.Tikhomirova, N. K., Averina, O. A., Chemodanov,
D. V., Ishutina, I. E., Sotnikova, L. F., Neverovsky, A. E., Zernii, E.
Y., Philippov, P. P., and Senin, I. I. (2015) Development of dry eye
syndrome under conditions of general anesthesia, Proc. Int. Sci.
Pract. Conf. “Current problems and advances in
medicine”, Vol. 2, pp. 60-62.
13.Gild, W. M., Posner, K. L., Caplan, R. A., and
Cheney, F. W. (1992) Eye injuries associated with anesthesia. A closed
claims analysis, Anesthesiology, 76, 204-208.
14.Wipperman, J. L., and Dorsch, J. N. (2013)
Evaluation and management of corneal abrasions, Am. Fam.
Physician, 87, 114-120.
15.Lemp, M. A. (1995) Report of the national eye
institute/industry workshop on clinical trials in dry eyes, CLAO
J., 21, 221-232.
16.Bhattacharya, D., Ning, Y., Zhao, F., Stevenson,
W., Chen, R., Zhang, J., and Wang, M. (2015) Tear production after
bilateral main lacrimal gland resection in rabbits, Invest.
Ophthalmol. Vis. Sci., 56, 7774-7783.
17.Gautheron, P. D., Lotti, V. J., and Le Douarec,
J. C. (1979) Tear film breakup time prolonged with unmedicated
cellulose polymer inserts, Arch. Ophthalmol., 97,
1944-1947.
18.Gulidova, O. V., Liubitskii, O. B., Klebanov, G.
I., and Chesnokova, N. B. (1999) Changes in the antioxidative activity
of tears during experimental eye burns, Bull. Exp. Biol. Med.,
128, 571-574.
19.Zernii, E. Y., Baksheeva, V. E., Iomdina, E. N.,
Averina, O. A., Permyakov, S. E., Philippov, P. P., Zamyatnin, A. A.,
and Senin, I. I. (2016) Rabbit models of ocular diseases: new relevance
for classical approaches, CNS Neurol. Disord. Drug Targets,
15, 267-291.
20.Gogia, R., Richer, S. P., and Rose, R. C. (1998)
Tear fluid content of electrochemically active components including
water soluble antioxidants, Curr. Eye Res., 17,
257-263.
21.Choy, C. K., Cho, P., Chung, W. Y., and Benzie,
I. F. (2001) Water-soluble antioxidants in human tears: effect of the
collection method, Invest. Ophthalmol. Vis. Sci., 42,
3130-3134.
22.Aragona, P., Aguennouz, M., Rania, L., Postorino,
E., Sommario, M. S., Roszkowska, A. M., De Pasquale, M. G., Pisani, A.,
and Puzzolo, D. (2015) Matrix metalloproteinase 9 and transglutaminase
2 expression at the ocular surface in patients with different forms of
dry eye disease, Ophthalmology, 122, 62-71.
23.Runstrom, G., Mann, A., and Tighe, B. (2013) The
fall and rise of tear albumin levels: a multifactorial phenomenon,
Ocul. Surf., 11, 165-180.
24.Chen, Y., Mehta, G., and Vasiliou, V. (2009)
Antioxidant defenses in the ocular surface, Ocul. Surf.,
7, 176-185.
25.Behndig, A., Svensson, B., Marklund, S. L., and
Karlsson, K. (1998) Superoxide dismutase isoenzymes in the human eye,
Invest. Ophthalmol. Vis. Sci., 39, 471-475.
26.Gukasyan, H. J., Kim, K. J., Lee, V. H., and
Kannan, R. (2007) Glutathione and its transporters in ocular surface
defense, Ocul. Surf., 5, 269-279.
27.Hao, Y. H., Kuang, Z. H., Xu, Y., Walling, B. E.,
and Lau, G. W. (2013) Pyocyanin-induced mucin production is associated
with redox modification of FOXA2, Respir. Res., 14, doi:
10.1186/1465-9921-14-82.
28.Grover, V. K., Kumar, K. V., Sharma, S., Sethi,
N., and Grewal, S. P. (1998) Comparison of methods of eye protection
under general anaesthesia, Can. J. Anaesth., 45,
575-577.
29.Schmidt, P., and Boggild-Madsen, N. B. (1981)
Protection of the eyes with ophthalmic ointments during general
anaesthesia, Acta Ophthalmol. (Copenh.), 59, 422-427.
30.Siffring, P. A., and Poulton, T. J. (1987)
Prevention of ophthalmic complications during general anesthesia,
Anesthesiology, 66, 569-570.
31.Ganidagli, S., Cengi, M., Becerik, C., Oguz, H.,
and Kilic, A. (2004) Eye protection during general anaesthesia:
comparison of four different methods, Eur. J. Anaesthesiol.,
21, 665-667.
32.Cejkova, J., and Cejka, C. (2015) The role of
oxidative stress in corneal diseases and injuries, Histol.
Histopathol., 30, 893-900.
33.Pinazo-Duran, M. D., Gallego-Pinazo, R.,
Garcia-Medina, J. J., Zanon-Moreno, V., Nucci, C., Dolz-Marco, R.,
Martinez-Castillo, S., Galbis-Estrada, C., Marco-Ramirez, C.,
Lopez-Galvez, M. I., Galarreta, D. J., and Diaz-Llopis, M. (2014)
Oxidative stress and its downstream signaling in aging eyes, Clin.
Interv. Aging, 9, 637-652.
34.Tsubota, K., Kawashima, M., Inaba, T., Dogru, M.,
Ogawa, Y., Nakamura, S., Shinmura, K., Higuchi, A., and Kawakita, T.
(2010) The era of antiaging ophthalmology comes of age: antiaging
approach for dry eye treatment, Ophthalmic Res., 44,
146-154.
35.Brzheskiy, V. V., Efimova, E. L., Vorontsova, T.
N., Alekseev, V. N., Gusarevich, O. G., Shaidurova, K. N., Ryabtseva,
A. A., Andryukhina, O. M., Kamenskikh, T. G., Sumarokova, E. S.,
Miljudin, E. S., Egorov, E. A., Lebedev, O. I., Surov, A. V., Korol, A.
R., Nasinnyk, I. O., Bezditko, P. A., Muzhychuk, O. P., Vygodin, V. A.,
Yani, E. V., Savchenko, A. Y., Karger, E. M., Fedorkin, O. N., Mironov,
A. N., Ostapenko, V., Popeko, N. A., Skulachev, V. P., and Skulachev,
M. V. (2015) Results of a multicenter, randomized, double-masked,
placebo-controlled clinical study of the efficacy and safety of
visomitin eye drops in patients with dry eye syndrome, Adv.
Ther., 32, 1263-1279.
36.Petrov, A., Perekhvatova, N., Skulachev, M.,
Stein, L., and Ousler, G. (2016) SkQ1 ophthalmic solution for dry eye
treatment: results of a phase 2 safety and efficacy clinical study in
the environment and during challenge in the controlled adverse
environment model, Adv. Ther., 33, 96-115.
37.Yani, E. V., Katargina, L. A., Chesnokova, N. V.,
Beznos, O. V., Savchenko, A. Yu., Vigodin, V. A., Gudkova, E. Yu.,
Zamyatnin, A. A., Jr., and Skulachev, M. V. (2012) The first experience
of using vizomitin drug in the treatment of “dry eye”,
Prakt. Med., 1, 134-137.