REVIEW: Bacteriophages as Therapeutic Preparations: What Restricts Their Application in Medicine
V. V. Vlassov1,a*, N. V. Tikunova1, and V. V. Morozova1
1Institute of Chemical Biology and Fundamental Medicine, Siberian Branch of the Russian Academy of Sciences, 630090 Novosibirsk, Russia* To whom correspondence should be addressed.
Received June 15, 2020; Revised August 18, 2020; Accepted August 18, 2020
The increasing prevalence of bacterial pathogens with multiple antibiotic resistance requires development of new approaches to control infections. Phage therapy is one of the most promising approaches. In recent years, research organizations and a number of pharmaceutical companies have intensified investigations aimed at developing bacteriophage-based therapeutics. In the United States and European countries, special centers have been established that experimentally apply phage therapy to treat patients who do not respond to antibiotic therapy. This review describes the features of bacteriophages as therapeutic tools, critically discusses the results of clinical trials of bacteriophage preparations, and assesses the prospects for using phage therapy to treat certain types of infectious diseases.
KEY WORDS: bacteriophages, phage therapy, bacterial infections, multidrug resistance, synthetic bacteriophagesDOI: 10.1134/S0006297920110061
INTRODUCTION
Multiple antibiotic resistance of pathogenic bacteria is seen today as a global concern. Mortality rate of the infections caused by these bacteria grows every year, and WHO experts believe that it may reach 10 million deaths per year by 2050 and exceed cancer mortality (https://www.who.int/news-room/detail/29-04-2019-new-report). The most dangerous multidrug-resistant bacteria are those of the ESKAPE group: Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species. This situation calls for a vigorous search for new products and approaches to combat pathogenic bacteria. Phage therapy is now considered as one of much promising approaches. This therapy was first applied a century ago for treatment of acute intestinal diseases and skin infections [1, 2].
Bacteriophages were used most extensively and successfully in the Soviet Union, mainly for the treatment of wound infections, including postoperative complications [3-5], and intestinal diseases, including cholera outbreaks [6-8].
After the advent of antibiotics, which provided a successful solution to the problem of bacterial infections in the first years of application, the interest in phage therapy subsided. One of the reasons for this was that science and technology was not ready for manufacturing of complex biopharmaceutical products with bacteriophages. In 1960-2000, phage therapy existed in a few centers – in Poland and Georgia, and the only country to manufacture bacteriophage products industrially was the Soviet Union (later the Russian Federation). Bacteriophage therapeutics were used as a single-agent therapy [9-11] or in combination therapy with antibiotics [10, 12, 13].
Everything has changed in recent years. Armed with modern methods, we can now characterize bacteriophages in detail; we can make changes in their genomes and even design synthetic bacteriophages; we can also make preparations of individual bacteriophages under GMP conditions. Interest in phage therapy has recently been fueled by a series of reports on successful use of bacteriophages for the treatment of critically ill patients infected with multidrug-resistant microorganisms [14-17]. For the second year in a row, the United States and Europe are organizing large congresses on phage therapy focusing on the practical use of bacteriophages (https://www.kisacoresearch.com/events/phage-futures-europe; https://phage-futures.com/events/phage-futures-2019). Several companies have emerged that develop bacteriophage-based products; organizations in many countries are conducting clinical trials of these products. However, the authors of this review believe that analysis of the properties of bacteriophages and critical consideration of the results of bacteriophage research in experimental models of infections and the results of clinical trials give no cause for euphoria and reveal considerable problems that need to be addressed in order to create truly effective phage therapy methods.
PROPERTIES OF BACTERIOPHAGES AS POTENTIAL ANTIBACTERIAL
THERAPEUTICS
Bacteriophages, or bacterial viruses, are one of the most abaundant forms of life on our planet. They live everywhere, in water and on land, and specific bacteriophage can be likely found for each bacterial strain. Bacteriophages are extremely effective as bactericidal agents. As potential therapeutic agents, they can offer several benefits. Bacteriophages are natural objects, so their therapeutic applications cause no harm to environment. They do not damage the cells of higher organisms; they are nontoxic and highly specific; and when they kill the target pathogenic bacteria, they do not harm normal microflora of the macroorganism (host). Bacteriophages regulate composition of the microbiota; healthy people carry more than 1000 species of phages in their intestines. The most common phages found in human intestines are of the order Caudovirales (of the families Myoviridae, Siphoviridae, and Podoviridae), which are the same order as those used in phage therapy [18, 19].
Bacteriophages are capable of penetrating layers of epithelial cells in the gastrointestinal tract (GIT) by the mechanism termed transcytosis; i.e., phages can pass through the mucous membranes into the blood and then into the respiratory system, into the excretory organs, or to that place in the body where bacteria are proliferating. They enter bladder directly from the intestines. It is believed that every day, about 109 phage particles penetrate by means of transcytosis from the intestines into other tissues and organs, forming a phage community, which influences human health and immune status [20, 21]. Bacteriophages are able to overcome some of the physiological barriers, e.g., the blood–brain barrier, and get inside bones affected by osteomyelitis [22]. In the presence of target bacteria, phages multiply in the affected areas of the body, such as in an infected wound, and once phages have devoured the pathogen, they themselves fall prey to tissue macrophages, with liver and kidneys participating in the elimination of phages from the body [23].
High specificity of bacteriophages is not only their advantage, but their drawback too. In order to reliably suppress an infectious agent, one would require a range of phages specific to all the strains of the infectious agent. To treat a patient successfully, one needs to select a phage product specific to the strain that caused the infection. Moreover, a possibility exists that during therapy, the target bacterium may develop resistance to the phage; in this case, one will need to search for a new one [13, 14]. This is one of the challenges of phage therapy, and this is why bacteriophages cannot be regarded as the first-line drugs. Most bacterial infections can be successfully treated with one or two antibiotics, which are economical to manufacture, have long shelf-life, and provide wide specificity. Bacteriophages become a necessity only if antibiotic therapy fails, hence, the market for them is smaller. It is important that bacteriophages can be used in combination with antibiotics [24, 25].
Bacteria have many defense mechanisms against bacteriophages, which block each of the stages in the infection of the bacterial cell by the phage. The most well-known defense mechanisms include: changes in the receptor (a bacterial surface protein recognized by a phage particle) to prevent adsorption of the phage on the bacterium; changes in proteins whose functions are needed for the phage nucleic acid to penetrate into the cell; restriction–modification systems that protect bacterial DNA from restriction enzymes and allow these enzymes to selectively cleave phage DNA; and the CRISPR-Cas type systems, which ensure recognition and selective destruction of the phage nucleic acids that were previously encountered by the bacteria [26]. Bacteria can protect themselves against bacteriophages in the same way as against antibiotics and immune system cells, i.e., by forming a physical barrier – biofilm. Some bacteriophages are able to destroy biofilms with special enzymes and to lyse bacteria they contain [27, 28]. One of the ways to combat bacterial resistance is to use bacteriophage cocktails [29-31]. If phages kill bacteria faster than they replicate, the risk of resistance formation decreases.
Administering bacteriophages into the body induces an immune response, whose strength depends on the nature of the phage, method of administration, and characteristics of the organism [32, 33]. It was shown that dendritic cells react to bacteriophages similarly to other infectious agents; contact with them stimulates the innate and adaptive immune responses [34-36]. It was suggested that the immune response of the organism could reduce efficacy of the repeated use of the same bacteriophage product in a patient. Bacteriophages are part of the microbiota in healthy people; huge quantities of phages inhabit human body, and their interaction with the host immune system appears to be very complex and, as of today, not well understood [37, 38]. Experiments showed that in order to achieve a clinically significant immune response, it is necessary to administer large quantity of bacteriophages, much greater than is needed for phage therapy. Several studies found no connection between the production of neutralizing antibodies against bacteriophages and clinical outcome of the therapy [32, 39-41]. However, other studies showed that such connection existed [39, 42]. It is likely that phages lyse bacteria before the immune system produces antibodies against the phages. It was reported that administering bacteriophages could have a beneficial effect on the functions of immune system; i.e., administering bacteriophages reduced the level of proinflammatory cytokines and infiltration of leukocytes into the tissue [36, 38, 43]. However, there are observations to the contrary [44, 45].
Studies revealed the possibility of a synergistic effect, which was observed in several cases of combined use of antibiotics and bacteriophages [46]. The developed resistance to bacteriophage due to the changes in the structure of bacterial receptor or other proteins could adversely affect viability of the bacterium because these changes increase its sensitivity to antibiotics [47].
BACTERIOPHAGE THERAPY PRODUCTS
Bacteriophages are isolated from a wide variety of environmental sources. At present, research organizations in different countries possess a large number of bacteriophages, many of which were investigated using modern molecular biological methods: their genomes were decoded and their specificity and antibacterial properties were studied. Several countries have bacteriophage banks: the Félix d′Hérelle Reference Center for Bacterial Viruses at the University of Laval, the Leibniz-Institute DSMZ–German Collection of Microorganisms, and the Cell Cultures and Bacteriophage Bank of Korea, which provide bacteriophages on a commercial basis. Companies that develop phage therapy products have their own collections, access to which is limited.
Practice showed that selecting bacteriophage for the treatment of a given patient could require a wide search across the different collections. For example, in the well-known case of successful cure of a patient infected with A. baumannii with multiple antibiotic resistance, nine different bacteriophages were applied, which were found in three different collections [14].
If the treatment of a patient necessitates an urgent search for bacteriophage, one could consult Phage Directory (https://phage.directory), a special agency that coordinates the efforts of phage therapy organization from more than 20 countries. Phage Directory can quickly provide contacts for the exchange of phages required for the patient treatment.
Two approaches are used when developing bacteriophage therapy products. The first one is to create a cocktail of bacteriophages acting against different strains of a given pathogen or even several pathogens involved in the development of a given type of infection. In Georgia, the Biochimfarm Georgi company (AO Biokhimfarm) produces and upgrades twice a year the product Pyofag, which contains phages against Streptococcus, Staphylococcus, Escherichia coli, P. aeruginosa, and Proteus, and Intestifag, which contains phages against bacteria most relevant in the etiology of intestinal diseases. These cocktails are used to treat infectious and intestinal diseases. In Russia, NPO Mikrogen (http://www.bacteriofag.ru) produces more than ten different complex bacteriophage products for the treatment of bacterial infections in humans. Another company, SPC Mikromir, produces several veterinary products of bacteriophages and phage-containing gels designed to normalize microbial composition of the skin and oral cavity (at present, the gels manufactured by SPC Mikromir are not considered medical drugs).
The second type of phage therapy involves a personalized approach, i.e., selection of a phage or creation of a phage cocktail against the specific pathogen, or several pathogens, found in a patient. This approach has long been used in Poland, at the Ludwik Hirszfeld Institute of Immunology and Experimental Therapy, and in Georgia, at the Phage Therapy Center (http://medigroup.ge), which have been selecting bacteriophages from their collections for individual patients and even searching for new phages, if they could not find the necessary one in their collections.
Today, phage therapy uses lytic phages only [48, 49], which are unable to integrate their genome into the genome of bacteria in order to avoid horizontal transfer of unwanted genes. The bacteriophage should be tested for the absence of unwanted genes that determine its toxicity and antibiotic resistance and absence of the genetic elements responsible for the transfer of phage genes into the bacterial genome, which can be achieved using pyrosequencing techniques [50, 51].
Obvious requirements are established in modern medicine for bacteriophage products: they must be produced under GMP conditions and the product itself must be sterile and must not contain bacterial cell components, e.g., pyrogenic substances, which may cause unwanted side effects.
At present, an unresolved issue remains of designing dosage forms of bacteriophages that ensure their stability during storage and with different methods of administration into the body. Some phages are stable in solution; others are storable as dry products obtained by lyophilization in the presence of stabilizing agents such as sugars (trehalose, sorbitol) or milk proteins [52, 53]. For stabilization, phage particles could be incorporated into liposomes [54-57]; to protect phages from attacks of immune system, it was proposed to modify their surface with polyethylene glycol [58].
Modern synthetic biology methods are capable of combining bacteriophage genomes and even synthesizing fully artificial genomes [59-61]. This opens up a fundamental possibility of obtaining bacteriophages with modified specificity and bacteriophages that carry genes encoding products that are toxic to bacterial cells and affect certain bacterial genes [60]. Designing of chimeric bacteriophages was described; these phages showed increased lytic activity and infected an expanded spectrum of host bacteria [62, 63]. Bacteriophages were obtained that contained genes encoding enzymes that destroyed bacterial biofilms [64]. Elements were added into the phage genome, which ensured production of the CRISPR-Cas genomic editing system in the bacterial cell to selectively break the bacterial genes responsible for drug resistance. Administering this phage triggered death of the drug-resistant bacteria, without damaging the cells of the same microorganism that did not contain the target genetic elements [65, 66].
METHODS FOR ADMINISTERING BACTERIOPHAGES INTO THE BODY
Bacteriophages can be administered into the body by various methods: oral intake, inhalation, topical use, or injection (intravenous, intramuscular, or subcutaneous at the site of injury). When administered orally, phages are exposed to an acidic environment in the stomach; to reduce the damaging effect one should first neutralize the acidic environment or place the bacteriophage product into an appropriate capsule. When administered orally, bacteriophages can be detected, although not always, in bloodstream [23, 41, 67]. Inhalation delivery of phages is usually achieved through the use of nebulizers [25, 68-70]. When administered topically, e.g., as part of creams, ointments, emulsions, or hydrogels, phages can be used to treat burns, purulent wounds in diabetic feet, acne, and other bacterial skin lesions. The easiest way is to irrigate the wound and/or a napkin with a solution of the bacteriophage product. When injected intravenously, phages enter almost all the organs and tissues. In this case, phages are cleared from the bloodstream within 60 minutes.
Bacteriophages can also be used as an antibacterial coating for implants to prevent infections that occur during implantation of prostheses in orthopedics [71-73].
TESTING BACTERIOPHAGE PRODUCTS IN ANIMAL EXPERIMENTS
Applicability of phage therapy for a wide range of infectious diseases was studied using various animal models [55, 74-77]. Unfortunately, almost all these models happened to be inconsistent with the actual models of human diseases. Bacteremia often develops in humans as a secondary infection that accompanies injuries, viral lesions, or a weakened immune system. As the infection develops, bacteria leave the initial site and form metastatic lesions in various parts of the body. Therapy begins, as a rule, when a developed infectious process is underway. In almost all the experiments described, healthy animals were infected with large doses of bacteria and, simultaneously or after a short time, were injected with bacteriophage products. In this experiment scheme the infection has not enough time to spread beyond the injection site, the animals, in fact, play the role of a test tube. The results of the experiments showed that administration of bacteriophages achieved therapeutic effect if applied immediately or shortly after infection of the animal. When phage products were administered few hours after the infection, the observed effect was in some cases noticeably weaker. An important unexpected result of the study was strong dependence of the efficacy of bacteriophage products on the dose. One could have expected that once administered into the infected animal, phages would be multiplying rapidly in the host bacteria, and a minimum dose of phages would suffice to achieve therapeutic effect. This was the possibility that was always mentioned when describing theoretical benefits of phages as therapeutics. However, in the animal experiments, good therapeutic result was observed only after administering high doses of bacteriophages, such that have not been used in known clinical studies [55, 74, 78-89].
The main results of the animal experiments are as follows: bacteriophages are harmless to mammals and effectively destroy bacteria under conditions whereby bacteria are easily reachable (table).
Experiments on using bacteriophages in animal models
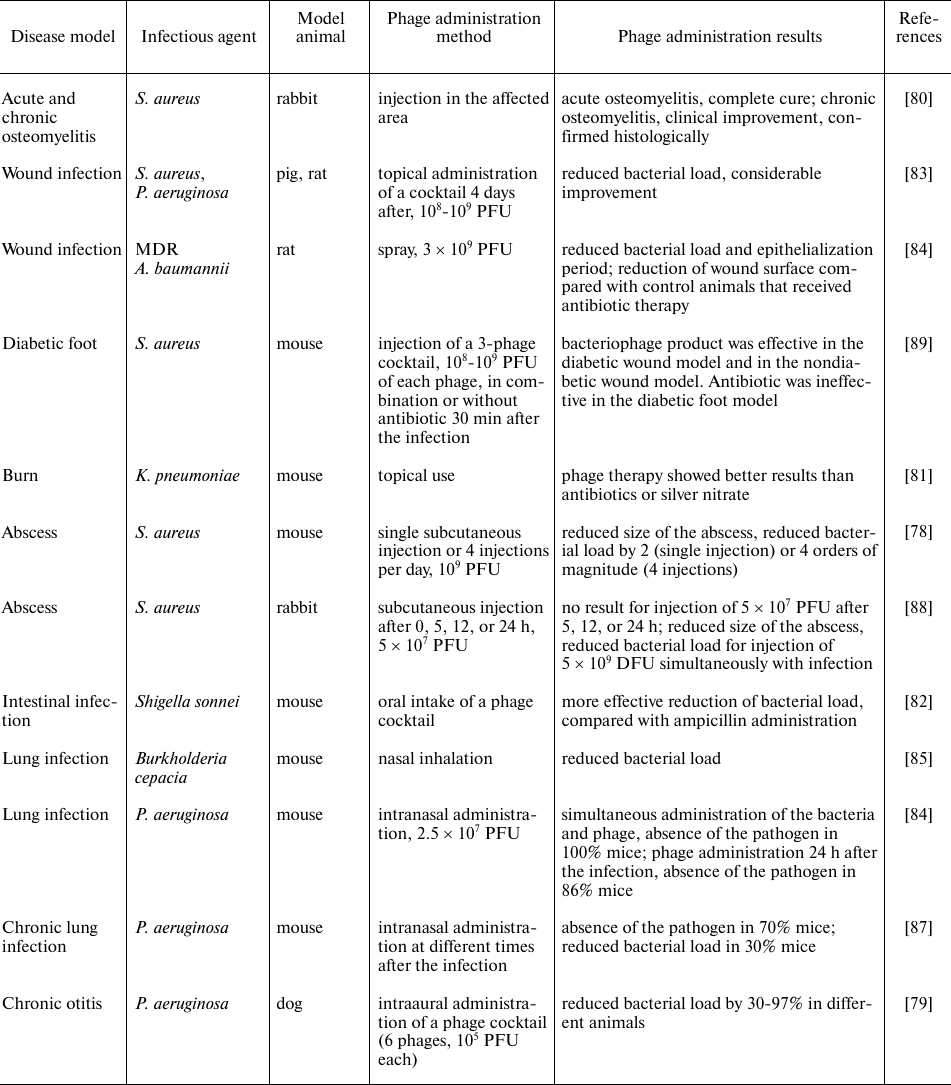
Note. PFU, plaque-forming units.
CLINICAL STUDIES OF BACTERIOPHAGE PRODUCTS AND PHAGE APPLICATIONS
IN PRACTICAL MEDICINE
Effectiveness of bacteriophage treatment has been evidenced by positive results described in the literature in the middle of the last century, when there was no practice of using placebo and control groups in clinical trials and negative results typically escaped publication [74, 90-93]. In the case of infections, however, the placebo effect can be profound.
In recent years, interest in phage therapy has intensified, partly because of the several broadly publicized cases of curing critically ill patients with bacteriophage products. However, significance of these events was exaggerated by the media; dozens of publications appeared about each patient who was saved by phages, and none about the unsuccessful cases, since the latter were of no use to journalists. Analysis of the cases of curing seriously ill patients with bacteriophage products revealed intricacies of the treatment and challenges that arose.
Thus, a case was described of successful use of the phage therapy in a 68-year-old patient suffering from diabetes and necrotizing pancreatitis complicated by a drug-resistant A. baumannii strain. Antibiotic therapy proved ineffective, and the patient received therapy with bacteriophages selected by testing bacteria isolated from the patient. Bacteriophages were injected intravenously and transdermally into the abscess. As a result of the treatment, the A. baumannii infection was suppressed, and the patient recovered [14].
There was another well-known case of curing a patient who was in a critical condition. A 15-year-old patient with cystic fibrosis and disseminated mycobacterial abscesses after lung transplantation was treated with a three-component phage cocktail. Lytic phages that effectively killed the infectious strain of Micobacterium abscessus were developed using methods of synthetic biology. Researchers noted that intravenous phage treatment was well tolerated and was accompanied by an objective clinical improvement, including closure of the sternum wound, improved liver function, and healing of inflamed skin lymph nodes [17].
In the cases described, the patients continued to receive antibiotics simultaneously with bacteriophages for ethical reasons. Long-term treatment with high doses of phages was needed. The product selection process was complicated and involved many organizations; one even had to construct phages using synthetic biology methods.
In recent years, a series of clinical trials were conducted on bacteriophage products, but not all of those studies adhered to the modern standards [94-103]. Only a few of them showed phage therapy to be clinically effective [96-98, 103]. Unfortunately, the results of most other experiments cannot be considered noteworthy. To date, the main result of the clinical trials remains that bacteriophages are indeed safe, no matter how they are administered, including by intravenous injection. One could hardly call this news, since phages have proved to be safe over many decades of their use in medicine.
In 2005, the Institute of Immunology and Experimental Therapy, Polish Academy of Sciences (Wroclaw, Poland) began clinical studies of phage therapy for bacterial infections (clinical study no. NCT00945087). The results of this study have not yet been published.
In 2009, BioContol Ltd. reported positive results of the phage therapy for ear infections caused by P. aeruginosa [98]. Later, however, these results were not mentioned anywhere.
Nestlé sponsored trials in Bangladesh of the anti-E. coli phage product for treatment of diarrhea in children [100-102]. No evidence was obtained on efficacy of the therapy. The failure was explained by the fact that only 60% of the patients had E. coli in their feces; for the rest of them, the product was useless.
Much discussion occurred around the clinical trials of a phage cocktail containing 12 bacteriophages applied in the treatment of burn wounds infected with P. aeruginosa and E. coli, the so-called Phagoburn Trial (http://www.phagoburn.eu, European Clinical Trials database, no. 2014-000714-65, and Clinical Trials gov, no. NCT02116010). The phage products were administered topically, daily for seven days. The results of the trials showed no effective therapeutic action of bacteriophages. The standard treatment with silver sulfadiazine proved to be more effective than phage therapy. The failure of these trials was attributed to instability of the phage product and insufficient concentration of phages [99].
In 2016-2017, a randomized, placebo-controlled, double-blind clinical study was conducted of the prebiotic supplement PreforPro, consisting of four bacteriophages (clinical study no. NCT03269617, funded by the University of Colorado, United States), to assess its safety and effects on gastrointestinal microbiome. During the study, data were obtained on the safety of bacteriophages and on some statistically significant changes in the gastrointestinal tract microbiome after administering the product [104, 105].
In 2017-2018, a randomized, placebo-controlled, double-blind clinical study was conducted to examine safety and efficacy of treating urinary tract infections with the adapted Pyofag product in patients undergoing transurethral resection of the prostate. The study was conducted at the National Center of Urology in Tbilisi, Georgia, and funded by Balgrist University Hospital, Switzerland (clinical study no. NCT03140085) [106]. The results confirmed safety of the product and showed decrease in the titers of infectious bacteria (from 1 to 5 log) in six out of nine patients [107].
Recently, a study was conducted at Westmead Hospital, Australia. Fourteen patients with Staphylococcus-induced infective endocarditis and sepsis were injected intravenously with the AB-SA01 phage cocktail, which included three different bacteriophages (AmpliPhi BioSciences, United States). The cocktail was administered twice a day for two weeks. As a result, researchers observed decrease in the inflammatory responses and reduced bacterial load [103]. However, there was no control group in this study. AmpliPhi Biosciences received FDA approval to conduct trials of the intravenous phage product for treatment of purulent wounds and skin infections caused by the methicillin-resistant S. aureus (MRSA) strains.
As of today, phage therapy is not licensed for large-scale use in the United States and European countries. Nevertheless, in 2018 the Center for Innovative Phage Applications and Therapeutics (IPATH) was organized in the United States to investigate and apply phage therapy as an eIND (experimental investigational new drug) at the University of California San Diego School of Medicine. In Poland, the Ludwik Hirszfeld Institute of Immunology and Experimental Therapy uses phage therapy as part of the experimental treatment in accordance with the Declaration of Helsinki (Article 37, Unproven Interventions in Clinical Practice of the Declaration of Helsinki, www.wma.net), which permits the use of unproven therapy methods in patients for which the officially approved drugs and treatments were shown to be ineffective [108]. According to the Declaration of Helsinki, bacteriophage therapy is permissible in other European countries as well, but the bureaucratic process of obtaining approval for experimental treatment is far from easy. Therefore, patients are seeking opportunities to receive phage therapy in Georgia, Poland, and more recently in the United States.
PHAGE THERAPY PROBLEMS
Phage therapy looks like an ideal personalized approach to the treatment of infectious diseases: the action principle of bacteriophages has been proven; their efficacy has been shown by model systems; reliable cases of success have been described where phage therapy saved lives or considerably improved the patient’s health. Bacteriophages are safe; they are classified by the FDA as “Generally Recognized as Safe” and may therefore undergo clinical trials in some countries. However, introduction of phages into medical practice occurs very slowly.
One of the reasons is that molecular mechanisms underlying the relations between phages and bacteria and behavior of phages in the body remain poorly understood. Phages are fundamentally different from the conventional chemotherapy drugs; they can multiply in the patient’s body depending on the concentration of the host bacteria, and they can mutate, which is not the case with conventional drugs. That is why it is difficult to determine the dosage of phage products, and unless the phage gets inside the target pathogenic bacterium, its therapeutic effect is zero. Also, we know little about pharmacokinetics and pharmacodynamics of bacteriophages. Optimal dosage forms of phage products to treat various types of infections have yet to be determined.
Practical questions remain: What should be the approved phage-based product? Should it contain only specific bacteriophages with a precisely calibrated composition, which have passed the entire set of trials, the same way as chemical drugs? Or should the formulation include bacteriophages or bacteriophage cocktails tailored to a specific patient? What should the standard procedure for selecting bacteriophage product for the treatment of specific patients affected by certain bacterial strains involve? What should be the standardized techniques for bacteriophage purification to ensure their sterility and safety?
A major task is to develop platforms for quick identification of the pathogenic bacteria and their sensitivity to bacteriophage product. Currently, this process takes at least three to five days, which in many cases is unacceptably long, making it easier for the attending physician to immediately prescribe a broad-spectrum antibiotic, which will be effective against the infectious agent in 50% of the cases.
That is why great hopes are pinned on the use of synthetic biology methods to obtain bacteriophages with desired properties such as expanded specificity, increased lytic activity, and ability to infect only those bacteria that contain target genetic elements, such as drug resistance genes. Several works have been published that show potential of synthetic biology to obtain phages with altered individual functions and characteristics. However, the problems that stand in the way of creating therapeutic bacteriophages are profound and plentiful. Thus, some see an outstanding example of designing highly specific bacteriophages in works where the phages were equipped with genes that encode the genomic editing systems that disrupt specific bacterial genes only [109-111]. However, equipping phages with these high-precision weapons does not solve the problem of creating a therapeutic drug. Targeting specific bacterial strains has never been an issue. On the contrary, it is the narrow specificity of bacteriophages that is a problem.
On top of the problems associated with in-depth research of bacteriophages, there is also a number of economic and bureaucratic barriers that hinder widespread introduction of phage therapy.
Pharmaceutical industry shows little enthusiasm in bacteriophages because of intellectual property concerns. Moreover, narrow specificity of the phage products and development of phage resistance in bacteria can often render it impossible to use an unchanged standard phage product for a long time. Most often, bacteriophages are needed in critical cases only, for a relatively narrow circle of patients for whom all the existing antibiotics shown ineffective. According to the experts, only a few thousand such cases occur in the United States annually. Therefore, pharmaceutical companies see no economic benefit in manufacturing phage products.
The development of phage therapy has stumbled against another crucial roadblock, namely that regulatory authorities in most countries impose same requirements on the phage products as on chemical drugs. Since bacteriophages are not substances with a strictly fixed, unchanging structure, they in principle cannot meet these requirements. In addition, although bacteriophages do not infect human cells, they still are viruses. This word alone makes medical authorities nervous and general public too. When discussing the possibility of using bacteriophages as therapeutic agents, they raise fringe concerns, refuted by the long-term clinical use of bacteriophages. They reiterate worries about the possibility of bacteriophages transferring fragments of genetic material between bacteria, although the human body contains a huge variety of different bacteriophages, and administering another related phage cannot create any additional problems. Especially since therapeutic phages are deprived of systems for the transfer of genetic elements.
Insistence of the regulating authorities on the standard could be, to some extent, met by products with a fixed composition, i.e., cocktails of certain thoroughly studied bacteriophages. However, these products are not flexible enough to achieve efficacy against constantly mutating bacterial strains. In fact, composition of the phage product could be altered in the course of production to adapt to newly emerging strains of infectious agents. Recent advances in the treatment of terminal patients with phage products showed that phage therapy succeeded only when personalized phage cocktails or even when specially constructed phages were used [14, 17]. This type of phage therapy, too, is not compatible with the existing regulations.
It should be noted that there is a case of successful application of simplified drug approval procedures. By that we mean production of the influenza vaccines, which require regular modification.
In this situation, several countries are making efforts to develop special rules for bacteriophage products. Belgium is now taking steps to introduce a scheme whereby phages are used as magistral preparations [112], which pharmacists can compose themselves from the standard bacteriophages, following prescription of the attending physician. Bacteriophages in these preparations must meet the requirements of pharmacopoeia. However, these magistral preparations may include, in addition to standard bacteriophages, unauthorized ones if the latter have a certificate issued by a laboratory accredited with the Belgian regulatory bodies. Magistral phage preparations can also be used to treat patients, and responsibility for their use lies with the attending physician and/or the pharmacist. In Georgia, personalized phage products can also be made as magistral preparations at pharmacies licensed by the Georgian Ministry of Health.
In Russia, a country that used to be a leader in the production and application of bacteriophages, today only the phage products manufactured by NPO Mikrogen are sold in the pharmacy network and used as pharmacotherapeutic medical immunobiological preparations according to the State Register of the Ministry of Health of the Russian Federation. Products of other manufacturers are not considered medicinal drugs. Phage therapy in Russia is not developing because the authorities do not allow the use of personalized phage therapy methods and the Mikrogen bacteriophage products are not on the list of vital and essential drugs for medical use. Furthermore, the standard products manufactured by Russian companies cannot deliver the main benefit of phage therapy, i.e., the personalized approach.
Obviously, the bureaucratic barriers must be lifted in the interests of patients. Bacteriophages pose no danger to humans, as proven by their century-long use in medicine.
CONCLUSIONS
Phage therapy obviously offers much promise for treating bacterial infections. Its capabilities manifested themselves most conspicuously in personalized applications, when patients who were not responding to antibiotic therapy received phage cocktails tailored to their specific needs.
Bacteriophages cannot replace antibiotics, which are inexpensive, have a wide specificity, and turn out to be effective in a plenty of cases; therefore, antibiotics will remain the first-choice drugs for treating infectious diseases. However, phages are capable of dealing with severe infections, which do not respond to antibiotic therapy, and they enable personalized therapy in patients that preserve their normal microflora. Phage therapy cannot completely solve the problem of antibiotic-resistant infectious agents. The most promising applications of phage therapy are associated with the treatment of infectious lesions that do not respond to antibiotic therapy, where phages are best administered topically. In addition, phage therapy could be useful in cases where antibiotics could damage microbiota of the patient due to the broad spectrum of their action or negatively affect the already weakened patient with a serious illness. Phages can be used successfully in places where they could be easily transported and from where they cannot be quickly eliminated.
Development of the effective bacteriophage products and treatment strategies requires extensive fundamental research. We need to investigate thoroughly the bacterium–phage coevolution mechanisms; study pharmacodynamics and pharmacokinetics of the phages in the body; develop methods for constructing bacteriophages with desired properties using synthetic biology methods; and design technologies for obtaining stable bacteriophage products and means of their delivery. Full-fledged clinical trials need to be conducted to clarify issues related to dosages, administration methods, and possible therapeutic applications of bacteriophage products.
Broad international cooperation is necessary to create enabling conditions for quick identification and delivery of bacteriophages required for treating individual patients.
The existing approaches to certification of the therapeutic drugs cannot be applied to bacteriophage products. They completely block the road for the development of phage therapy, best implemented as personalized treatment, which involves selection of bacteriophages for the treatment of a specific patient infected with the specific bacteria. The task of successful development of phage therapy, as well as other therapies based on viruses, cells, and cellular products, necessitates creation of the new principles for licensing such procedures.
Funding. This work was financially supported by the Russian Foundation for Basic Research (project no. 18-29-08015).
Ethics declarations. The authors declare no conflict of interest in financial or any other sphere. This article does not contain any studies with human participants or animals performed by any of the authors.
REFERENCES
1.D’Herrelle, F. (1935) Bakteriofag i
fenomen vyzdorovleniya (The Bacteriophage and the Phenomenon of
Cure), Tiflis Gos. Univ., Tiflis.
2.Chanishvili, N. (2012) Phage therapy –
history from Twort and d’Herelle through soviet experience to
current approaches, Adv. Virus Res., 83, 4-40.
3.Pokrovskaya, M. P., Kaganova, L. S., Morozenko, M.
A., Bulgakova, A. G., and Skatsenko, E. E. (1941) Lechenie ran
bakteriofagami (Treatment of Wounds with Bacteriophages) Medgiz,
Moscow, Leningrad.
4.Tsulukidze, A. P. (1941) Revisiting bacteriophage
application techniques in surgical practice, Vest. Khirurga,
6, 679-685.
5.Kokin, G. A. (1941) The use of bacteriophages in
surgery, Sov. Meditsina, 9, 15-18.
6.Sapir, I. B. (1939) Nablyudeniya i zamechaniya
po povodu lecheniya dizenterii bakteriofagom (Observations and
Remarks on the Treatment of Dysentery with Bacteriophage) Mosk. Obl.
Inst. Infekts. Bolezn., Moscow.
7.Belikova, M. A. (1941) Experience in using phages
to prevent dysentery in the infants and young children of Stalingrad,
Zh. Mikrobiol. Epidemiol. Immunol., 5-6, 168-183.
8.Ermol’eva, Z. V. (1942) Kholera
(Cholera), Medgiz, Moscow.
9.Zhukov-Verezhnikov, N. N., Permitina, L. D.,
Berillo, E. A., Komissarov, V. P., Bardymov, V. M., Khvoles, A. G., and
Ugryumov, L. B. (1978) Study of the therapeutic effect of bacteriophage
preparations in the complex treatment of purulent surgical diseases,
Sov. Meditsina, 12, 64-66.
10.Kochetkova, V. A., Mamontov, A. S., Moskovtseva,
R. L., Erastova, E. I., Trofimov, E. I., Popov, M. I., and Dzhubalieva,
S. K. (1989) Phage therapy of postoperative pyoinflammatory
complications in cancer patients, Sov. Meditsina, 6,
23-26.
11.Bryusov, P. G., Zubritskii, V. F., Islamov, R.
N., Nizovoi, A. V., and Fominykh, V. M. (2011) Phage prophylaxis and
phage therapy of surgical infections, Voen. -Med. Zhurn.,
4, 34-39.
12.Khairullin, I. N., Pozdeev, O. K., and
Shaimardanov, R. Sh. (2002) Efficacy of specific bacteriophages in the
treatment and prevention of surgical and postoperative infections,
Kazan. Med. Zhurn., 83, 258-261.
13.Morozova, V. V., Kozlova, Y. N., Ganichev, D. A.,
and Tikunova, N. V. (2018) Bacteriophage treatment of infected diabetic
foot ulcers, Methods Mol. Biol., 1693, 151-158, doi:
10.1007/978-1-4939-7395-8_13.
14.Schooley, R. T., Biswas, B., Gill, J. J.,
Hernandez-Morales, A., and Lancaster, J. (2017) Development and use of
personalized bacteriophage-based therapeutic cocktails to treat a
patient with a disseminated resistant Acinetobacter baumannii
infection, Antimicrob. Agents Chemother., 61, e00954-17,
doi: 10.1128/AAC.00954-17.
15.Chan, B. K., Turner, P. E., Kim, S., Mojibian, H.
R., Elefteriades, J. A., and Narayan, D. (2018) Phage treatment of an
aortic graft infected with Pseudomonas aeruginosa, Evol. Med.
Public Health., 1, 60‐66, doi:
10.1093/emph/eoy005.
16.Onsea, J., Soentjens, P., Djebara, S.,
Merabishvili, M., Depypere, M., et al. (2019) Bacteriophage application
for difficult-to-treat musculoskeletal infections: development of a
standardized multidisciplinary treatment protocol, Viruses,
11, 891, doi: 10.3390/v11100891.
17.Dedrick, R. M., Guerrero-Bustamante, C. A.,
Garlena, R. A., Russell, D. A., Ford, K., et al. (2019) Engineered
bacteriophages for treatment of a patient with a disseminated
drug-resistant Mycobacterium abscessus, Nat. Med.,
25, 730‐733, doi: 10.1038/s41591-019-0437-z.
18.Dutilh, B. E., Cassman, N., McNair, K., Sanchez,
S. E., Silva, G. G., et al. (2014) A highly abundant bacteriophage
discovered in the unknown sequences of human faecal metagenomes,
Nat. Commun., 5, 4498, doi: 10.1038/ncomms5498.
19.Sausset, R., Petit, M. A., Gaboriau-Routhiau, V.,
and De Paepe, M. (2020) New insights into intestinal phages, Mucosal
Immunol., 13, 205‐215, doi:
10.1038/s41385-019-0250-5.
20.Górski, A., Wazna, E., Dąbrowska, B.
W., Dąbrowska, K., Switała-Jeleń, K., and
Miedzybrodzki, R. (2006) Bacteriophage translocation, FEMS Immunol.
Med. Microbiol., 46, 313-319, doi:
10.1111/j.1574-695X.2006.00044.x.
21.Nguyen, S., Baker, K., Padman, B. S., Patwa, R.,
Dunstan, R. A., et al. (2017) Bacteriophage transcytosis provides a
mechanism to cross epithelial cell layers, mBio., 8,
e01874-17, doi: 10.1128/mBio.01874-17.
22.Fish, R., Kutter, E., Bryan, D., Wheat, G., and
Kuhl, S. (2018) Resolving digital Staphylococcal osteomyelitis using
bacteriophage – a case report, Antibiotics (Basel),
7, 87, doi: 10.3390/antibiotics7040087.
23.Huh, H., Wong, S., St Jean, J., and Slavcev, R.
(2019) Bacteriophage interactions with mammalian tissue: therapeutic
applications, Adv. Drug Deliv. Rev., 145, 4‐17,
doi: 10.1016/j.addr.2019.01.003.
24.Akturk, E., Oliveira, H., Santos, S. B., Costa,
S., Kuyumcu, S., Melo, L. D. R., and Azeredo, J. (2019) Synergistic
action of phage and antibiotics: parameters to enhance the killing
efficacy against mono and dual-species biofilms, Antibiotics
(Basel), 8, 103, doi: 10.3390/antibiotics8030103.
25.Lin, Y., Chang, R., Britton, W. J., Morales, S.,
Kutter, E., and Chan, H. K. (2018) Synergy of nebulized phage PEV20 and
ciprofloxacin combination against Pseudomonas aeruginosa, Int. J.
Pharm., 551, 158-165, doi:
10.1016/j.ijpharm.2018.09.024.
26.Samson, J. E., Magadán, A. H., Sabri, M.,
and Moineau, S. (2013) Revenge of the phages: defeating bacterial
defences, Nat. Rev. Microbiol., 11, 675‐687, doi:
10.1038/nrmicro3096.
27.Basu, S., Agarwal, M., Bhartiya, S. K., Nath, G.,
and Shukla, V. (2015) An in vivo wound model utilizing
bacteriophage therapy of Pseudomonas aeruginosa biofilms,
Ostomy Wound Manage., 61, 16-23.
28.Fong, S. A., Drilling, A., Morales, S., Cornet,
M. E., Woodworth, B. A., et al. (2017) Activity of bacteriophages in
removing biofilms of Pseudomonas aeruginosa isolates from
chronic rhinosinusitis patients, Front. Cell. Infect.
Microbiol., 7, 418, doi: 10.3389/fcimb.2017.00418.
29.Chan, B. K., Abedon, S. T., and Loc-Carrillo, C.
(2013) Phage cocktails and the future of phage therapy, Future
Microbiol., 8, 769‐783, doi: 10.2217/fmb.13.47.
30.Yuan, Y., Wang, L., Li, X., Tan, D., Cong, C.,
and Xu, Y. (2019) Efficacy of a phage cocktail in controlling phage
resistance development in multidrug resistant Acinetobacter
baumannii, Virus Res., 272, 197734, doi:
10.1016/j.virusres.2019.197734.
31.Górski, A., Międzybrodzki, R.,
Łobocka, M., Głowacka-Rutkowska, A., Bednarek, A., et al.
(2018) Phage therapy: what have we learned? Viruses, 10,
288, doi: 10.3390/v10060288.
32.Łusiak-Szelachowska, M., Zaczek, M., and
Weber-Dąbrowska, B. (2014) Phage neutralization by sera of
patients receiving phage therapy, Viral Immunol., 27,
295‐304, doi: 10.1089/vim.2013.0128.
33.Majewska, J., Beta, W., Lecion, D.,
Hodyra-Stefaniak, K., Kłopot, A., et al. (2015) Oral application
of T4 phage induces weak antibody production in the gut and in the
blood, Viruses, 7, 4783-99, doi: 10.3390/v7082845.
34.Hodyra-Stefaniak, K., Miernikiewicz, P.,
Drapała, J., Drab, M., Jończyk-Matysiak, E. J., et al.
(2015) Mammalian host-versus-phage immune response determines phage
fate in vivo, Sci. Rep., 5, 14802, doi:
10.1038/srep14802.
35.Górski, A., Międzybrodzki, R.,
Borysowski, J., Dąbrowska, K., Wierzbicki, P., et al. (2012)
Phage as a modulator of immune responses: practical implications for
phage therapy, Adv. Virus Res., 83, 41‐71, doi:
10.1016/B978-0-12-394438-2.00002-5.
36.Górski, A., Międzybrodzki, R.,
Jończyk-Matysiak, E., Żaczek, M., and Borysowski, J.
(2019) Phage-specific diverse effects of bacterial viruses on the
immune system, Future Microbiol., 14, 1171‐1174,
doi: 10.2217/fmb-2019-0222.
37.Sinha, A., and Maurice, C. F. (2019)
Bacteriophages: uncharacterized and dynamic regulators of the immune
system, Mediators Inflamm., 2019, 3730519, doi:
10.1155/2019/3730519.
38.Van Belleghem, J. D., Dąbrowska, K.,
Vaneechoutte, M., Barr, J., and Bollyky, P. (2018) Interactions between
bacteriophage, bacteria, and the mammalian immune system,
Viruses, 11, 10, doi: 10.3390/v11010010.
39.Dąbrowska, K., Miernikiewicz, P.,
Piotrowicz, A., Hodyra, K., Owczarek, B., et al. (2014) Immunogenicity
studies of proteins forming the T4 phage head surface, J.
Virol., 88, 12551-12557, doi: 10.1128/JVI.02043-14.
40.Zaczek, M., Łusiak-Szelachowska, M.,
Jończyk-Matysiak, E., Weber-Dąbrowska, B., Miedzybrodzki,
R., et al. (2016) Antibody production in response to staphylococcal
MS-1 phage cocktail in patients undergoing phage therapy, Front.
Microbiol., 7, 1-14, doi: 10.3389/fmicb.2016.01681.
41.Dąbrowska, K. (2019) Phage therapy: what
factors shape phagepharmacokinetics and bioavailability? Systematic and
critical review, Med. Res. Rev., 1, 1-26, doi:
10.1002/med.21572.
42.Bochkareva, S. S., Aleshkin, A. V., Ershova, O.
N., Novikova, L. I., Karaulov, A. V., Kiseleva, I. A., et al. (2017)
Anti-phage antibody response in phage therapy against
healthcare-associated infections (HAIs), Infektsionnye Bolezn.,
15, 35-40, doi: 10.20953/1729-9225-2017-1-35-40.
43.Weber-Dąbrowska, B., Zimecki, M., and
Mulczyk, M. (2000) Effective phage therapy is associated with
normalization of cytokine production by blood cell cultures, Arch.
Immunol. Ther. Exp. (Warsz), 48, 31-37.
44.Roach, D. R., Chollet-martin, S., Noël, B.,
and Granger, V. (2019) Human neutrophil response to Pseudomonas
3 bacteriophages, BioRxiv, doi: 10.1101/786905.
45.Roach, D. R., Leung, C. Y., Henry, M., Morello,
E., Singh, D., Di Santo, J. P., Weitz, J. S., and Debarbieux, L. (2017)
Synergy between the host immune system and bacteriophage is essential
for successful phage therapy against an acute respiratory pathogen,
Cell Host Microbe, 22, 38-47.e4, doi:
10.1016/j.chom.2017.06.018.
46.Dickey, J., and Perrot, V. (2019) Adjunct phage
treatment enhances the effectiveness of low antibiotic concentration
against Staphylococcus aureus biofilms in vitro, PLoS
One, 14, e0209390, doi: 10.1371/journal.pone.0209390.
47.Rodriguez-Gonzalez, R. A., Leung, C. Y., Chan, B.
K., Turner, P. E., and Weitz, J. S. (2020) Quantitative models of
phage-antibiotic combination therapy, mSystems, 5,
e00756-19, doi: 10.1128/mSystems.00756-19.
48.Poglazov, B. F. (1973) Morphogenesis of T-even
bacteriophages, Karger Publishers.
49.Letarov, A. V. (2019) Sovremennye kontsepsii
biologii bakteriofagov, DeLi, Moscow.
50.Tikunova, N. V., Voroshilova, N. N., Polygach, O.
A., Morozova, V. V., Tikunov, A. Yu., Kuril’shchikov, A. M., and
Vlasov, V. V. (2016) Genetic characteristics and antibacterial
spectrume of bacteriophages in the industrial series of the product
Piobakteriofag polyvalent purified, Epidemiol. Vaktsinoprof.,
15, 93-100, doi: 10.31631/2073-3046-2016-15-2-93-100.
51.Polygach, O. A., Voroshilova, N. N., Tikunova, N.
V., Morozova, V. V., Tikunov, A. Yu., Krylov, V. N., Yunusova, A. A.,
and Dabizheva, A. N. (2018) Modern approaches to developing the phage
base of the medicinal and preventive product of Pseudomonas
aeruginosa bacteriophages, Epidemiol. Vaktsinoprof.,
2, 37-45, doi: 10.24411/2073-3046-2018-10004.
52.Malik, D. J., Sokolov, I. J., Vinner, G. K.,
Mancuso, F., Cinquerrui, S., et al. (2017) Formulation, stabilisation
and encapsulation of bacteriophage for phage therapy, Adv. Colloid
Interface Sci., 249, 100‐133, doi:
10.1016/j.cis.2017.05.014.
53.Manohar, P., and Ramesh, N. (2019) Improved
lyophilization conditions for long-term storage of bacteriophages,
Sci. Rep., 9, 15242, doi: 10.1038/s41598-019-51742-4.
54.Chadha, P., Katare, O. P., and Chhibber, S.
(2017) Liposome loaded phage cocktail: Enhanced therapeutic potential
in resolving Klebsiella pneumoniae mediated burn wound
infections, Burns, 43, 1532‐1543, doi:
10.1016/j.burns.2017.03.029.
55.Chang, R. Y. K., Wallinb, M., Lina, Y., Leung, S.
S. Y., Wang, Y., Morales, S., and Chan, H.-K. (2018) Phage therapy for
respiratory infections, Adv. Drug Deliv. Rev., 133,
76‐86, doi: 10.1016/j.addr.2018.08.001.
56.Otero, J., García-Rodríguez, A.,
Cano-Sarabia, M., Maspoch, D., Marcos, R., Cortés, P., and
Llagostera, M. (2019) Biodistribution of liposome-encapsulated
bacteriophages and their transcytosis during oral phage therapy,
Front. Microbiol., 10, 689, doi:
10.3389/fmicb.2019.00689.
57.Singla, S., Harjai, K., Katare, O. P., and
Chhibber, S. (2016) Encapsulation of bacteriophage in liposome
accentuates its entry in to macrophage and shields it from neutralizing
antibodies, PLoS One, 11, e0153777, doi:
10.1371/journal.pone.0153777.
58.Kim, K. P., Cha, J. D., Jang, E. H., Klumpp, J.,
Hagens, S., Hardt, W.-D., Lee, K.-J., Loessner, M. J. (2008) PEGylation
of bacteriophages increases blood circulation time and reduces T-helper
type 1 immune response, Microb. Biotechnol., 1,
247‐257, doi: 10.1111/j.1751-7915.2008.00028.x.
59.Rita Costa, A., Milho, C., Azeredo, J., and
Pires, D. P. (2018) Synthetic biology to engineer bacteriophage
genomes, Methods Mol. Biol., 1693, 285‐300, doi:
10.1007/978-1-4939-7395-8_21.
60.Pires, D. P., Cleto, S., Sillankorva, S.,
Azeredo, J., and Lu, T. K. (2016) Genetically engineered phages: a
review of advances over the last decade, Microbiol. Mol. Biol.
Rev., 80, 523‐543, doi: 10.1128/MMBR.00069-15.
61.Ando, H., Lemire, S., Pires, D. P., and Lu, T. K.
(2015) Engineering modular viral scaffolds for targeted bacterial
population editing, Cell Syst., 1, 187‐196, doi:
10.1016/j.cels.2015.08.013.
62.Lu, T. K., and Koeris, M. S. (2011) The next
generation of bacteriophage therapy, Curr. Opin. Microbiol.,
14, 524‐531, doi: 10.1016/j.mib.2011.07.028.
63.Kilcher, S., Studer, P., Muessner, C., Klumpp,
J., and Loessner, M. J. (2018) Cross-genus rebooting of custom-made,
synthetic bacteriophage genomes in L-form bacteria, Proc. Natl.
Acad. Sci. USA, 115, 567‐572, doi:
10.1073/pnas.1714658115.
64.Lu, T. K., and Collins, J. J. (2007) Dispersing
biofilms with engineered enzymatic bacteriophage, Proc. Natl. Acad.
Sci. USA, 104, 11197‐11202, doi:
10.1073/pnas.0704624104.
65.Cobb, L. H., Park, J., Swanson, E. A., Beard, M.
C., McCabe, E. M., et al. (2019) CRISPR-Cas9 modified bacteriophage for
treatment of Staphylococcus aureus induced osteomyelitis and
soft tissue infection, PLoS One, 14, e0220421, doi:
10.1371/journal.pone.0220421.
66.Yehl, K., Lemire, S., Yang, A. C., Ando, H.,
Mimee, M., et al. (2019) Engineering phage host-range and suppressing
bacterial resistance through phage tail fiber mutagenesis, Cell,
179, 459‐469, doi: 10.1016/j.cell.2019.09.015.
67.Barr, J. J. (2017) A bacteriophages journey
through the human body, Immunol. Rev., 279,
106‐122, doi: 10.1111/imr.12565.
68.Cooper, C. J., Denyer, S. P., and Maillard, J. Y.
(2014) Stability and purity of a bacteriophage cocktail preparation for
nebulizer delivery, Lett. Appl. Microbiol., 58,
118‐122, doi: 10.1111/lam.12161.
69.Carrigy, N. B., Chang, R.Y., Leungm, S. S. Y.,
Harrison, M., Petrova, Z., et al. (2017) Anti-tuberculosis
bacteriophage D29 delivery with a vibrating mesh nebulizer, jet
nebulizer, and soft mist inhaler, Pharm. Res., 34,
2084-2096, doi: 10.1007/s11095-017-2213-4.
70.Astudillo, A., Leung, S. S. Y., Kutter, E.,
Morales, S., and Chan, H. K. (2018) Nebulization effects on structural
stability of bacteriophage PEV 44, Eur. J. Pharm. Biopharm.,
125, 124‐130, doi: 10.1016/j.ejpb.2018.01.010.
71.Samokhin, A. G., Kozlova, Yu. N., Korneev, D. V.,
Taranov, O. S., Fedorov, E. A., Pavlov, V. V., Morozova, V. V., and
Tikunova, N. V. (2018) Experimental study of the antibacterial activity
of the lytic Staphylococcus aureus bacteriophage ph20 and lytic
Pseudomonas aeruginosa bacteriophage ph57 during modelling of
its impregnation into poly(methylmetacrylate) orthopedic implants (bone
cement), Vestn. Ross. Akad. Med. Nauk, 73, 59-68, doi:
10.15690/vramn905.
72.Aleshkin, A. V., Zul’karneev, E. R.,
Kiseleva, I. A., Emel’yanenko, K. A., Emel’yanenko, A. M.,
and Boinovich, L. B. (2018) Experimental application of
organic–inorganic hybrid coatings with adsorbed bacteriophages
for reducing the risk of STEC infections, Byull. Experiment. Biol.
Med., 165, 473-476.
73.Morris, J., Kelly, N., Elliott, L., Grant, A.,
Wilkinson, M., Hazratwala, K., and McEwen, P. (2019) Evaluation of
bacteriophage anti-biofilm activity for potential control of orthopedic
implant-related infections caused by Staphylococcus aureus,
Surg. Infect. (Larchmt), 20, 16-24, doi:
10.1089/sur.2018.135.
74.Gelman, D., Eisenkraft, A., Chanishvili, N.,
Nachman, D., Glazer, C. S., and Hazan, R. (2018) The history and
promising future of phage therapy in the military service, J. Trauma
Acute Care Surg., 85, S18-S26, doi:
10.1097/TA.0000000000001809.
75.Kortright, K. E., Chan, B. K., Koff, J. L., and
Turner, P. E. (2019) Phage therapy: a renewed approach to combat
antibiotic-resistant bacteria, Cell Host Microbe, 25,
219-232, doi: 10.1016/j.chom.2019.01.014.
76.Abedon, S. T. (2019) Use of phage therapy to
treat long-standing, persistent, or chronic bacterial infections,
Adv. Drug Deliv. Rev., 145, 18‐39, doi:
10.1016/j.addr.2018.06.018.
77.Principi, N., Silvestri, E., and Esposito, S.
(2019) Advantages and limitations of bacteriophages for the treatment
of bacterial infections, Front. Pharmacol., 10, 513, doi:
10.3389/fphar.2019.00513.
78.Capparelli, R., Parlato, M., Borriello, G.,
Salvatore, P., and Iannelli, D. (2007) Experimental phage therapy
against Staphylococcus aureus in mice, Antimicrob. Agents
Chemother., 51, 2765-2773.
79.Hawkins, C., Harper, D., Burch, D., Anggard, E.,
and Soothill, J. (2010) Topical treatment of Pseudomonas aeruginosa
otitis of dogs with a bacteriophage mixture: a before/after clinical
trial, Vet. Microbiol., 145, 309-313.
80.Kishor, C., Mishra, R. R., Saraf, S. K., Kumar,
M., Srivastav, A. K., and Nath, G. (2016) Phage therapy of
staphylococcal chronic osteomyelitis in experimental animal model,
Indian J. Med. Res., 143, 87-94.
81.Kumari, S., Harjai, K., and Chhibber, S. (2011)
Bacteriophage versus antimicrobial agents for the treatment of murine
burn wound infection caused by Klebsiella pneumoniae B5055,
J. Med. Microbiol., 60, 205-210.
82.Mai, V., Ukhanova, M., Reinhard, M. K., Li, M.,
and Sulakvelidze, A. (2015) Bacteriophage administration significantly
reduces Shigella colonization and shedding by Shigella-challenged mice
without deleterious side effects and distortions in the gut microbiota,
Bacteriophage, 5, e1088124.
83.Mendes, J. J., Leandro, C., Corte-Real, S.,
Barbosa, R., Cavaco-Silva, P., Melo-Cristino, J., Gorski, A., and
Garcia, M. (2013) Wound healing potential of topical bacteriophage
therapy on diabetic cutaneous wounds, Wound Repair Regen.,
21, 595-603.
84.Pabary, R., Singh, C., Morales, S., Bush, A.,
Alshafi, K., Bilton, D., Alton, E. W., Smithyman, A., and Davies, J. C.
(2016) Antipseudomonal bacteriophage reduces infective burden and
inflammatory response in murine lung, Antimicrob. Agents
Chemother., 60, 744-751.
85.Semler, D. D., Goudie, A. D., Finlay, W. H., and
Dennis, J. J. (2014) Aerosol phage therapy efficacy in Burkholderia
cepacia complex respiratory infections, Antimicrob. Agents
Chemother., 58, 4005-4013.
86.Shivaswamy, V. C., Kalasuramath, S. B., Sadanand,
C. K., Basavaraju, A. K., Ginnavaram, V., Bille, S., Ukken, S. S., and
Pushparaj, U. N. (2015) Ability of bacteriophage in resolving wound
infection caused by multidrug-resistant Acinetobacter baumannii
in uncontrolled diabetic rats, Microb. Drug Resist., 21,
171-177.
87.Waters, E. M., Neill, D. R., Kaman, B., Sahota,
J. S., Clokie, M. R., Winstanley, C., and Kadioglu, A. (2017) Phage
therapy is highly effective against chronic lung infections with
Pseudomonas aeruginosa, Thorax, 72, 666-667.
88.Wills, Q. F., Kerrigan, C., and Soothill, J. S.
(2005) Experimental bacteriophage protection against Staphylococcus
aureus abscesses in a rabbit model, Antimicrob. Agents
Chemother., 49, 1220-1221.
89.Albac, S., Medina, M., Labrousse, D., Hayez, D.,
Bonnot, D., et al. (2020) Efficacy of bacteriophages in a
Staphylococcus aureus nondiabetic or diabetic foot infection
murine model, Antimicrob. Agents Chemother., 64,
e01870-19, doi: 10.1128/AAC.01870-19.
90.Morozova, V. V., Vlassov, V. V., and Tikunova, N.
V. (2018) Applications of bacteriophages in the treatment of localized
infections in humans, Front. Microbiol., 9, 1696, doi:
10.3389/fmicb.2018.01696.
91.Sulakvelidze, A., Alavidze, Z., and Morris, J. G.
(2001) Bacteriophage therapy, Antimicrob. Agents Chemother.,
45, 649-659, doi: 10.1128/AAC.45.3.649-659.2001.
92.Chanishvili, N. (2009) A Literature Review of
the Practical Application of Bacteriophage Research, Nova Science
Publishers, New York.
93.Chanishvili, N. (2016) Bacteriophages as
therapeutic and prophylactic means: summary of the soviet and
post-soviet experiences, Curr. Drug Deliv., 13, 309-323,
doi: 10.2174/156720181303160520193946.
94.Schmidt, C. (2019) Phage therapy’s latest
makeover, Nat. Biotechnol., 37, 58-586, doi:
10.1038/s41587-019-0133-z.
95.Altamirano, F., and Barr, J. (2019) Phage therapy
in the postantibiotic era, Clin. Microbiol. Rev., 32,
e00066-18, doi: 10.1128/CMR.00066-18.
96.Rhoads, D. D., Wolcott, R. D., Kuskowski, M. A.,
Wolcott, B. M., Ward, L. S., and Sulakvelidze, A. (2009) Bacteriophage
therapy of venous leg ulcers in humans: results of a phase I safety
trial, J. Wound Care, 18, 240-243.
97.Markoishvili, K., Tsitlanadze, G., Katsarava, R.,
Morris, J. G., and Sulakvelidze, A. (2002) A novel sustained-release
matrix based on biodegradable poly(ester amide) s and impregnated with
bacteriophages and an antibiotic shows promise in management of
infected venous stasis ulcers and other poorly healing wounds, Int.
J. Dermatol., 41, 453-458, doi:
10.1046/j.1365-4362.2002.01451.x.
98.Wright, A., Hawkins, C. H., Anggård, E. E.,
and Harper, D. R. (2009) A controlled clinical trial of a therapeutic
bacteriophage preparation in chronic otitis due to antibiotic-resistant
Pseudomonas aeruginosa; a preliminary report of efficacy, Clin.
Otolaryngol., 34, 349-357, doi:
10.1111/j.1749-4486.2009.01973.x.
99.Jault, P., Leclerc, T., Jennes, S., Pirnay, J.
P., Que, Y. A., et al. (2018) Efficacy and tolerability of a cocktail
of bacteriophages to treat burn wounds infected by Pseudomonas
aeruginosa (PhagoBurn): a randomised, controlled, double-blind
phase 1/2 trial, Lancet Infect. Dis., 19, 35-45, doi:
10.1016/S1473-3099(18)30482-1.
100.Sarker, S. A., McCallin, S., Barretto, C.,
Berger, B., Pittet, A. C., et al. (2012) Oral T4-like phage cocktail
application to healthy adult volunteers from Bangladesh,
Virology, 434, 222-232, doi:
10.1016/j.virol.2012.09.002.
101.Sarker, S. A., Sultana, S., Reuteler, G.,
Moine, D., Descombes, P., Charton, F., et al. (2016) Oral phage therapy
of acute bacterial diarrhea with two coliphage preparations: a
randomized trial in children from Bangladesh, EBioMedicine,
4, 124-137, doi: 10.1016/j.ebiom.2015.12.023.
102.McCallin, S., Sarker, S. A., and Barretto, C.
(2013) Safety analysis of a Russian phage cocktail: from metagenomic
analysis to oral application in healthy human subjects,
Virology, 443, 187-196, doi:
10.1016/j.virol.2013.05.022.
103.Fabijan, A. P., Lin, R. C. Y., Ho, J.,
Maddocks, S., Ben Zakour, N. L., and Iredell, J. R. (2020) Safety of
bacteriophage therapy in severe Staphylococcus aureus infection,
Nat. Microbiol., 5, 465-472, doi:
10.1038/s41564-019-0634-z.
104.Febvre, H. P., Rao, S., Gindin, M., Goodwin, N.
D. M., Finer, E., et al. (2019) PHAGE study: effects of supplemental
bacteriophage intake on inflammation and gut microbiota in healthy
adults, Nutrients, 11, E666, doi: 10.3390/nu11030666.
105.Gindin, M., Febvre, H. P., Rao, S., Wallace, T.
C., and Weir, T. L. (2019) Bacteriophage for gastrointestinal health
(PHAGE) study: evaluating the safety and tolerability of supplemental
bacteriophage consumption, J. Am. Coll. Nutr., 38, 68-75,
doi: 10.1080/07315724.2018.1483783.
106.Leitner, L., Sybesma, W., Chanishvili, N.,
Goderdzishvili, M., Chkhotua, A., et al. (2017) Bacteriophages for
treating urinary tract infections in patients undergoing transurethral
resection of the prostate: a randomized, placebo-controlled,
double-blind clinical trial, BMC Urol., 17, 90, doi:
10.1186/s12894-017-0283-6.
107.Ujmajuridze, A., Chanishvili, N.,
Goderdzishvili, M., Leitner, L., Mehnert, U., Chkhotua, A., Kessler, T.
M., and Sybesma, W. (2018) Adapted bacteriophages for treating urinary
tract infections, Front. Microbiol., 9, 1832, doi:
10.3389/fmicb.2018.01832.
108.Międzybrodzki, R., Borysowski, J.,
Weber-Dąbrowska, B., Wojciech Fortuna, W., et al. (2012) Clinical
aspects of phage therapy, Adv. Virus Res., 83, 73-121,
doi: 10.1016/B978-0-12-394438-2.00003-7.
109.Goren, M., Yosef, I., and Qimron, U. (2017)
Sensitizing pathogens to antibiotics using the CRISPR-Cas system,
Drug Resist. Updat., 30, 1-6, doi:
10.1016/j.drup.2016.11.001.
110.Park, J. Y., Moon, B. Y., Park, J. W.,
Thornton, J. A., Park, Y. H., and Seo, K. S. (2017) Genetic engineering
of a temperate phage-based delivery system for CRISPR/Cas9
antimicrobials against Staphylococcus aureus, Sci. Rep.,
7, 44929, doi: 10.1038/srep44929.
111.Pursey, E., Sünderhauf, D., Gaze, W. H.,
Westra, E. R., and Van Houte, S. (2018) CRISPR-Cas antimicrobials:
challenges and future prospects, PLoS Pathog., 14,
e1006990, doi: 10.1371/journal.ppat.1006990.
112.Pirnay, J. P., Verbeken, G., Ceyssens, P. J.,
Huys, I., De Vos, D., Ameloot, C., and Fauconnier, A. (2018) The
magistral phage, Viruses, 10, 64, doi:
10.3390/v10020064.