The Use of Human Induced Pluripotent Stem Cells for Testing Neuroprotective Activity of Pharmacological Compounds
E. V. Novosadova1,a*, E. L. Arsenyeva1, S. A. Antonov1, Y. N. Vanyushina1, T. V. Malova1, A. A. Komissarov1, S. N. Illarioshkin2, L. G. Khaspekov2, L. A. Andreeva1, N. F. Myasoedov1, V. Z. Tarantul1, and I. A. Grivennikov1,b*
1Institute of Molecular Genetics, Russian Academy of Sciences, 123182 Moscow, Russia2Research Center of Neurology, 125367 Moscow, Russia
* To whom correspondence should be addressed.
Received June 20, 2019; Revised July 23, 2019; Accepted August 6, 2019
Development of therapeutic preparations involves several steps, starting with the synthesis of chemical compounds and testing them in different models for selecting the most effective and safest ones to clinical trials and introduction into medical practice. Cultured animal cells (both primary and transformed) are commonly used as models for compound screening. However, cell models display a number of disadvantages, including insufficient standardization (primary cells) and disruption of cell genotypes (transformed cells). Generation of human induced pluripotent stem cells (IPSCs) offers new possibilities for the development of high-throughput test systems for screening potential therapeutic preparations with different activity spectra. Due to the capacity to differentiate into all cell types of an adult organism, IPSCs are a unique model that allows examining the activity and potential toxicity of tested compounds during the entire differentiation process in vitro. In this work, we demonstrated the efficiency of IPSCs and their neuronal derivatives for selecting substances with the neuroprotective activity using two classes of compounds – melanocortin family peptides and endocannabinoids. None of the tested compounds displayed cyto- or embryotoxicity. Both melanocortin peptides and endocannabinoids exerted neuroprotective effect in the neuronal precursors and IPSC-derived neurons subjected to hydrogen peroxide. The endocannabinoid N-docosahexaenoyl dopamine exhibited the highest neuroprotective effect (~70%) in the differentiated cultures enriched with dopaminergic neurons; the effect of melanocortin Semax was ~40%. The possibility of using other IPSC derivatives for selecting compounds with the neuroprotective activity is discussed.
KEY WORDS: test-system, embryotoxicity, neuroprotection, induced pluripotent stem cells, oxidative stressDOI: 10.1134/S0006297919110075
Abbreviations: alpha-MSH, alpha-melanocyte-stimulating hormone; BDNF, brain-derived neurotrophic factor; DA neuron, dopaminergic neuron; DMSO, dimethyl sulfoxide; EC, endocannabinoid; G418, geneticin; GDNF, glial cell-derived neurotrophic factor; IPSC, induced pluripotent stem cell; MC, melanocortin; N-ADA, N-arachidonoyl dopamine; N-DDA, N-docosahexaenoyl dopamine; PGP, melanocortin family peptide Pro-Gly-Pro.
One of the essential problems of modern pharmacology and medicine is
creation of efficient and safe drugs, which requires development of
high-throughput, reproducible, and cost-effective technologies for
compound screening. Currently, the average cost of a new drug
development is estimated as several billion dollars, and it usually
takes 10-15 years for a preparation to be introduced into clinical
practice, starting from its synthesis and testing. The development of a
new therapeutic preparation incudes several steps, such as synthesis of
prospective compounds, their testing in different models in order to
select the most active and safest ones, clinical tests, and
introduction to medical practice. Cultured animal cells (primary,
transformed, and transfected) are important test models used for the
screening of novel compounds. Drug testing in cell cultures is a
mandatory step in the development of therapeutic preparations that
precedes testing in laboratory animals. Various types of cells cultures
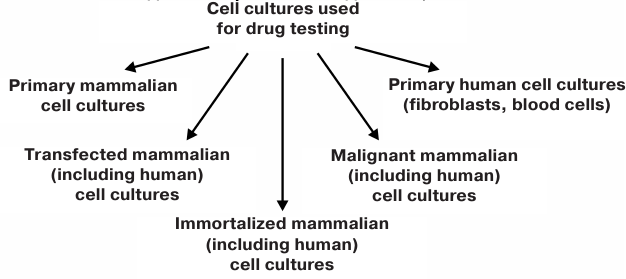
used for screening of pharmacological compounds are shown in Fig. 1.
Fig. 1. Animal cell cultures used for testing pharmacological compounds.
Mammalian cell cultures can be classified into two main types: primary and transformed. Mammalian cell cultures artificially transfected to contain a gene responsible for the development of one or another human disease must be also mentioned, as we have used such cultures for testing various peptide compounds for their neuroprotective activity [1, 2].
However, animals and animal-derived cell cultures are often non-adequate models for screening therapeutic preparations because of species-specific responses. It is known that some drugs successfully tested in such models later proved to be unsuitable or even toxic in humans [3, 4].
Reprogramming of somatic cells and production of induced pluripotent stem cells (IPSCs) from mammals, including humans, has opened new prospects in transplantation and studies of molecular and cellular mechanisms of human diseases in vitro [5, 6]. This technology has created new opportunities for the development of models for severe human pathologies, including neurodegenerative disorders, and facilitated development of test systems for the high-throughput in vitro screening of components of therapeutic preparations tailored to individual patients [7-9].
The most important characteristic of IPSCs is their capacity to differentiate into all cell types of an adult organism. Various derivatives have been generated from human IPSCs, including retinal pigment epithelium, motoneurons, cortical neurons, GABAergic, dopaminergic, and sensor neurons, Purkinje cells, oligodendrocytes, endothelial cells, cardiomyocytes, hematopoietic cells, osteogenic cells, neutrophils, etc. [5, 6]. The majority of these cell were derived from patients with genetic pathologies and, therefore, displayed altered genotypes typical for particular diseases. Such cells have been used as models for both molecular genetic investigation of disease pathogenesis and screening of potential therapeutic preparations [6].
As a model system, human IPSCs present several advantages as a model system for investigating the functions and pathologies of the nervous system, as well as for searching efficient treatments for neurological disorders because of the following:
1) impossibility to obtain neurons from adult human brain (with exception of surgical removal of malignant tumors);
2) inability of differentiated human neurons to grow in cell culture;
3) possibility of production of unlimited amounts of IPSCs and their derivatives;
4) possibility of obtaining IPSCs from a particular donor, as well as from patients with neurodegenerative diseases;
5) possibility of targeted differentiation of IPSCs to neuronal or glial lineage in vitro with the generation of particular type of neurons.
In our laboratory, we have used human IPSCs to develop a test system for the high-throughput screening of various pharmacological compounds and evaluation of their cytotoxicity, embryotoxicity, and neuroprotective potential [10, 11].
The objective of this study was to examine the efficiency of the developed and patented test system in the screening of compounds with the neuroprotective activity using two groups of chemical compounds, such as peptides of the melanocortin family [alpha-melanocyte-stimulating hormone (alpha-MSH), Semax (Met-Glu-His-Phe-Pro-Gly-Pro), and PGP (Pro-Gly-Pro)] and endocannabinoids [N-docosahexaenoyl dopamine (N-DDA) and N-arachidonoyl dopamine (N-ADA)].
MATERIALS AND METHODS
Cell culturing and synthesis of compounds. Human IPSCs and their derivatives produced in [12] were used in this work. Culturing and evaluation of IPSC proliferative activity were carried out as described previously [13]. Melanocortins (MCs) were synthesized at the Division of Chemistry of Physiologically Active Compounds, Institute of Molecular Genetics, Russian Academy of Sciences; endocannabinoids (ECs) were synthesized in the Laboratory of Oxylipins, Shemyakin–Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences.
Evaluation of compound cytotoxicity in IPSCs. IPSCs were seeded at 40,000 cells per well in 24-well plates and cultured in 1 ml of mTeSR1 medium (STEMCELL Technologies, Canada) in a CO2 incubator (Sanyo, Japan) at 37°C and 5% CO2. Neuronal precursors produced from the IPSCs as described previously [14] were seeded at 20,000 cells per well in a 96-well plate and cultured in the neuronal medium containing DMEM/F12 (Gibco, USA), 2% serum replacement (Gibco), 1% B27 supplement (50×; Gibco), 2 mM glutamine (ICN Biomedicals, USA), 1 mM non-essential amino acids (PanEko, Russia), 50 units/ml penicillin-streptomycin (PanEko), 10 μM SB431542 (Stemgent, USA), 80 ng/ml Noggin (Stemgent) in 5% CO2 at 37°C and. The plates were coated with Matrigel (Corning, USA) prior to the experiments. Next day, the medium in the wells was replaced with the medium containing investigated compounds at the final concentration of 0.001, 0.1, and 10 µM and antibiotic geneticin (G418; Gibco) at the concentrations of 50, 100, and 200 μg/ml. After 48 h, the medium was replaced with the fresh medium containing the corresponding compound. On day 3 of incubation, the proliferative activity of the cells was examined with the MTT assay. For this purpose, an aliquot (400 μl) of freshly prepared 0.3 mg/ml MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] was added to each well and incubated for 3 h in a CO2-incubator. Next, the MTT solution was replaced with DMSO (400 μl per well) followed by incubation for 2 h at room temperature on a shaker (Eppendorf, Germany). The optical density in the wells was recorded at 600 nm with an Infinite M200 Pro plate reader (Tecan, Switzerland).
Effect of MC peptides on the formation of embryoid bodies from IPSCs. In order to produce embryoid bodies, IPSC colonies were detached from the support using dispase (Gibco). Next, the colonies were transferred to a 24-well ultra-low attachment with (Costar, USA) at 160,000 cells per well in 1 ml of mTeSR1 growth medium for non-differentiated cells containing 50 μg/ml G418; MC peptides were added to the wells at the concentrations of 0.001, 0.1, and 10 μM, and the cells were incubated for 24 h at 37°C in 5% CO2. Next, half of the medium was replaced with the medium for the embryoid body growth containing DMEM/F12 (Gibco), 20% fetal bovine serum (Hyclone, Denmark), 2 mM L-glutamine, 0.1 mM β-mercaptoethanol, 1 mM non-essential amino acids, and 50 units/ml penicillin-streptomycin. After 24 h, the medium was completely replaced with the embryoid body growth medium containing G418 and MC peptides in the corresponding concentrations. Embryoid bodies were counted under the microscope 24-48 h after cell seeding. For this purpose, the entire cell suspension was transferred to a round-bottom 96-well plate (100 μl per well) to provide the maximal accuracy in determining the number of embryoid bodies in each case.
Generation of differentiated cultures enriched with dopaminergic neurons. Neuronal precursors were seeded into Petri dishes treated with Matrigel at 200,000 cells per cm². Next day, the medium was replaced with the type I differentiation medium containing DMEM/F12, 2% serum replacement, 1% B27 supplement (50×), 2 mM glutamine, 1 mM non-essential amino acids, 50 units/ml penicillin-streptomycin, 100 ng/ml recombinant Sonic hedgehog protein (PeproTech, USA), 20 ng/ml FGF8 (PeproTech), and 10 μM purmorphamine (Stemgent). The cells were cultured for 10 days; the medium was changed every other day. Next, the cells were transferred to the type II differentiation medium containing DMEM/F12, 2% serum replacement, 1% B27 supplement (50×), 2 mM glutamine, 1% amino acid mixture, 50 units/ml penicillin-streptomycin, 20 ng/ml brain-derived neurotrophic factor (BDNF; PeproTech), 20 ng/ml glial cell-derived neurotrophic factor (GDNF; PeproTech), 200 μM ascorbic acid (PanEko), and 4 μM forskolin (Stemgent) followed by incubation for 14-21 days at 37°C in 5% CO2, (the medium was replaced every 48 h).
Assessment of neuroprotective effect of MC peptides and ECs on the neuronal precursors and dopaminergic neurons under oxidative stress. Neuronal precursors were seeded into a Matrigel-coated 96-well plate at 20,000-30,000 cells per well and incubated for 24 h. Dopaminergic neurons were produced by culturing under conditions described in the above section. Following generation of mature neurons, the medium was replaced with the test medium (DMEM/F12, 2 mM L-glutamine, 50 units/ml of penicillin-streptomycin) containing MC peptides (Semax, alpha-MSH, PGP; 50 μl per well) at the final concentration of 1 nM or ECs (N-DDA, N-ADA) at the final concentrations of 2.5 or 5.0 μM. An equal volume of vehicle (PBS for MC peptides and DMSO for ECs) was used as a control. The cells were incubated for 40 min; then, 50 µl of the medium containing 200 μM Н2O2 was added per well, and the cells were incubated for another 3 h. Next, the medium was replaced with the test medium, and the number of viable neuronal precursors was determined on the next day using the MTT assay.
Immunofluorescence assay. Dopaminergic (DA) neurons were fixed with 4% paraformaldehyde and stained with polyclonal rabbit anti-tyrosine hydroxylase antibodies (1 : 1000; Abcam, USA) and monoclonal mouse antibodies anti-βIII-tubulin antibodies (1 : 2000; Abcam, USA) with the following detection using goat polyclonal antibodies against mouse and rabbit immunoglobulins conjugated with Alexa 488 and Alexa 546 dyes, respectively (1 : 2000; Thermo Fischer, USA) as described previously [12]. Stained cells were counted under an AXIO Imager Z1 fluorescence microscope (Zeiss, Germany). The intensity of fluorescence signal in the plate wells was evaluated with an Infinite M200 Pro plate reader (Tecan). The images were analyzed for the number of nuclei and cells using the ImageJ-win64 program (USA), plugin ITCN.
Statistical analysis. Results are shown as mean values ± standard deviation of the mean for 3-4 independent experiments. In each experiment, 3-4 replicates per point were carrier out. The significance of differences between the experimental groups was estimated using dispersion analysis (one-way ANOVA); significance level was set at p < 0.05.
RESULTS
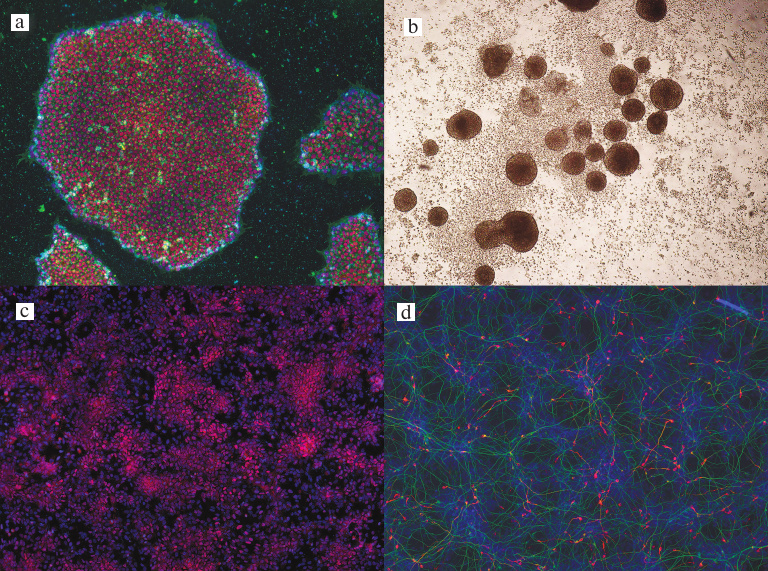
As mentioned above, we have previously developed a test system based on human IPSCs for the high-throughput screening of pharmacological compounds for cytotoxicity, embryotoxicity, and neuroprotective potential [10, 11]. Non-differentiated IPSCs, embryonic bodies, neuronal precursors, and cells cultures differentiated into neuronal lineage and enriched with DA neurons were used for verifying the developed test system (see Fig. 2 for the microphotographs of the used cell cultures).
Fig. 2. IPSC cultures at different stages of differentiation: a) non-differentiated IPSCs stained for SSEA-4 (green) and Oct3/4 (red); b) embryonic bodies without staining; c) neuronal precursors stained for Sox1 (red); d) differentiated neurons stained for βIII-tubulin (green) and tyrosine hydroxylase (red); ×100. Cell nuclei were stained with DAPI (blue).
Cytotoxicity and embryotoxicity of the tested compounds and proliferative activity of treated cells were evaluated with the MTT assay as the simplest technique for a high-throughput screening. The neuroprotective potential of the investigated compounds in differentiated cell cultures was determined by immunofluorescence staining from the expression of specific markers (βIII-tubulin for neurons and tyrosine hydroxylase for DA neurons). The peptide Semax, which is a basic component of therapeutic preparation with the established neuroprotective properties, was used as a positive control [15, 16]; the antibiotic geneticin (G418) exhibiting the cytotoxic effect on eukaryotic cells was used as a negative control.
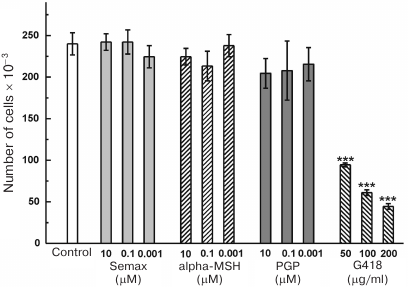
Effect of MC peptides on the IPSC proliferative activity and ability to form embryoid bodies. The effect of 0.001-10 μM MC peptides on the survival and proliferation of non-differentiated IPSCs treated with G418 (50, 100, and 200 μg/ml) (Fig. 3). At all the tested concentrations, G418 caused pronounced cell death. Semax, alpha-MSH, and PGP did not affect proliferation of IPSCs in a wide concentration range and did not display the cytotoxic activity (Fig. 3).
Fig. 3. Effect of MC peptides and G418 on the proliferative activity of IPSCs; n = 3; *** p ≤ 0.005.
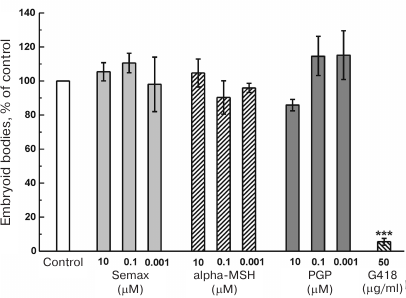
Next, we studied the effect of the tested compounds on the embryoid body formation, which models initial stages of embryonic development. For this purpose, IPSCs were detached from the support and placed in ultra-low attachment plates to eliminate cell attachment to the support. Under these conditions, IPSCs enter initial stages of differentiation and form embryoid bodies. The number of formed embryoid bodies was evaluated after 72 h of cell incubation in the presence of tested MC peptides by direct counting under a microscope (Fig. 4). We found that the MC peptides did not produce any significant effect on the number of embryoid bodies. At 50 μg/ml, G418 significantly decreased the number of formed embryoid bodies (Fig. 4), while 100 and 200 μg/ml G418 completely inhibited embryoid body formation (data not shown).
Fig. 4. Effect of MC peptides and G418 on the ability of IPSCs to form embryoid bodies; n = 4; *** p ≤ 0.005.
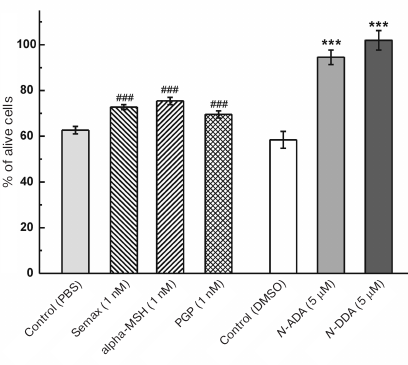
Evaluation of neuroprotective effect of MC peptides and ECs on human IPSCs at different stages of neuronal differentiation. We investigated the ability of MC peptides (Semax, alpha-MSH, and PGP) and ECs (N-DDA and N-ADA) to exhibit the neuroprotective effect under conditions of oxidative stress induced by hydrogen peroxide. For this purpose, neuronal precursors were seeded into 96-well plates coated with Matrigel. The medium was replaced after 24 h with simultaneous addition of the investigated compounds. Next, the cells were incubated for 1 h, followed by addition of hydrogen peroxide at the indicated concentrations. After 24 h, the number of viable cells was estimated using the MTT assay (Fig. 5). We found that both MC peptides and ECs produced the neuroprotective effect in the generated IPSC-derived neuronal precursors. In particular, the number of survived cells in the presence of Semax, alpha-MSH, and PGP was approximately 15, 20, and 10% higher, respectively, than in the control. The number of survived cells in the presence of N-ADA and N-DDA was by 36 and 47% higher, respectively, than in the control.
Fig. 5. Effect of MC peptides and ECs on the survival of neuronal precursors under oxidative stress induced by 200 μM Н2O2. The number of alive cells in the presence of investigated compounds without Н2O2 addition was taken as 100%; n = 4; *** p ≤ 0.001 in comparison with control (PBS); ### p ≤ 0.001 in comparison with control (DMSO).
To obtain cultures enriched with DA neurons, neuronal precursors were seeded into 96-well plates coated with Matrigel and cultured as described in the “Materials and Methods” section. After 15 days of culturing, the medium was replaced with the new one containing the studied compounds (1.0 nM MC peptides, 2.5 or 5.0 μM of ECs). The cells were incubated for 1 h followed by addition of Н2O2 to the final concentration of 200 μM. After 24 h, the cells were fixed and stained immunofluorescently using antibodies against βIII-tubulin (total neuronal population) and tyrosine hydroxylase (DA neurons). The neuroprotective effect of the analyzed MC peptides varied (table). Thus, Semax increased the number of survived cells by 40% vs. control in both total (Tub+) and DA (TH+) neurons, while alpha-MSG demonstrated the same protective activity only for the DA neurons (TH+) (table).
Effect of MC peptides (alpha-MSH and Semax) and ECs (N-ADA and
N-DDA) on the survival of differentiated neurons under oxidative
stress

Note: Tub+, cells stained with antibodies against
βIII-tubulin (total neuronal population); TH+, cells
stained with antibodies against tyrosine hydroxylase (DA neurons);
n = 4; ** p ≤ 0.01; *** p ≤ 0.005.
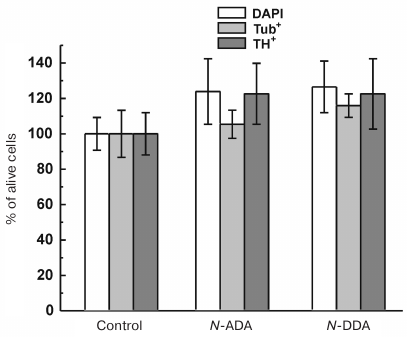
We also studied the effect of 2.5 and 5.0 μM ECs (N-DDA and N-ADA) on the survival of differentiated neurons under the Н2O2-induced oxidative stress. No protective effect of 2.5 μM ECs was observed (Fig. 6), while at the concentration of 5.0 μM, both ECs demonstrated reliable neuroprotective properties (table). The number of survived cells in the presence of N-ADA in both cell populations was ~30% higher than in the control; N-DDA demonstrated the highest neuroprotective effect among the all investigated compounds (~70%) (table).
Fig. 6. Effect of 2.5 μM ECs (N-ADA and N-DDA) on the survival of neurons under oxidative stress. The number of alive cells in the presence of ECs without Н2O2 addition was taken as 100%; n = 3.
DISCUSSION
Because of the normal karyotype and capacity to differentiate into all types of cells of an adult organism, IPSCs represent a unique model for fundamental, pharmacological, and toxicological studies [7, 9, 17]. By transforming consequently through the differentiation stages from immature precursors to terminally differentiated cells, IPSCs allow to evaluate various aspects of the actions of pharmacological compounds. Testing compounds in human IPSCs has made it possible to replace similar experiments in animal cells. In recent years, the use of IPSCs and their differentiated derivatives as test systems for drug screening has received wide recognition [6, 9, 17]. In particular, hepatocytes and cardiomyocytes have been used to search for compounds for the treatment of hypercholesterolemia and some cardiovascular diseases [18, 19]. Much attention has been paid to the use of IPSCs differentiated into neuronal lineage for selecting compounds with the neuroprotective activity [20-23].
Our research group has developed and patented an IPSC-based test system for screening of various compounds for their cytotoxicity, embryotoxicity, and neuroprotective activity [10, 11]. This test system allows to evaluate not only the cytotoxicity of the investigated compound, but also its embryotoxicity and teratogenic properties, since formation of embryoid bodies mimics the pre-implantation stage of human embryo. Further differentiation of embryoid bodies in vitro for 3-5 days is similar to the embryo development from the blastocyst (3.5 days) to the gastrula (7.5 days) [24], which makes it possible to relate embryonic development in vitro to embryogenesis in in vivo. Here, we demonstrated that MC peptides do not affect the proliferative activity of IPSCs and their ability to form embryoid bodies, i.e., lack cytotoxicity and embryotoxicity. At the same, the antibiotic geneticin caused the death of non-differentiated IPSCs and blocked their ability to form embryoid bodies, thus displaying pronounced cytotoxic properties. The capacity of IPSCs to differentiate into neurons allows identification of compounds with neurotoxic and neuroprotective activities. We analyzed the effect of two groups of compounds (MC peptides and ECs) at different stages of neuronal differentiation. Both MC peptides and ECs are considered as potential therapeutic preparations with neuroprotective properties [15, 16, 25, 26]. However, the vast majority of studies demonstrating their beneficial effect have been conducted in either human tumor cells or animal-derived cells [25-27]. Thus, N-ADA and N-DDA were found to exhibit neuroprotective properties in the primary neuronal cultures derived from the rat brain [25, 26]. Similar effects were observed for the MC peptide Semax [15, 16].
The data of these studies should be extrapolated to “normal” human cells with great care. Here, we investigated for the first time the effect of MC peptides and ECs on the neuronal population of human cells at different stages of differentiation. All the tested compounds demonstrated neuroprotective effect in cells at the neuronal precursor stage subjected to the hydrogen peroxide-induced oxidative stress. Furthermore, the neuroprotective efficiency was different for different groups of compounds. The number of cells that survived oxidative stress in the presence of ECs was ~40% higher than in the control; while the MC peptides increased cell survival by 10-20% only. The differentiated cultures enriched with DA neurons were protected by the investigated compounds to a varying degree. While ECs at the concentration of 2.5 μM did not display neuroprotection properties, at higher concentration (5.0 μM) N-DDA demonstrated the highest neuroprotection with regards to DA neurons (~70%). The neuroprotective effect 5.0 μM of N-ADA was only ~30%. The MC peptides Semax and alpha-MSH also exhibited the neuroprotective effect in these cultures (~40%) (table).
Hence, in this work, we confirmed the efficiency of the developed test system in the evaluation of cytotoxicity, embryotoxicity, and neuroprotective potential of the tested compounds. The question on the molecular mechanisms of this neuroprotective effect remains open. It is possible that it is mediated through specific receptors located on the IPSC surface. The presence of receptors for MC peptides and ECs has been established by us earlier [13, 28]. It was demonstrated that the neuroprotective effect of ECs is associated with the presence of respective type 1 and 2 receptors on the cells [29].
Apparently, testing therapeutic preparations on specialized cells at certain stages of differentiation is very important. However, a potential negative effect on the population of surrounding cells should be taken into account. The number of glial cells that surround neuron in the human brain is approximately the same as the number of neurons themselves. So far, the studies on the involvement of glial cells in the development of neurodegenerative disorders have been very scarce; although, it has been found that astrocytes, for example, play an important role in the development of some neurodegenerative diseases [30, 31]. That is why we plan in the future to generate glial cultures from IPSCs in order to develop similar test system for screening potential drugs with the neuroprotective activity. Earlier, we have used similar test system based on the primary cultures of rat brain neurons and glial cells [27]. It seems especially important to use IPSCs obtained from the patients with genetic forms of neurodegenerative diseases, because this will give the possibility not only to monitor effects on specialized neurons undergoing degeneration in the brain, but also to evaluate the contribution of glial cells to this process [31, 32].
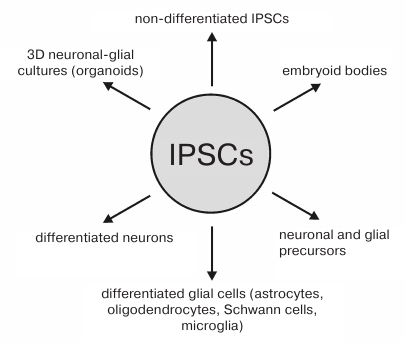
In Fig. 7, we summarized the possible uses of IPSCs and their derivatives in the selection of compounds with the neuroprotective activity.
Fig. 7. IPSCs and their neuronal and glial derivatives used for screening compounds with the neuroprotective activity.
The use of three-dimensional neuronal-glial cultures (organoids) that was first described in 2013, is considered a very promising approach for development of more complex models for investigating human nervous system pathologies and screening potential therapeutic preparations with neuroprotective activity [33]. Currently, such cultures are widely used as models for studying brain pathologies [34, 35]. Despite the attractiveness of this in vitro method, there are certain limitations that prevent considering formed organoids as an adequate model of complex three-dimensional structures formed form by the cells in the brain. This is related mostly to different sets of trophic and differentiation factors present in an organism in vivo and used by researchers in vitro. Nevertheless, numerous studies are underway pursuing production of more standardized organoids [36].
It must be also emphasized that the developed test system can be used for high-throughput screening of chemical compounds for their neuroprotective activity. The use of 96- and 384-well plates and highly sensitive fluorescence or cytochemical detection methods, as well as modern analyzers, offer promise of simultaneous screening of thousand and more compounds per day. At present, such approaches are known and used in a number of foreign laboratories [37-42]. In the case of differentiated neuronal cultures, the problem that still needs to be addressed is a long period of time required for the terminal differentiation of these cells in vitro (more than 3-4 weeks).
In conclusion, our experiments provided evidence that the use of IPSCs and their neuronal derivatives at different stages of differentiation is a promising approach for screening neuroprotective compounds, which allowed us to identify a number of compounds with neuroprotective activity.
Funding. This work was supported by the Russian Foundation for Basic Research (project 17-04-01661a) and the Program of the Presidium of the Russian Academy of Sciences “Fundamental studies for biomedical technologies”. The equipment used in the study was provided by the Center of Collective Use of the Institute of Molecular Genetics, Russian Academy of Sciences (Center for Cell and Gene Technologies).
Acknowledgements. The authors express gratitude to V. V. Bezuglov (Shemyakin–Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences) for kindly providing endocannabinoids for this study.
Conflict of interest. The authors declare no conflict of interest in financial or any other sphere.
Ethical approval. This article does not contain any studies with human participants or animals performed by any of the authors.
REFERENCES
1.Grivennikov, I. A., Dolotov, O. V., Inozemtseva, L.
S., Antonov, S. A., Kobylyanskii, A. G., and Myasoedov, N. F. (2011)
The use of primary cultures of mammalian nerve and glial cells for
selection of compounds with neuroprotective activity, Vestn.
Biotekhnol. Fiz.-Khim. Biol. Yu. A. Ovchinnikova, 7,
24-31.
2.Novosadova, E. V., Grivennikov, I. A., Bobrysheva,
I. V., Grigorenko, A. P., Andreeva, L. A., Rogaeva, E. I., and
Tarantul, V. Z. (2012) Semax positively affects viability of the
transgenic pheochromocytoma line PC12 carrying human presenilin-1
mutant gene (hPS1), Vestn. Biotekhnol. Fiz.-Khim. Biol. Yu. A.
Ovchinnikova, 8, 15-21.
3.Shefer, K., Shpilmann, Kh., and Fetter, K. (2010)
Drug Therapy during Pregnancy and Lactation [in Russian]
(Romanov, B. K., ed.), Logosfera, Moscow.
4.Kalter, H. (2003) Teratology in the 20th century:
environmental causes of congenital malformations in humans and how they
were established, Neurotoxicol. Teratol., 25,
131-282.
5.Takahashi, K., Tanabe, K., Ohnuki, M., Narita, M.,
Ichisaka, T., Tomoda, K., and Yamanaka, S. (2007) Induction of
pluripotent stem cells from adult human fibroblasts by defined factors,
Cell, 131, 861-872; doi: 10.1016/j.cell.2007.11.019.
6.Elitt, M. S., Barbar, L., and Tesar, P. J. (2018)
Drug screening for human genetic diseases using iPSC models, Hum.
Mol. Genet., 27, R89-R98; doi: 10.1093/hmg/ddy186.
7.Piccinno, M. S., Petrachi, T., Resca,
E., Strusi, V., Bergamini, V., Mulas, G.
A., Mari, G., Dominici, M., and Veronesi, E. (2018)
Label-free toxicology screening of primary human mesenchymal cells and
iPS-derived neurons, PLoS One, 13, e0201671; doi:
10.1371/journal.pone.0201671.
8.Vu, M., Li, R., Baskfield, A., Lu, B., Farkhondeh,
A., Gorshkov, K., Motabar, O., Beers, J., Chen, G., Zou, J.,
Espejo-Mojica, A. J., Rodriguez-Lopez, A., Almeciga-Diaz, C. J.,
Barrera, L. A., Jiang, X., Ory, D. S., Marugan, J. J., and Zheng, W.
(2018) Neural stem cells for disease modeling and evaluation of
therapeutics for Tay–Sachs disease, Orphanet. J. Rare Dis., 13,
152; doi: 10.1186/s13023-018-0886-3.
9.Cota-Coronado, A., Ramirez-Rodriguez, P. B.,
Padilla-Camberos, E., Diaz, N. F., Flores-Fernandez, J. M.,
Avila-Gonzalez, D., and Diaz-Martinez, N. E. (2018) Implications of
human induced pluripotent stem cells in metabolic disorders: from drug
discovery toward precision medicine, Drug Discov. Today, 24, 334-341;
doi: 10.1016/j.drudis.2018.10.001.
10.Novosadova, E. V., Andreeva, L. A.,
Arsen’eva, E. L., Grivennikov, I. A., Illarioshkin, S. N.,
Lebedeva, O. S., Makarova, I. V., Manuilova, E. S., Myasoedov, N. F.,
and Tarantul, V. Z. (2016) Use of human induced pluripotent stem cells
for testing of cyto- and embryotoxicity of pharmacological compounds,
RF Patent 2599847 C1.
11.Novosadova, E. V., Andreeva, L. A.,
Arsen’eva, E. L., Grefenshtein, M. A., Grivennikov, I. A.,
Illarioshkin, S. N., Inozemtseva, L. S., Lebedeva, O. S., Manuilova, E.
S., and Myasoedov, N. F. (2018) A method for testing neuroprotective
activity of compounds in vitro, and the relevant test-system, RF
Patent 2646446 C1.
12.Novosadova, E. V., Nekrasov, E. D., Chestkov, I.
V., Surdina, A. V., Vasina, E. M., Bogomazova, A. N., Manuilova, E. S.,
Arsenyeva, E. L., Simonova, V. V., Konovalova, E. V., Fedotova, E. Yu.,
Abramycheva, N. Yu., Khaspekov, L. G., Grivennikov, I. A., Tarantul, V.
Z., Kiselev, S. L., and Illarioshkin, S. N. (2016) A platform for
studying molecular and cellular mechanisms of Parkinson’s disease
based on human induced pluripotent stem cells, Sovr. Tehnol.
Med., 8, 155-164.
13.Novosadova, E. V., Manuilova, E. S.,
Arsen’eva, E. L., Andreeva, L. A., Lebedeva, O. S., Grivennikov,
I. A., and Myasoedov, N. F. (2016) Investigation of the effect of
alpha-melanocyte-stimulating hormone on proliferation and early stages
of differentiation of human induced pluripotent stem cells, Dokl.
Biochem. Biophys., 467, 141-144; doi:
10.1134/S1607672916020174.
14.Novosadova, E. V., Arsenyeva, E. L., Manuilova,
E. S., Khaspekov, L. G., Bobrov, M. Y., Bezuglov, V. V., Illarioshkin,
S. N., and Grivennikov, I. A. (2017) Neuroprotective properties of
endocannabinoids N-arachidonoyl dopamine and N-docosahexaenoyl dopamine
examined in neuronal precursors derived from human pluripotent stem
cells, Biochemistry (Moscow), 82, 1367-1372; doi:
10.1134/S0006297917110141.
15.Asmarin, I. P., Nezavibat’ko, V. N.,
Miasoedov, N. F., Kamenskii, A. A., Grivennikov, I. A.,
Ponomareva-Stepnaia, M. A., Andreeva, L. A., Kaplan, A. Ia., Koshelev,
V. B., and Riasina, T. V. (1997) A nootropic adrenocorticotropin analog
4-10-Semax (15-year experience in its design and study), Zh. Vyssh.
Nerv. Deiat. Im. I. P. Pavlova, 47, 420-430.
16.Myasoedov, N. F., Skvortsova, V. I., Nasonov, E.
L., Zhuravleva, E. Iu., Grivennikov, I. A., Arsenyeva, E. L., and
Sukhanov, I. I. (1999) Investigation of mechanisms of neuroprotective
effect of Semax in acute period of ischemic stroke, Zh. Nevrol.
Psikhiatr. Im. S. S. Korsakova, 99, 15-19.
17.Novosadova, E. V., and Grivennikov, I. A. (2014)
Induced pluripotent stem cells: from derivation to application in
biochemical and biomedical research, Biochemistry (Moscow),
79, 1425-1441; doi: 10.1134/S000629791413001X.
18.Cayo, M. A., Mallanna, S. K., and Di Furio, F.
(2017) A drug screen using human iPSC-derived hepatocyte-like cells
reveals cardiac glycosides as a potential treatment for
hypercholesterolemia, Stem Cell, 20, 478-489; doi:
10.1016/j.stem.2017.01.011.
19.Del’Alamo, J. C., Lemons, D., and Serrano,
R. (2016) High throughput physiological screening of iPSC-derived
cardiomyocytes for drug development, Biochim. Biophys. Acta,
1863, 1717-1727.
20.Malik, N., Efthymiou, A. G., Mather, K., Chester,
N., Wang, X., Nath, A., Rao, M. S., and Steiner, J. P. (2014) Compounds
with species and cell type specific toxicity identified in a 2000
compound drug screen of neural stem cells and rat mixed cortical
neurons, Neurotoxicology, 45, 192-200; doi:
10.1016/j.neuro.2014.10.007.
21.Peng, J., Liu, Q., Rao, M. S., and Zeng, X.
(2013) Using human pluripotent stem cell-derived dopaminergic neurons
to evaluate candidate Parkinson’s disease therapeutic agents in
MPP+ and rotenone models, J. Biomol. Screen,
18, 522-533; doi: 10.1177/1087057112474468.
22.Little, D., Ketteler, R., Gissen, P., and Devine,
M. J. (2019) Using stem cell-derived neurons in drug screening for
neurological diseases, Neurobiol. Aging, 78, 130-141;
doi: 10.1016/j.neurobiolaging.2019.02.008.
23.Garcia-Leon, J. A., Vitorica, J., and Gutierrez,
A. (2019) Use of human pluripotent stem cell-derived cells for
neurodegenerative disease modeling and drug screening platform,
Future Med. Chem., 11, 1305-1322; doi:
10.4155/fmc-2018-0520.
24.Desbaillets, I., Ziegler, U., Groscurth, P., and
Gassmann, M. (2000) Embryoid bodies: an in vitro model of mouse
embryogenesis, Exp. Physiol., 85, 645-651.
25.Bobrov, M. Y., Lizhin, A. A., Andrianova, E. L.,
Gretskaya, N. M., Frumkina, L. E., Khaspekov, L. G., and Bezuglov, V.
V. (2008) Antioxidant and neuroprotective properties of N-arachidonoyl
dopamine, Neurosci. Lett., 431, 6-11.
26.Bobrov, M. Y., Lyzhin, A. A., Andrianova, E. L.,
Gretskaya, N. M., Zinchenko, G. N., Frumkina, L. E., Khaspekov, L. G.,
and Bezuglov, V. V. (2006) Antioxidant and neuroprotective properties
of N-docosahexaenoyl dopamine, Bull. Exp. Biol. Med.,
142, 425-427.
27.Niu, N., and Wang, L. (2015) In vitro
human cell line models to predict clinical response to anticancer
drugs, Pharmacogenomics, 16, 273-285; doi:
10.2217/pgs.14.170.
28.Bobrov, M. Yu., Bezuglov, V. V., Khaspekov, L.
G., Illarioshkin, S. N., Novosadova, E. V., and Grivennikov, I. A.
(2017) Expression of type I cannabinoid receptors at different stages
of neuronal differentiation of human fibroblasts, Bull. Exp. Biol.
Med., 163, 272-275; doi: 10.1007/s10517-017-3782-2.
29.Vendel, E., and de Lange, E. C. (2014) Functions
of the CB1 and CB2 receptors in neuroprotection at the level of the
blood-brain barrier, Neuromol. Med., 16, 620-642; doi:
10.1007/s12017-014-8314-x.
30.Hauser, D. N., and Cookson, M. R. (2011)
Astrocytes in Parkinson’s disease and DJ-1, J. Neurochem.,
117, 357-358.
31.Krencik, R., and Ullian, E. M. (2013) A cellular
star atlas: using astrocytes from human pluripotent stem cells for
disease studies, Front. Cell. Neurosci., 7, 25; doi:
10.3389/fncel.2013.00025.
32.Di Domenico, A., Carola, G., Calatayud, C.,
Pons-Espinal, M., Munoz, J. P., Richaud-Patin, Y., Fernandez-Carasa,
I., Gut, M., Faella, A., Parameswaran, J., Soriano, J., Ferrer, I.,
Tolosa, E., Zorzano, A., Cuervo, A. M., Raya, A., and Consiglio, A.
(2019) Patient-specific iPSC-derived astrocytes contribute to
non-cell-autonomous neurodegeneration in Parkinson’s disease,
Stem Cell Rep., 12, 213-229; doi:
10.1016/j.stemcr.2018.12.011.
33.Lancaster, M. A., Renner, M., and Martin, C.-A.
(2013) Cerebral organoids model human brain development and
microcephaly, Nature, 501, 373-379; doi:
10.1038/nature12517.
34.Amin, N. D., and Pas, S. P. (2018) Building
models of brain disorders with three-dimensional organoids,
Neuron, 100, 389-405; doi:
10.1016/j.neuron.2018.10.007.
35.Bordoni, M., Rey, F., Fantini, V., Pansarasa, O.,
Di Giulio, A. M., Carelli, S., and Cereda, C. (2018) From neuronal
differentiation of iPSCs to 3D neuro-organoids: modelling and therapy
of neurodegenerative diseases, Int. J. Mol. Sci., 19,
3972; doi: 10.3390/ijms19123972.
36.Eremeev, A. V., Volovikov, E. A., Shuvalova, L.
D., Davidenko, A. V., Khomyakova, E. A., Bogomiakova, M. E., Lebedeva,
O. S., Zubkova, O. A., and Lagarkova, M. A. (2019) Necessity is the
mother of invention or inexpensive, reliable, and reproducible protocol
for generating organoids, Biochemistry (Moscow), 84,
321-328; doi: 10.1134/S0006297919030143.
37.McArdle, P., Engberg, S., Bennett, N., Blackett,
C., and Wigglesworth, M. (2017) Enabling 1536-well high-throughput
cell-based screening through the application of novel centrifugal plate
washing, SLAS Discov., 22, 732-742; doi:
10.1177/2472555216683650.
38.Sherman, S. P., and Bang, A. G. (2018)
High-throughput screen for compounds that modulate neurite growth of
human induced pluripotent stem cell-derived neurons, Dis. Model
Mech., 11, No. 2, dmm031906; doi: 10.1242/dmm.031906.
39.Han, Y., Miller, A., Mangada, J., Liu, Y., and
Swistowski, A. (2009) Identification by automated screening of a small
molecule that selectively eliminates neural stem cells derived from
hESCs but not dopamine neurons, PLoS One, 4, e7155; doi:
10.1371/journal.pone.0007155.
40.Wang, C., Ward, M. E., Chen, R., Liu, K., Tracy,
T. E., Chen, X., Xie, M., Sohn, P. D., Ludwig, C., Meyer-Franke, A.,
Karch, C. M., Ding, S., and Li, G. (2017) Scalable production of
iPSC-derived human neurons to identify tau-lowering compounds by
high-content screening, Stem Cell Rep., 9, 1221-1233;
doi: 10.1016/j.stemcr.2017.08.019.
41.Traub, S., and Heilker, R. (2019) hiPS
cell-derived neurons for high-throughput screening, Methods Mol.
Biol., 1994, 243-263; doi: 10.1007/978-1-4939-9477-9_23.
42.Little, D., Luft, C., Pezzini-Picart, O., Mosaku,
O., Ketteler, R., Devine, M. J., and Gissen, P. (2019) Seeding induced
pluripotent stem cell-derived neurons onto 384-well plates, Methods
Mol. Biol., 1994, 159-164; doi:
10.1007/978-1-4939-9477-9_14.