REVIEW: HIF-Dependent Mechanisms of Relationship between Hypoxia Tolerance and Tumor Development
Dzhuliia Sh. Dzhalilova1,a* and Olga V. Makarova1
1Federal State Budgetary Institution “Research Institute of Human Morphology”, 117418 Moscow, Russia* To whom correspondence should be addressed.
Received March 1, 2021; Revised July 2, 2021; Accepted July 14, 2021
Oxygen deficiency is one of the key pathogenetic factors determining development and severity of many diseases, including inflammatory, infectious diseases, and cancer. Lack of oxygen activates the signaling pathway of the hypoxia-inducible transcription factor HIF in cells that has three isoforms, HIF-1, HIF-2, HIF-3, regulating expression of several thousand genes. Throughout tumor progression, HIF activation stimulates angiogenesis, promotes changes in cell metabolism, adhesion, invasiveness, and ability to metastasize. HIF isoforms can play opposite roles in the development of inflammatory and neoplastic processes. Humans and laboratory animals differ both in tolerance to hypoxia and in the levels of expression of HIF and HIF-dependent genes, which may lead to predisposition to the development of certain oncological disorders. In particular, the ratio of different histogenetic types of tumors may vary among people living in the mountains and at the sea level. However, despite the key role of hypoxia at almost all stages of tumor development, basal tolerance to oxygen deficiency is not considered as a factor of predisposition to the tumor growth initiation. In literature, there are many works characterizing the level of local hypoxia in various tumors, and suggesting fundamental approaches to its mitigation by HIF inhibition. HIF inhibitors, as a rule, have a systemic effect on the organism, however, basal tolerance of an organism to hypoxia as well as the level of HIF expression are not taken into account in the process of their use. The review summarizes the literature data on different HIF isoforms and their role in tumor progression, with extrapolation to organisms with high and low tolerance to hypoxia, as well as on the prevalence of various types of tumors in the populations living at high altitudes.
KEY WORDS: hypoxia tolerance, high altitude, tumor progression, HIF, angiogenesisDOI: 10.1134/S0006297921100011
Abbreviations: EMT, epithelial–mesenchymal transition; HAP, hypoxia-activated prodrugs; HIF, hypoxia-inducible factor; HSP, heat shock protein; MCT1, monocarboxylate transporter 1; MCT4, monocarboxylate transporter 4; PHD, prolyl hydroxylase domain proteins; ROS, reactive oxygen species; UV-B, ultraviolet-B; VEGF, vascular endothelial growth factor; VHL, Von Hippel–Lindau E3-ubiquitin-ligase complex.
INTRODUCTION
According to the literature, hypoxia is one of the key factors regulating development of various tumors [1, 2]. It was demonstrated that the local oxygen deficiency in the tumor correlates with its aggressiveness, rate of metastases development, and resistance to therapy [2, 3]. Therefore, the study of the molecular and biological mechanisms of the relationship between hypoxia and tumor growth will make it possible to determine targets for the development of new effective therapies.
Cellular response to hypoxia is realized through the signaling pathway of the transcription factor HIF (hypoxia-inducible factor) [2, 4, 5]. HIF is a heterodimer consisting of one of oxygen-regulated α-subunit isoforms (HIF-1α, HIF-2α, or HIF-3α) and a constitutively expressed HIF-1β subunit (ARNT, Aryl Hydrocarbon Nuclear Receptor Translocator) [4-6]. The cytoplasmic subunit HIF-α synthesized de novo under sufficient oxygen concentration is regulated by hydroxylation of the proline residues by prolyl hydroxylases (Prolyl Hydroxylase Domain proteins – PHD1, PHD2, and PHD3), which contributes to proteasome degradation of HIF by the Von Hippel–Lindau (VHL) E3-ubiquitin-ligase complex [7, 8]. Activity of hydroxylases depends on the oxygen content in the tissues: at low concentration or in the absence of O2, it is suppressed, which leads to the decrease in HIF-α hydroxylation. As a result, the HIF-α subunit accumulates in the cytoplasm of the cells and then is translocated into the nucleus, where it dimerizes with the HIF-β-subunit. In the nucleus, the HIF-α/β dimer interacts with the hypoxia response elements (HRE), which are located in the promoters of oxygen-dependent genes encoding proteins involved in systemic and cellular adaptation to hypoxia, such as erythropoietin, vascular endothelial growth factor (VEGF) and others [9, 10].
Many mechanisms of hypoxia-independent stabilization HIF-α-subunits under normoxia were characterized. This condition is called pseudohypoxia and originates from the sensitivity of prolyl hydroxylases not only to the changes of O2 concentration, but also to the level of reactive oxygen species (ROS), iron, ascorbate, and intermediate products of the Krebs cycle [5, 11, 12]. Mutations in the genes encoding proteins involved in the Krebs cycle stabilize HIF-α subunits via inhibition of their hydroxylation [12]. For example, mutations in the isocitrate dehydrogenase 1/2 (IDH1/2) genes, which are found in more than 80% of glioblastomas, are associated with activation of HIF-1α [13, 14]. Lactate can induce HIF-1α activation by suppressing PHD-mediated proline hydroxylation [15]. Increase in the succinate and fumarate levels due to mutations in the genes encoding the corresponding enzymes leads to inhibition of prolyl hydroxylases and stabilization of HIF-1α [12]. It was demonstrated that the increase in pyruvate concentration also causes pseudohypoxic conditions [16]. Pseudohypoxia-mediated HIF-1 activation is a key mechanism for the development of a number of myelodysplastic syndromes [11].
Moreover, activation of the HIF-α subunit occurs during changes in the content of cytokines and growth factors, such as IL-1β, transforming growth factor TGF-β, heat shock protein HSP90, activation of the transcription factor NF-κB, signaling pathways PI3K/AKT/mTOR, MAPK, etc. [9, 17-19]. Thus, HIF activation during tumor progression can occur as a result of the action of many factors, that mostly determines development and outcome of the disease.
Hypoxia inducible factor (HIF) regulates many cellular and molecular biological mechanisms of tumor development, including: angiogenesis [20, 21], maintenance of the cancer stem cell population [22, 23], metabolic reprogramming [24, 25]; regulation of autocrine growth factors [26]; epithelial–mesenchymal transition (EMT) [27-30], invasion [31, 32], metastasis [33-35], and also affects resistance of tumors to radiation therapy [36] and chemotherapy [37].
ROLE OF HYPOXIA AT DIFFERENT STAGES OF TUMOR GROWTH
During the development of malignant tumors, stages of initiation, promotion, and progression are commonly identified [38-40]. Greten and Grivennikov [40] characterized two main mutually dependent events during initiation of the tumor growth. Firstly, the cell acquires a number of genetic mutations, and as their number grows, the likelihood of tumor formation increases. Cells with a limited number of mutations can stimulate the growth of benign tumors, while individual cells with a number of mutations sufficient for autonomous existence form a clone of malignant tumor cells [41, 42]. Secondly, for further development of tumors, proliferation of the transformed clones is necessary. Mutations occurring during the development of tumors followed by active cell proliferation lead to initiation of hypoxia and HIF activation [2].
In various tumors, HIF-dependent expression of many genes could be increased due to either genetic changes or intratumoral hypoxia. Increase in the HIF activity is associated with both mutations leading to dysfunction of tumor suppressors (primarily VHL) and mutations that activate protooncogenes and various signaling pathways. It was demonstrated on experimental models and clinical material that the levels of HIF-1α or HIF-2α correlate with the rate of tumor growth, vascularization, and metastasis [2]. Increase in the HIF-1 activity during the tumor development occurs either as a result of decrease in its ubiquitination or increase in the HIF-1 protein synthesis. For example, decrease of the function of suppressor genes such as p53 and VHL decreases ubiquitination of HIF-1, while activation of the PI3K/AKT/mTOR, MEK/ERK signaling pathways and dysfunction of PTEN (Phosphatase and Tensin homolog deleted on chromosome 10) promotes increase in the HIF-1 protein synthesis [2, 43, 44]. EGFR (Epidermal Growth Factor Receptor) and HER2 (Human Epidermal Growth Factor Receptor) tyrosine kinases activation in prostate and breast cancer, respectively, also increases the HIF-1α synthesis as a result of the PI3K/AKT signaling pathway activation. It stimulates the mTOR activity, which, in its turn, promotes the HIF-1α mRNA translation into the protein [45, 46]. Thus, one of the consequences of many key genetic changes in tumor cells is increase in the HIF activity.
Hypoxia, initiation of tumor development, increase of cell proliferation and survival. Hypoxia plays a key role in the initiation of tumors because many genes, including protooncogenes and tumor suppressors, are controlled by HIF [9, 10, 47]. Koshiji et al. [48] demonstrated that HIF-1 leads to genetic instability via limiting the repair of mismatched DNA nucleotides by inhibiting expression of MSH2 (MutS homolog 2) and MSH6 (MutS homolog 6) genes. It has been established that HIF also promotes activation of the protooncogene, K-Ras [49]. The so-called cancer stem cells detected in hypoxic microenvironment of various tumors such as esophagus, stomach, breast, ovaries, and kidney cancer [22, 23, 50-53] play an important role in the process of tumor development.
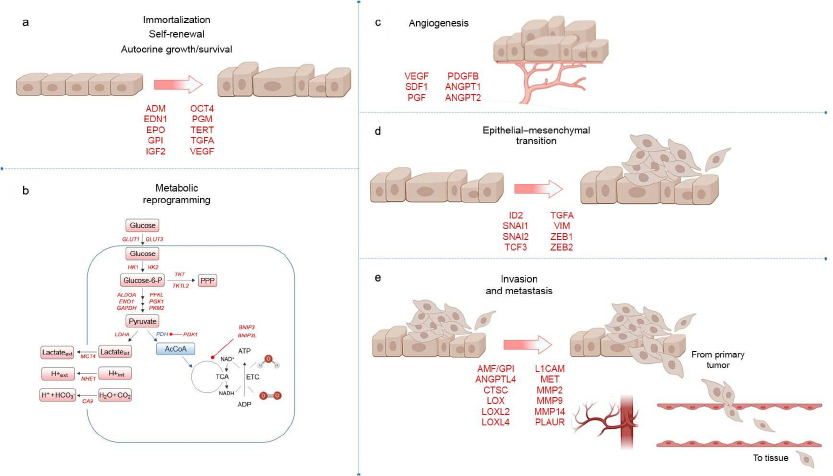
Cancer stem cells are more resistant to chemotherapy and radiotherapy than non-stem cells [51, 53]. The HIF-1 and HIF-2 expression is increased in the stem cells of many tumors, in particular, in malignant glioma, breast cancer, and in myeloblasts of acute myeloid leukemia. According to the results of in vitro and in vivo experimental studies, HIF activation maintains the cancer stem cells phenotype [23, 53, 54]. Cancer stem cells immortalization depends on the HIF-regulated increase in the TERT telomerase activity, pluripotency factors (NANOG and OCT4 – Octamer Binding Protein 4), glycolytic enzymes glucose-phosphate isomerase (GPI) and phosphoglycerate mutase (PGM), which block cell aging [55-57]. Hypoxia facilitates dedifferentiation of pancreatic cancer cells into stem cell-like phenotypes with high oncogenic potential by activating the HIF-1 and Notch signaling pathways [58].
Tumor cells, in contrast to normal cells, are characterized by increase in the proliferation rate and decrease in the death rate due to increased expression of secreted growth or survival factors. Tumor cells express membrane receptors related to the secreted growth factors, which leads to autocrine signaling. Growth or survival factors that are encoded by the HIF-regulated genes and are involved in autocrine signaling include: transforming growth factor-α (TGFA) in clear cell renal carcinoma [59]; insulin-like growth factor-2 (IGF2) and vascular endothelial growth factor (VEGF) in colorectal, gastric and pancreatic cancer [60-62]; endothelin 1 (EDN1) in breast, prostate and ovarian cancer [63]; adrenomedullin in pancreatic and prostate cancer [64] and erythropoietin (EPO) in breast and prostate cancer, kidney and melanoma [65]. These genes control key tumor processes – invasion, proliferation, angiogenesis, and metastasis.
Hypoxia and changes in the metabolism of tumor cells. Under normoxia, mitochondria maintain oxidative phosphorylation at the level required for ATP production (aerobic metabolism). However, under hypoxia, metabolism switches to anaerobic glycolysis. In tumor cells, as well as in normal cells, HIF activation promotes adaptation to hypoxia, angiogenesis, and metabolic changes. It is known that tumor cells generate ATP via glycolysis even under conditions of normal oxygen supply [66]. Otto Warburg [67] found that even with sufficient oxygen, cancer cells consume much more glucose than normal cells, which is used to form lactic acid. This discovery is called “aerobic glycolysis” or “Warburg effect”. Warburg believed that the change in respiration caused metabolic disturbances, and this contributed to the tumor progression. Later it was established that some genes that control oncogenesis, such as RAS, c-MYC, and p53, are involved in the Warburg effect regulation [68]. Many studies have demonstrated that HIF-1, as the main regulator of the response to hypoxia of both normal and tumor cells, plays an essential role in the process of aerobic glycolysis providing energy to tumor cells and preventing their damage by hypoxic stress [2, 25, 69].
HIF-1 mediates expression of the genes encoding glucose transport proteins (GLUT1, GLUT3) and glycolytic enzymes (ALDOA – aldolase A, ENO1 – enolase 1, GAPDH – glyceraldehyde-3-phosphate dehydrogenase, HK1, HK2 – hexokinase 1 and 2, PFKL – phosphofructokinase L, PGK1 – phosphoglycerate kinase 1, PKM2 – pyruvate kinase M2, LDHA – lactate dehydrogenase A), which convert glucose into lactate [2, 47]. Aerobic glycolysis is faster than mitochondrial oxidative phosphorylation. Glucose uptake by the metastatic tumor cells is so significant in comparison with the normal cells, that it serves as a basis for clinical screening of malignant tumors metastasis in cancer patients. For this purpose, patients are injected with 18F-fluorodeoxyglucose, which is visualized using positron emission tomography [70]. Thus, HIF-1 stimulates glycolysis in tumor cells by increasing glucose uptake and activity of glycolytic enzymes, therefore creating an ineffective but rapid way of generating energy.
Activation of HIF and HIF-dependent genes in proliferating tumor cells contributes not only to metabolic changes, but also to formation of the special microenvironment. A critical effect of enhanced glucose uptake and glycolysis activation by tumor cells is intercellular acidosis in tumor tissues. Tumor cells contribute to reduction of extracellular pH by activating the HIF-1 target genes complex encoding the synthesis of plasma membrane proteins, such as monocarboxylate transporter 4 (MCT4), sodium hydrogen antiporter (NHE-1), and carbonic anhydrase 9 (CA9) [71]. Decrease of pH in the intercellular space stimulates activity of proteinases, which destroy the extracellular matrix and disrupt ability of the cells to interact. Metabolic changes, glycolysis activation, and formation of the acidic environment facilitate cell proliferation and tumor progression [71]. It has been demonstrated that increase in the lactate concentration in many carcinomas is associated with an increased metastasis risk [72].
The main components of the tumor cell microenvironment are blood and lymphatic vessels, fibroblasts, immune cells, and extracellular matrix [73]. Cancer-Associated Fibroblasts (CAF) are one of the main stroma components, and they can make up to 80% of all tumor cells. Paracrine signaling between the tumor cells and fibroblasts is affected by oxygen deficiency. Hypoxia was demonstrated to stimulate secretion of the chemokine CXCL13 by tumor-associated myofibroblasts in the prostate cancer progression [74]. Tumor cells under hypoxic conditions can secrete paracrine signaling molecules that promote progenitor cells reprogramming in CAF [75], and HIF-1 regulates some of these signaling molecules, such as TGF-β, bFGF (basic fibroblast growth factor), and PDGFB (platelet growth factor B) [2, 26, 59].
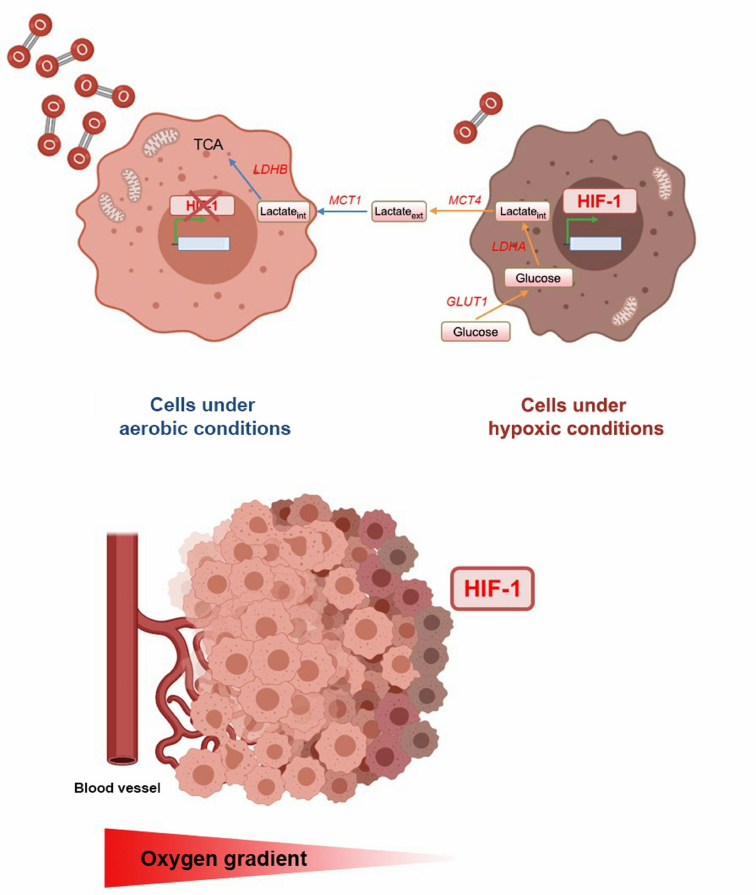
In terms of oxygen level in different areas, tumors are heterogeneous and have both aerobic and hypoxic regions. Tumor cells adjacent to blood vessels are oxygenated to a higher degree and exhibit oxidative metabolism, while cells located at a distance from blood vessels are insufficiently oxygenated and glycolysis predominates in them. The existence of “metabolic symbiosis” between the tumor cells from hypoxic and aerobic regions was demonstrated (Fig. 1) [76]. In the not adequately oxygenated cells, HIF-1 mediates high expression of glucose transporters, glycolytic enzymes, including LDHA and MCT4. These changes in the expression of these genes lead to increase in the glucose uptake, its conversion into lactate, and removal of the latter from the cell. In contrast, the well-oxygenated cells express monocarboxylate 1 (MCT1) transporter and lactate dehydrogenase B (LDHB), which mediate the lactate uptake and its conversion to pyruvate used for oxidative phosphorylation. Lactate produced by glycolysis in hypoxic cells is secreted by MCT4, taken up by aerobic tumor cells via MCT1, and converted to pyruvate by LDHB. It is next used for oxidative phosphorylation by aerobic tumor cells to produce ATP. Due to this mechanism, more glucose remains for the metabolism and survival of hypoxic tumor cells [76]. Intratumoral hypoxic foci surrounded by normoxic cells serve as a kind of platform for progression of the infiltrative tumor growth. It has been demonstrated that invasion occurs precisely from the hypoxic areas of the tumor with low pH [77].
Fig. 1. Effect of oxygen concentration gradient on the development of intratumoral heterogeneity. Cells are well oxygenated around the vessels, i.e., are under aerobic conditions, and cells located at a distance from the vessels are under hypoxic conditions, and the HIF-1 expression is activated in them. Under hypoxia, cell metabolism changes towards glycolysis, which leads to excessive lactate formation. Lactate is removed from hypoxic cells by MCT4 and absorbed by well-oxygenated cells via MCT1. This interaction between the cells enables efficient use of glucose, which promotes tumor progression.
Partial pressure of oxygen in the tumor is significantly lower than in the normal tissues surrounding the tumor [78]. Since different organs and tissues of organism are supplied with oxygen to different extent, the hypoxia level in tumors may vary depending on their histogenesis (Table 1). For example, oxygen partial pressure in the soft tissue sarcomas is less than 22 mmHg [79], while in the progressive head and neck squamous cell carcinoma pO2 is less than 5 mmHg [80]. Tumors with the lowest oxygen partial pressure are characterized by high rate of invasion and metastasis [1, 81]. It was demonstrated that in patients suffering from cervical cancer with partial oxygen pressure in the tumor less than 10 mmHg (1% oxygen) life expectancy with surgery or radiotherapy is significantly lower than in patients with higher degree of tumor oxygenation [82].
Table 1. Parameters of partial pressure and
oxygen content in human tumors of various localizations and in normal
tissues
Hypoxia and angiogenesis. Tumor cells undergo adaptive and genetic changes that allow them to survive and proliferate in a hypoxic environment. During the tumor development and progression, hypoxia occurs due to excessive cell proliferation and insufficient blood supply, which activates angiogenesis, regulated by the HIF-dependent factor VEGF [2, 20, 88]. Oxygen deficiency induces imbalance between the production of pro-angiogenic and anti-angiogenic factors, which leads to excessive, rapid and chaotic formation of blood vessels with significant increase in their number. Such vessels are characterized by thin walls and variety of shapes and sizes [2, 89]. HIF-1 controls expression of several genes encoding angiogenic growth factors, including VEGF, SDF1 (Stromal-Derived Factor 1), PGF (Placental Growth Factor), PDGFB, and angiopoietin (ANGPT 1 and 2) [2, 20, 88].
HIF-1 and HIF-2 expression in endothelial cells has different effects on vascularization. In experimental models, inhibition of HIF-1 activity significantly suppresses tumor vascularization, in particular, in the case of prostate cancer [21, 90]. While HIF1A gene deletion reduces tumor vascularization and tumor growth, HIF2A deletion, on the contrary, enhances angiogenesis with formation of immature vessels and increase in the degree of tumor hypoxia [91, 92].
Hypoxia and metastasis of tumors. Tumor progression is associated with metastasis, which is largely determined by interaction of the tumor cells with microenvironment and involves the process of epithelial–mesenchymal transition (EMT) [30]. HIF-1 regulates the process of tumor metastasis, changing adhesion and mobility of the tumor cells and activating EMT. The hypoxia-induced EMT is characterized by the decrease in expression of the genes encoding epithelium-associated proteins – E-cadherin and β-catenin, and by the increase in expression of the genes encoding proteins of mesenchymal phenotype – N-cadherin, vimentin, α-SMA (α-Smooth Muscle Actin) and chemokine receptor CXCR4 [27, 28, 93]. HIF-1 promotes transcription activation of the genes encoding repressors (ID2 – Inhibitor of Differentiation 2, SNAI1, SNAI2 – Snail Family Transcriptional Repressor 1 and 2, TCF3 – Transcription Factor 3, ZEB1 and ZEB2 – Zinc Finger E-boxbinding Homeobox 1 and 2) that block E-cadherin and other proteins expression, thus providing cell adhesion and normal epithelial tissues architectonics [27-29]. In addition, HIF-1 activates the TGFA and vimentin (VIM) expression. These proteins contribute to the changes in the cytoskeleton structure and acquisition of the mesenchymal phenotype by the tumor cells [59]. Furthermore, HIF-1 activates matrix metalloproteinases MMP1, 2 and MMP9, LOX (Lysyl Oxidase), TWIST, and the MET protooncogene, which are responsible for migration and invasiveness [2, 32, 33]. LOX expression by metastatic cells increases their ability to form colonies in tissues of various organs. HIF-1 activates transcription of the genes encoding proteases that destroy (CTSC – Cathepsin C, MMP2, MMP9, PLAUR – Urokinase Plasminogen Activator Receptor) or remodel (LOX, LOXL2, LOXL4) the extracellular matrix [2, 32-34].
An important link in the process of metastasis is the HIF-1-regulated intravasation – penetration of tumor cells into the blood vessels [94]. It was shown that the HIF-1-induced ANGPTL4 (Angiopoietin-Related Protein 4) decreases adhesion of the vascular endothelial cells in breast cancer, which promotes entry of the tumor cells into the vascular lumen [34]. According to Zhang et al. [35], L1CAM (L1 Cell Adhesion Molecule) and ANGPTL4 also decrease adhesion and initiate extravasation of the tumor cells into the organ parenchyma with formation of metastases. The role of HIF at different stages of tumor development is presented in Fig. 2.
Fig. 2. Role of HIF at different stages of tumor development. HIF-dependent genes encoding proteins that are involved in key stages of tumor development are highlighted with red color. a) Genes that control immortalization, stem cell self-renewal, autocrine growth and survival; b) genes involved in metabolic reprogramming. Enzymatic activity of pyruvate dehydrogenase (PDH) is inhibited by HIF-dependent PDH-kinase 1 (PDK1), which blocks conversion of pyruvate to acetyl-CoA for participation in the tricarboxylic acid (TCA) cycle; c) HIF activates genes that promote angiogenesis in the tumor; d) genes contributing to EMT; e) genes that control invasion and metastasis.
ROLE OF HYPOXIA AND HIF IN TUMORS OF DIFFERENT LOCALIZATION AND
DIFFERENT EXTENT OF DIFFERENTIATION
According to the literature, organs differ in their sensitivity to hypoxia [95]. The most sensitive to hypoxia are the phylogenetically younger parts of the brain – the cerebral and cerebellum cortex [96]. In addition to the brain, highly sensitive to hypoxia organs are heart, lungs, kidneys and liver. In comparison with other organs, heart and liver are normally characterized by the high consumption of O2 and increased HIF-1 expression level [97]. The organs with medium sensitivity to hypoxia include pancreas and adrenal glands, and the low-sensitivity organs include bones, cartilages, tendons, and ligaments [96]. It is known that even under physiological conditions, an oxygen gradient is observed inside a tissue and there are hypoxic areas, which are necessary for normal cells functioning [98]. Therefore, the role of HIF in tumor progression mostly depends on its location. However, there are no data in the literature on the relationship between the tumor development rate and initial oxygen status of the organ in which it is formed.
According to the immunohistochemical examination of biopsy materials, increased HIF-1α expression was detected in the cells of almost all human tumors [99, 100]. HIF-2α expression increased only in some tumor types, such as renal carcinoma, non-small cell lung cancer, and cerebellar hemangioblastoma [101-103]. HIF-1 promotes breast cancer progression, and its inhibition reduces metastasis [104]. In addition, increased HIF-1α expression in the colon, stomach, lungs, skin, ovary, pancreas, and prostate carcinomas correlates with high proliferative activity of tumor cells [99]. According to the immunohistochemical studies, increased HIF-1α expression in the tumor cells is associated with high degree of malignancy, poor prognosis, and resistance to various types of antitumor therapy for tumors of different localizations – head and neck, ovary, and esophagus [99, 100].
Increase in the expression of some HIF-dependent genes, in particular, VEGF, in glioma tissues is also a factor of poor prognosis [105]. It was demonstrated that C6 glioma cells respond to hypoxia by activating HIF-1 [106]. During the glioma progression, hypoxia promotes high VEGF production, which is a key regulator of angiogenesis, and, therefore, it plays an important role in the tumor growth progression [105].
In some tissues, HIF-1 stabilization has a suppressive effect on tumor growth. In particular, it was demonstrated that the loss of HIF-1α expression promotes the renal carcinoma cells proliferation [107]. In addition, in ovarian cancer, increased HIF-1α expression correlates with the intensity of apoptosis, and combination of the increased tumor cells death and high HIF-1α expression promotes patient survival. However, in ovarian cancer, in which the expression of both HIF-1α and mutant p53 was increased, the apoptosis level was low, and this was associated with the decrease in the patient’s life expectancy [108]. In the early stages of esophageal cancer in patients, combination of increased HIF-1α and BCL2 expression was associated with the resistance to photodynamic therapy [109]. Thus, the effect of increased HIF-1α expression depends on the tumor localization and presence of mutations in the genes that affect the balance between proapoptotic and antiapoptotic factors.
It was established that different HIF isoforms, HIF-1 and HIF-2, could play different roles in tumor progression. It was discovered in the mouse model of non-small cell lung cancer (NSCLC) that HIF-2α inhibition promoted the tumor cells proliferation, while decrease in HIF-1α activity did not affect oncogenesis [110]. Analysis of survival of the patients with NSCLC revealed that the increased HIF-2α expression was statistically significantly associated with poor prognosis, while the HIF-1α expression did not have such effect [103]. It was observed in the model of colon cancer associated with chronic colitis that increase in the HIF-1 expression in intestinal epithelial cells did not lead to tumor progression. The HIF-2α-mediated inflammation promotes the colon tumors development, and HIF-2α activation in acute colitis causes severe disease course [111].
Controversial data on the role of HIF and its subunits in the progression of tumors of different localizations are probably due to the complex interactions of many factors, activation and suppression of certain genes during the development of each specific tumor. Therefore, further studies of the role of HIF in the progression of tumors of different localization, degree of differentiation, and histogenesis taking into account the initial oxygen status of the tissue are required.
POTENTIAL OF TUMOR THERAPIES BASED ON HYPOXIA CORRECTION
Therapeutic approaches to the hypoxia level correction in tumors are based on increase in their oxygenation or decrease in oxygen consumption by cells. To reduce the level of hypoxia in tumors, methods such as hyperbaric oxygenation, hyperthermia, and carbogen inhalation in combination with administration of nicotinamide were suggested. These methods, despite their effectiveness, have not found wide application in oncology due to high cost, technical difficulties of their use, and side effects [112, 113].
In recent decades, the areas of pharmacological correction of hypoxia in tumors has been developing intensively. For this purpose, drugs are used to inhibit HIF, which is the main gene expression regulator in hypoxic cells.
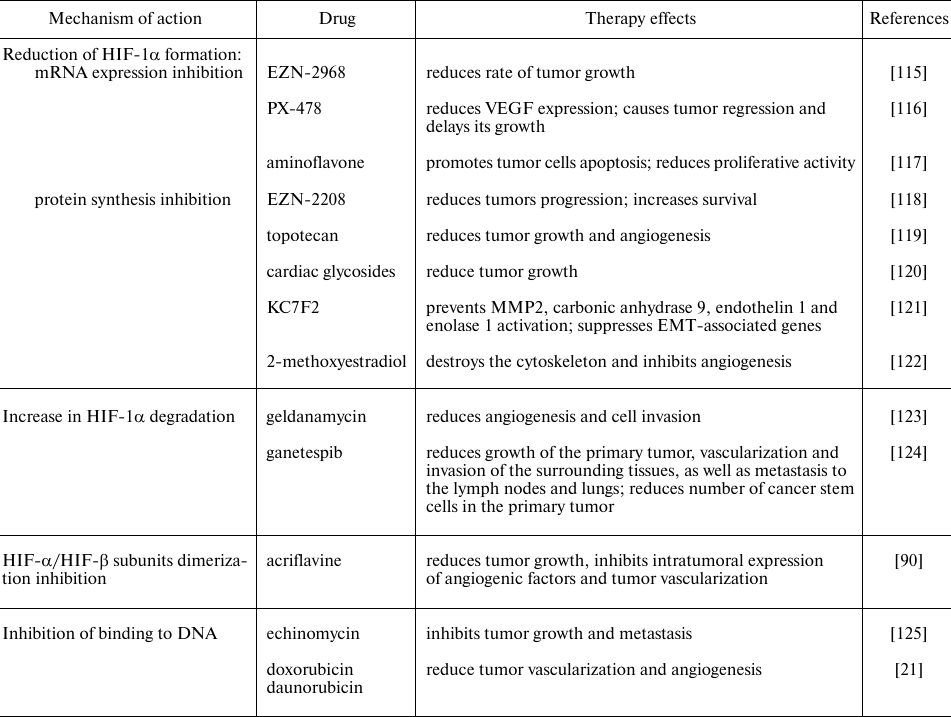
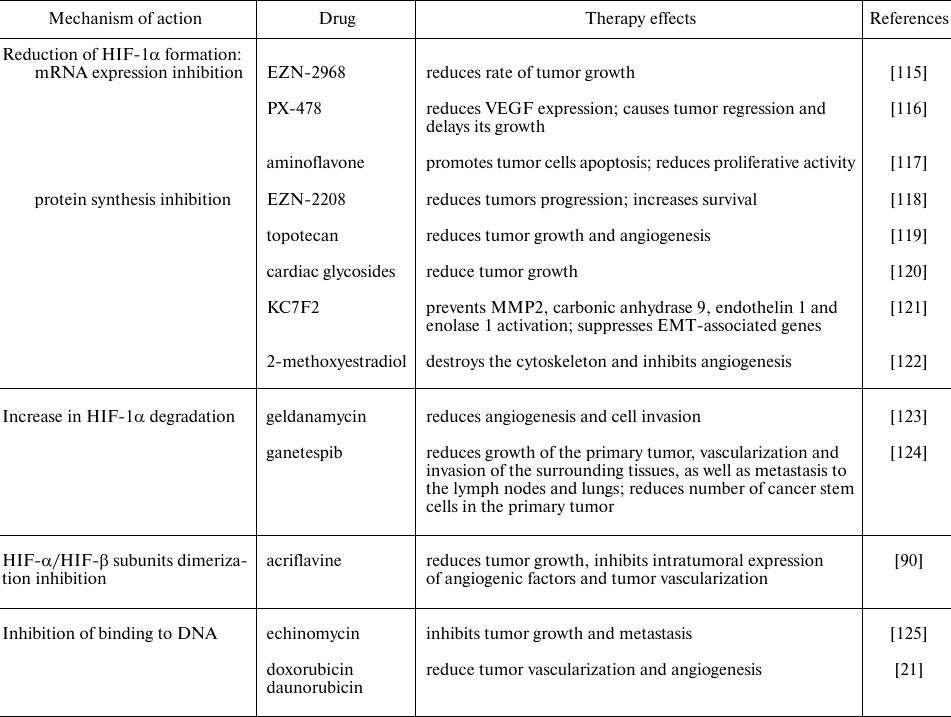
Many chemicals and drugs are capable of inhibiting HIF through various molecular mechanisms, in particular, through decrease in the mRNA expression, protein synthesis, HIF-1α- and HIF-1β-subunits dimerization, increase in HIF-1α degradation and prolyl hydroxylase activity, decrease in binding to DNA, etc. (Table 2). There are two main categories of HIF inhibitors: direct inhibitors that affect expression or function of HIF molecules, and indirect ones that regulate other molecules that indirectly affect HIF. Many of the potential HIF inhibitors are currently undergoing clinical trials [114].
Table 2. HIF inhibitors used in tumor
therapy
It should be taken into consideration that the strategy of the use of HIF-1 inhibitors in oncology could have an adverse effect on the course of concomitant diseases, which, on the contrary, require high HIF-1 expression level – ischemia, anemia, etc. [126]. In particular, in ischemic heart disease and ischemic stroke, the HIF-1-dependent increase in VEGF expression induces new blood vessels formation in the ischemic region, increases blood flow and oxygen supply, thereby reducing ischemia [127]. For example, increase of HIF-1 and HIF-2 activity for stimulation of erythropoietin synthesis is required for treatment of anemia associated with chronic kidney disease [128]. For this purpose, numerous prolyl hydroxylases inhibitors are used, which contribute to HIF-α subunit degradation in the proteasome. In addition, experimental models demonstrated that the increase in HIF-1 expression contributes to the decrease in the inflammatory bowel diseases severity [129].
Intravenous administration of anticancer chemotherapeutic drugs provides not only local effect (on tumor cells), but also a systemic effect, which can reduce effectiveness of the treatment and cause side effects. However, dependence of the therapeutic effect of antitumor drugs on the basal resistance of an organism to hypoxia and HIF expression level has not been studied. Effectiveness of the drugs may not be the same in organisms with different resistance to hypoxia. In particular, the effect of antihypoxants is more pronounced in animals susceptible to hypoxia [130].
One of the promising approaches to the treatment of tumors with severe hypoxia is the use of molecules with increased toxicity specifically for hypoxic cells. The Hypoxia-Activated Prodrugs (HAPs) are agents selectively activated under conditions of oxygen deficiency targeted to the destroying hypoxic tumor cells that are resistant to conventional treatments. HAPs are introduced in an inactive form, and they are biologically inert under normoxia, but they can undergo enzymatic reduction by oxidoreductases under hypoxic conditions to form biologically active compounds that are easily reoxidized in the presence of oxygen [131].
The first HAP studies were carried out by the group A. C. Sartorelli from Yale University; they demonstrated that mitomycin C was activated predominantly under hypoxic conditions and thus acquired the ability to selectively destroy hypoxic cells [132].
At present, efficacy of several HAPs has been evaluated, including porphyromycin, tirapazamine (SR4233), banoxantrone, evophosphamide, PR-104, and tarloxotinib [133]. Evophosphamide and tarloxotinib are currently undergo clinical trials.
Evophosphamide (TH-302) demonstrated high efficacy at the stage of preclinical studies, but it was not confirmed when evaluated in the phase III clinical trials. It may be due to the fact that intratumoral hypoxia level was not taken into account [134, 135]. In addition, the effectiveness of prodrugs acting on tumor cells in the hypoxic area may depend not only on the degree of local hypoxia in the tumor, but also on the individual resistance to oxygen deficiency of the whole organism.
Currently, the assessment of oxygen content in tumors is carried out mainly for research purposes, and there are several methods for this. Direct measurement of pO2 using an oxygen electrode inserted into a tumor has been widely used to determine oxygenation degree of the solid tumors. This procedure is invasive, making it difficult to perform repeated measurements. In addition, tumors are heterogeneous in terms of the hypoxia degree, and skilled staff is required to work with such electrodes [136]. A promising technique, oxygen-enhanced MRI (OE-MRI) suggested by O’Connor et al. is less invasive [137].
Hypoxia and HIF activation play a key role in the development of tumor cell resistance to various treatments. The main mechanism of HIF chemoresistance is an increase in the expression of transporter proteins (such as Multidrug Resistance 1 Protein (MDR1), Multidrug Resistance-associated Protein 1 (MRP1), and Breast Cancer Resistance Protein (BCRP)), which decrease concentration of the chemotherapeutic agents within tumor cells located under hypoxic conditions. Drugs used in the treatment of breast cancer, such as doxorubicin, paclitaxel, and vincristine, are substrates for MDR1-mediated concentration decrease, and activation of HIF-1 increases resistance of tumor cells to them [138, 139].
Sensitivity to chemotherapy mostly depends on the intensity of blood flow inside tumors; therefore, irregularity of vasculature in the tumors correlates with impaired delivery of chemotherapeutic agents [140]. In addition, changes in the pH gradient due to hypoxia can disturb activity of the pH-dependent chemotherapeutic agents (doxorubicin and docetaxel) and DNA alkylating agents such as temozolomide, chlorambucil, and ifosfamide. Other HIF-related mechanisms of resistance to chemotherapy are: increased autophagy, altered cell metabolism, and inhibition of apoptosis.
Hypoxia in tumors also contributes to radiation therapy resistance through a number of factors controlled by HIF. In particular, the HIF-1-mediated radioresistance of HeLa cells under hypoxic conditions was associated with the decrease in Bax and p53 expression and decrease in the radiation-induced apoptosis [141]. It was demonstrated that the HIF-dependent increase in expression of Human Epididymis Protein 4 (HE4) resulted in high resistance of gastric cancer cells to radiation therapy [142], and erythropoietin increased glioma cells survival after exposure [143].
ROLE OF HIF EXPRESSION AND BASAL RESISTANCE OF AN ORGANISM TO
HYPOXIA IN THE PROGRESSION OF TUMORS
Emergence of most tumors is associated with the influence of environmental factors and depends on the organism genetic characteristics [144]. It was discovered that the development of 95% of tumors is associated with interaction of environmental factors and genes, and 5% – only with genes [145]. Genetic factors affecting predisposition to cancer development include rare dominant mutations, as well as more common genetic polymorphisms that determine response of the individual to environmental factors [145]. In particular, variability in expression levels or isoforms of metabolic genes such as P450, glutathione S-transferase (GST), and N-acetyl transferase (NAT), and of HIF-1α significantly affects the individual response to carcinogens. It was shown that people differ significantly in the individual levels of expression of HIF-1α and HIF-dependent genes in leukocytes [146, 147]. Presence of HIF1A gene polymorphisms has been revealed in humans, which ensures high level of its expression. One of the most studied HIF-1α polymorphisms is 1772 C>T (rs11549465 C>T, Pro582Ser). This polymorphic variant determines increase in HIF-1α transcriptional activity both under normoxic and hypoxic conditions in comparison with the wild type [148]. Recently, it has been demonstrated that the HIF-1α rs11549465 C>T polymorphism is associated with increased risk of tumor development [149].
It is known that human and laboratory animals differ in resistance to hypoxia, i.e., hypoxic exposure of the same degree causes different reactions in individuals of the same population, age, and gender [97, 150-158]. However, despite the important role of hypoxia in the processes of oncogenesis, practically at all stages of tumor formation, the individual basal resistance of an organism to hypoxia is not considered as a factor of predisposition to the tumor growth initiation. Gorr et al. [3] note that the degree of basal resistance of an organism to oxygen deficiency can also determine the level of intratumoral hypoxia.
Experimental studies revealed tolerant and susceptible to hypoxia laboratory animals; sensitivity to acute mountain sickness and high-altitude pulmonary edema has been studied in humans [97, 150-158]. The resistance to hypoxia of laboratory animals is determined in a decompression chamber based on the “gasping time” at a critical height – the time before the animal assumes lateral position. The “gasping time” of the tolerant to hypoxia animals usually differs from the susceptible animals by at least 3-fold. Results of determination of the resistance to hypoxia in a decompression chamber revealed that a significant part of animals exhibit normal tolerance (40-58%), the high tolerant animals include from 20 to 42%, and susceptible animals – from 18 to 40% [97, 150, 151, 158].
It is known from the literature that resistance to hypoxia is associated with the activity of antioxidant defense enzymes [157]. Enzymatic system of protection against oxidative stress is more active in people living at high altitude, which is due to the constant exposure to hypoxia and ROS formation [157]. It has been demonstrated that the level of enzymes such as superoxide dismutase and catalase, which protect cells from oxidative stress, in the heart of tolerant to hypoxia Sprague-Dawley rats after hypoxic exposure is higher than in the susceptible animals [150]. Expression of the most studied heat shock proteins (HSP60, HSP70, and HSP90) stabilizing membranes in the myocardium of tolerant to hypoxia rats was significantly increased, which indicated their ability to overcome prolonged exposure to O2 deficiency and more effective adaptation [150, 153]. Our earlier data [158] also indicate that the animals with different resistance to hypoxia have different adaptive capabilities and predisposition to the development of inflammatory diseases: the content of the oxidative stress marker 8-isoprostane increased in the susceptible animals after hypoxic exposure, which is associated with the damage of cellular membranes and increase in the TGF-β level. In the myocardium of susceptible to hypoxia rats, the content of malondialdehyde, which is formed upon exposure to ROS and is one of the oxidative stress markers, is almost 8-fold higher [150]. Oxidative stress plays a key role in the tumor development. It was demonstrated that interval exposure to hypoxia leads to increase in ROS production, which promotes survival and proliferation of the tumor cells [159]. Apparently, organisms with different resistance to hypoxia can be predisposed to the development of tumor diseases to varying degrees, which is probably mostly due to the different levels of oxidative stress and antioxidant protection degree.
Nevertheless, the mechanisms of tumor development in organisms with different resistance to hypoxia have not been sufficiently studied. Expression of HIF-1, NF-κB, and VEGF probably plays an important role. Under normoxia, in people susceptible to high-altitude pulmonary edema, the serum HIF-1α level is higher than in tolerant people [156]. It has been demonstrated that the animals tolerant and susceptible to hypoxia differ in many parameters, including the content of HIF-1α and VEGF: in susceptible to hypoxia rats HIF-1α level in the neocortex under normoxic conditions is 1.7 times higher than in the tolerant ones [97, 150, 151]. We have demonstrated that in the case of a systemic inflammatory response, the higher HIF-1 expression in the susceptible to hypoxia animals is accompanied by increase in the NF-κB expression and a more severe course of the disease [151]. It was also demonstrated with acute and chronic colitis models that the course of these diseases is more severe in the mice susceptible to hypoxia [160, 161]. Taking into account the role of HIF in the regulation of many genes responsible for various processes in the body, it is obvious that the higher basal expression of HIF-1α in the susceptible to hypoxia organisms can affect both oncogenesis and resistance to therapy of various types of cancer. However, the HIF-1α expression levels in tumors in organisms with different resistance to hypoxia have not been studied yet.
We have previously demonstrated [162] that the B16 melanoma progression was more pronounced in the susceptible to hypoxia mice, which was manifested by the large volume of the primary node and relative area of necrosis, high proliferation rates assessed by the mitotic index and the number of Ki-67+ cells, as well as increased VEGF expression in the liver. The number of caspase 3-positive cells dying via apoptosis mechanism was higher in the tolerant to hypoxia mice. HIF-1α expression is increased in many types of human tumors, including melanoma [163, 164]. Constitutive HIF-1α expression was demonstrated in melanoma cells using immunohistochemical analysis, quantitative PCR, Western blotting, and immunofluorescence staining of cultured cells [163, 164]. HIF-1α expression in the patients with melanoma was detected in the majority of tumor samples (87.6%), but its level did not correlate with the prognosis of the disease course and life expectancy [163]. It has been demonstrated that increase of expression of both HIF-1α and HIF-2α correlates with the VEGF expression in melanoma; however, a stronger association was found for HIF-2α with VEGF and poor prognosis [163]. It is possible that there are also differences between the tolerant and susceptible to hypoxia organisms in the expression of HIF-2α, which is a more significant factor in the melanoma progression, however, to the best of our knowledge no such data have been published.
The data on hypoxia-resistant naked mole rats that live in extreme conditions of oxygen deficiency and are able to withstand an average 250 s of complete anoxia are of particular interest [165]. For a long time, it was believed that tumors do not develop in them, although cases of neuroendocrine carcinoma of the stomach and subcutaneous adenocarcinoma in these animals have recently been reported [166]. Possibly, the high degree of protection against tumor development in naked mole rats depends on the characteristics of metabolism, HIF-1 expression, and pronounced antioxidant protection [165, 167].
Thus, hypoxia plays a key role at different stages of tumor development. Different predisposition of people to the development different types of tumors may be due not only to external and genetic factors, age, gender, but also could be determined by individual resistance to hypoxia and expression levels of various isoforms of HIF factor. In this regard, further research is needed in this area.
TOLERANCE OF THE ORGANISM TO THE DEVELOPMENT OF TUMORS IN THE
POPULATION LIVING AT HIGH ALTITUDES
Systemic hypoxia affects the incidence of some types of cancer (Table 3). Epidemiological studies indicate that highlanders demonstrate low mortality rate from malignant tumors. Adaptation to chronic hypoxic effects in the mountains leads to hypoxia-dependent metabolic changes, including changes in carbohydrate and lipid metabolism. People living at high altitudes have low blood glucose levels [168], which may be a protective factor against the increased risk of tumor development associated with hyperglycemia [169]. Lipids are required to regulate many tumor-associated signaling pathways, and high plasma lipid concentrations are associated with increased risk of tumor development [170]. Highlanders were found to have lower levels of fatty acids in blood plasma, as well as cholesterol [171], which also determines low cancer incidence and mortality.
Table 3. Epidemiological data on morbidity
and mortality of various tumors in the population living at the high
altitude
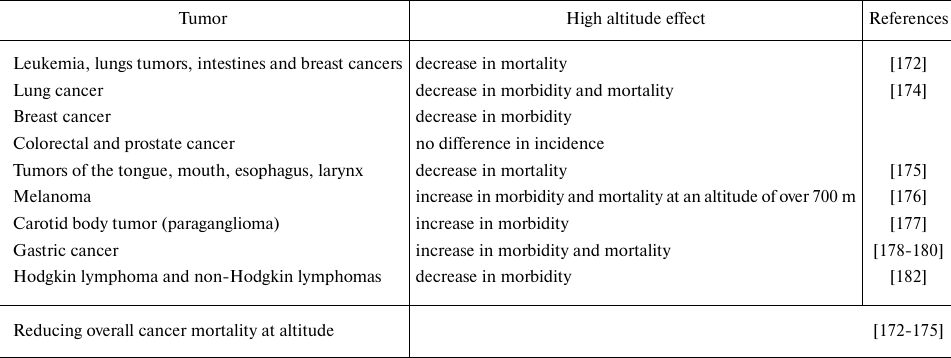
Although the overall malignant tumors mortality is negatively correlated with the increase in altitude [172-175], a number of features have been identified depending on the tumor histogenesis. While lymphoma, breast, esophagus, tongue, mouth, or larynx cancer demonstrated reduced mortality at high altitudes, mortality of liver and cervical cancer did not differ between the people living at sea level and in mountains [172, 175]. An inverse relationship was observed between the incidence and mortality rate of lung cancer and altitude [174]. Naturally, melanoma morbidity and mortality increase at altitude due to higher exposure to ultraviolet-B (UV-B) radiation [176]. Moreover, the incidence of carotid tumor, paraganglioma, increases in the highlands [177]. Altitude is considered as a very important environmental factor in the development of gastric cancer and affects its development both directly and indirectly. The direct effect of altitude on the gastric cancer development has been demonstrated in a number of studies: in Ecuador, in the high mountain provinces, mortality from stomach cancer is the highest [178, 179]. High incidence of stomach cancer is observed in the highlands of Spain, Iran, China, and Latin America [178, 179]. It should be noted that, in addition to altitude, a number of other factors have significant effect on the stomach cancer development, such as dietary habits and low selenium content in the soil in high mountain regions [180].
Decrease or increase in the incidence of malignant tumors of particular localization in the population living in high mountain areas is determined, in addition to hypoxia, by other environmental factors. Among these environmental factors affecting development of some types of cancer at high altitude the role of air pollution, UV-B and the associated vitamin D synthesis have been discussed. It has been suggested that the decrease in mortality of a number of tumors (lymphoma, breast, lung, and others) in the mountains may be due to the increased UV-B-dependent vitamin D synthesis, however, for prostate cancer there has been no association between high altitude and vitamin D level [174, 181]. Thus, protective role of the elevated vitamin D levels in cancer development and mortality is possible, but probably depends mostly on the tumor location, therefore, further research is needed in this area.
In general, the strongest environmental and adaptation factor at altitude is the low availability of oxygen, and this may make the largest contribution to the malignant tumor mortality. The data indicating that cancer mortality rate is reduced under high altitude conditions with low oxygen levels, at first glance, contradicts common sense, since intratumoral hypoxia itself is a factor for tumor growth and formation of more aggressive tumor phenotype [1, 81]. It should be noted that at altitude, due to the organism adaptation, the mechanisms for correcting hypoxia are activated, HIF is stimulated and hemoglobin content in the blood increases.
Thus, oxygen deficiency and HIF expression activation are pathogenetic factors in the development of tumor diseases. During the development and progression of tumors, HIF activation stimulates angiogenesis, promotes changes in cell metabolism, adhesion, ability to invade and metastasize. Various HIF isoforms – HIF-1, HIF-2 and HIF-3 – can play opposites role in the tumor development. Organisms of humans and laboratory animals differ both in resistance to hypoxia and in the expression levels of HIF isoforms and HIF-dependent genes, which may lead to predisposition to the development of certain cancers. In particular, incidence of various types of tumors varies in people living in highlands and at sea level. Further research is needed on the role of hypoxia in tumor formation that takes into account individual resistance to oxygen deficiency.
Funding. This work was financially supported by the State Budget project no. AAAA-A19-119021490067-4.
Ethics declarations. The authors declare no conflict of interest in financial or any other sphere. This article does not contain any studies with human participants or animals performed by any of the authors.
REFERENCES
1.Harris, A. L. (2002) Hypoxia – a key
regulatory factor in tumour growth, Nat. Rev. Cancer, 2,
38-47, doi: 10.1038/nrc704.
2.Semenza, G. L. (2010) Defining the role of
hypoxia-inducible factor 1 in cancer biology and therapeutics,
Oncogene, 29, 625-634, doi: 10.1038/onc.2009.441.
3.Gorr, T. A., Wichmann, D., Hu, J.,
Hermes‐Lima, M., Welker, A. F., et al. (2010) Hypoxia tolerance
in animals: biology and application, Physiol. Biochem. Zool.,
83, 733-752, doi: 10.1086/648581.
4.Semenza, G. L., and Wang, G. L. (1992) A nuclear
factor induced by hypoxia via de novo protein synthesis binds to
the human erythropoietin gene enhancer at a site required for
transcriptional activation, Mol. Cell Biol., 12,
5447-5454, doi: 10.1128/MCB.12.12.5447.
5.Ratcliffe, P., Koivunen, P., Myllyharju, J.,
Ragoussis, J., Bovee, J., et al. (2017) Update on hypoxia-inducible
factors and hydroxylases in oxygen regulatory pathways: from physiology
to therapeutics, Hypoxia, 5, 11-20, doi:
10.2147/HP.S127042.
6.Wang, G. L., Jiang, B. H., Rue, E. A., and Semenza,
G. L. (1995) Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS
heterodimer regulated by cellular O2 tension, Proc. Natl.
Acad. Sci. USA, 92, 5510-5514, doi:
10.1073/pnas.92.12.5510.
7.Ivan, M., Kondo, K., Yang, H., Kim, W., Valiando,
J., and Ohh, M. (2001) HIFalpha targeted for VHL mediated destruction
by proline hydroxylation: implications for O2 sensing,
Science, 292, 464-468, doi: 10.1126/science.1059817.
8.Jaakkola, P., Mole, D. R., Tian, Y. M., Wilson, M.
I., Gielbert, J., and Gaskell, S. J. (2001) Targeting of HIF-alpha to
the von Hippel–Lindau ubiquitylation complex by
O2-regulated prolyl hydroxylation, Science,
292, 468-472, doi: 10.1126/science.1059796.
9.Semenza, G. L. (2010) Oxygen homeostasis, Wiley
Interdiscip. Rev. Systems Biol. Med., 2, 336-361, doi:
10.1002/wsbm.69.
10.Schodel, J., Oikonomopoulos, S., Ragoussis, J.,
Pugh, C. W., Ratcliffe, P. J., and Mole, D. R. (2011) High-resolution
genome-wide mapping of HIF-binding sites by ChIP-seq, Blood,
117, e207-e217, doi: 10.1182/blood-2010-10-314427.
11.Hayashi, Y., Zhang, Y., and Yokota, A. (2018)
Pathobiological pseudohypoxia as a putative mechanism underlying
myelodysplastic syndromes, Cancer Discov., 8, 1438-1457,
doi: 10.1158/2159-8290.CD-17-1203.
12.Schönenberger, M. J., and Kovacs, W. J.
(2015) Hypoxia Signaling pathways: modulators of oxygen-related
organelles, Front. Cell Dev. Biol., 3, 42, doi:
10.3389/fcell.2015.00042.
13.Zhao, S., Lin, Y., Xu, W., Jiang, W., Zha, Z., et
al. (2009) Glioma-derived mutations in IDH1 dominantly inhibit IDH1
catalytic activity and induce HIF-1a, Science, 324,
261-265, doi: 10.1126/science.1170944.
14.Liu, S., Cadoux-Hudson, T., and Schofield, C. J.
(2020) Isocitrate dehydrogenase variants in cancer – cellular
consequences and therapeutic opportunities, Curr. Opin. Chem.
Biol., 57, 122-134, doi: 10.1016/j.cbpa.2020.06.012.
15.Sonveaux, P., Copetti, T., and De Saedeleer, C.
J. (2012) Targeting the lactate transporter MCT1 in endothelial cells
inhibits lactate-induced HIF-1 activation and tumor angiogenesis,
PLoS One, 7, e33418, doi:
10.1371/journal.pone.0033418.
16.Jung, S. Y., Song, H. S., Park, S. Y., Chung, S.
H., and Kim, Y. J. (2011) Pyruvate promotes tumor angiogenesis through
HIF-1-dependent PAI-1 expression, Int. J. Oncol., 38,
571-576, doi: 10.3892/ijo.2010.859.
17.Hellwig-Burgel, T., Rutkowski, K., Metzen, E.,
Fandrey, J., and Jelkmann, W. (1999) Interleukin-1beta and tumor
necrosis factor-alpha stimulate DNA binding of hypoxia-inducible
factor-1, Blood, 94, 1561-1567.
18.McMahon, S., Charbonneau, M., Grandmont, S.,
Richard, D. E., and Dubois, C. M. (2006) Transforming growth factor
beta1 induces hypoxia-inducible factor-1 stabilization through
selective inhibition of PHD2 expression, J. Biol. Chem.,
281, 24171-24181.
19.Rius, J., Guma, M., Schachtrup, C., Akassoglou,
K., Zinkernagel, A. S., et al. (2008) NF-kappaB links innate immunity
to the hypoxic response through transcriptional regulation of
HIF-1alpha, Nature, 453, 807-811, doi:
10.1038/nature06905.
20.Liao, D., and Johnson, R. S. (2007) Hypoxia: a
key regulator of angiogenesis in cancer, Cancer Metastasis Rev.,
26, 281-290.
21.Lee, K., Qian, D. Z., Rey, S., Wei, Ho., Liu, J.
O., and Semenza, G. L. (2009) Anthracycline chemotherapy inhibits HIF-1
transcriptional activity and tumor-induced mobilization of circulating
angiogenic cells, Proc. Natl. Acad. Sci. USA, 106,
2353-2358.
22.Li, Z., Bao, S., Wu, Q., Wang, H., Eyler, C., et
al. (2009) Hypoxia-inducible factors regulate tumorigenic capacity of
glioma stem cells, Cancer Cell, 15, 501-513, doi:
10.1016/j.ccr.2009.03.018.
23.Wang, Y., Liu, Y., Malek, S. N., Zheng, P., and
Liu, Y. (2011) Targeting HIF1α eliminates cancer stem cells in
hematological malignancies, Cell Stem Cell, 8, 399-411,
doi: 10.1016/j.stem.2011.02.006.
24.Korshunov, D. A., Kondakova, I. V., and Shashova,
E. E. (2019) Modern perspective on metabolic reprogramming in malignant
neoplasms, Biochemistry (Moscow), 84, 1129-1142, doi:
10.1134/S000629791910002X.
25.Vaupel, P., Schmidberger, H., and Mayer, A.
(2019) The Warburg effect: essential part of metabolic reprogramming
and central contributor to cancer progression, Int. J. Radiat.
Biol., 95, 912-919, doi: 10.1080/09553002.2019.1589653.
26.Lau, C. K., Yang, Z. F., Ho, D. W., Ng, M. N.,
Yeoh, G. C., et al. (2009) An Akt/hypoxia-inducible
factor-1alpha/platelet-derived growth factor-BB autocrine loop mediates
hypoxia-induced chemoresistance in liver cancer cells and tumorigenic
hepatic progenitor cells, Clin. Cancer Res., 15,
3462-3471, doi: 10.1158/1078-0432.CCR-08-2127.
27.Esteban, M. A., Tran, M., Harten, S. K., Hill,
P., Castellanos, M. C., et al. (2006) Regulation of E-cadherin
expression by VHL and hypoxia-inducible factor, Cancer Res.,
66, 3567-3575.
28.Kim, K., Lu, Z., and Hay, E. D. (2002) Direct
evidence for a role of beta-catenin/LEF-1 signaling pathway in
induction of EMT, Cell Biol. Int., 26, 463-476.
29.Krishnamachary, B., Zagzag, D., Nagasawa, H.,
Rainey, K., Okuyama, H., et al. (2006) Hypoxia-inducible
factor-1-dependent repression of E-cadherin in von Hippel-Lindau tumor
suppressor-null renal cell carcinoma mediated by TCF3, ZFHX1A, and
ZFHX1B, Cancer Res., 66, 2725-2731, doi:
10.1158/0008-5472.CAN-05-3719.
30.Lamouille, S., Xu, J., and Derynck, R. (2014)
Molecular mechanisms of epithelial–mesenchymal transition,
Nat. Rev. Mol. Cell Biol., 15, 178-196.
31.Krishnamachary, B., Berg-Dixon, S., and Kelly, B.
(2003) Regulation of colon carcinoma cell invasion by hypoxia-inducible
factor 1, Cancer Res., 63, 1138-1143.
32.Pennacchietti, S., Michieli, P., Galluzzo, M.,
Mazzone, M., Giordano, S., and Comoglio, P. M. (2003) Hypoxia promotes
invasive growth by transcriptional activation of the met protooncogene,
Cancer Cell, 3, 347-361.
33.Erler, J. T., Bennewith, K. L., Nicolau, M.,
Dornhöfer, N., Kong, C., et al. (2006) Lysyl oxidase is essential
for hypoxia-induced metastasis, Nature, 440, 1222-1226,
doi: 10.1038/nature04695.
34.Liu, Z. J., Semenza, G. L., and Zhang, H. F.
(2015) Hypoxia-inducible factor 1 and breast cancer metastasis, J.
Zhejiang Univ. Sci., 16, 32-43.
35.Zhang, H., Wong, C. C., Wei, H., Gilkes, D. M.,
Korangath, P., et al. (2011) HIF-1-dependent expression of
angiopoietin-like 4 and L1CAM mediates vascular metastasis of hypoxic
breast cancer cells to the lungs, Oncogene, 31,
1757-1770, doi: 10.1038/onc.2011.365.
36.Moeller, B. J., Richardson, R. A., and Dewhirst,
M. W. (2007) Hypoxia and radiotherapy: opportunities for improved
outcomes in cancer treatment, Cancer Metastas. Rev., 26,
241-248, doi: 10.1007/s10555-007-9056-0.
37.Rohwer, N., and Cramer, T. (2011)
Hypoxia-mediated drug resistance: novel insights on the functional
interaction of HIFs and cell death pathways, Drug Resist.
Updat., 14, 191-201.
38.Lyzhko, N. A. (2017) Molecular-genetic mechanisms
of initiation, promotion and progression of tumors, Russ. J.
Biother., 16, 7-17, doi:
10.17650/1726-9784-2017-16-4-7-17.
39.Liu, Y., Yin, T., Feng, Y., Cona, M. M., Huang,
G., et al. (2015) Mammalian models of chemically induced primary
malignancies exploitable for imaging-based preclinical theragnostic
research, Quant. Imaging Med. Surg., 5, 708-729, doi:
10.3978/j.issn.2223-4292.2015.06.01.
40.Greten, F. R., and Grivennikov, S. I. (2019)
Inflammation and cancer: triggers, mechanisms, and consequences,
Immunity, 51, 27-41, doi:
10.1016/j.immuni.2019.06.025.
41.Stratton, M. R., Campbell, P. J., and Futreal, P.
A. (2009) The cancer genome, Nature, 458, 719-724.
42.Hyndman, I. J. (2016) The contribution of both
nature and nurture to carcinogenesis and progression in solid tumours,
Cancer Microenvironment, 9, 63-69, doi:
10.1007/s12307-016-0183-4.
43.Ravi, R., Mookerjee, B., Bhujwalla, Z. M.,
Sutter, C. H., Artemov, D., et al. (2000) Regulation of tumor
angiogenesis by p53-induced degradation of hypoxia-inducible factor
1α, Genes Dev., 14, 34-44.
44.Zundel, W., Schindler, C., Haas-Kogan, D., Koong,
A., Kaper, F., et al. (2000) Loss of PTEN facilitates HIF-1-mediated
gene expression, Genes Dev., 14, 391-396.
45.Zhong, H., Chiles, K., Feldser, D., Laughner, E.,
Hanrahan, C., et al. (2000) Modulation of hypoxia-inducible factor
1α expression by the epidermal growth factor/phosphatidylinositol
3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells:
implications for tumor angiogenesis and therapeutics, Cancer
Res., 60, 1541-1545.
46.Laughner, E., Taghavi, P., Chiles, K., Mahon, P.
C., and Semenza, G. L. (2001) HER2 (neu) signaling increases the rate
of hypoxia-inducible factor 1α (HIF-1α) synthesis: novel
mechanism for HIF-1-mediated vascular endothelial growth factor
expression, Mol. Cell. Biol., 21, 3995-4004.
47.Watts, E. R., and Walmsley, S. R. (2019)
Inflammation and hypoxia: HIF and PHD isoform selectivity, Trends
Mol. Med., 25, 33-46, doi: 10.1016/j.molmed.2018.10.006.
48.Koshiji, M., To, K. K., Hammer, S., Kumamoto, K.,
Harris, A. L., and Modrich, P. (2005) HIF-1alpha induces genetic
instability by transcriptionally downregulating MutSalpha expression,
Mol. Cell, 17, 793-803.
49.Zeng, M., Kikuchi, H., Pino, M. S., and Chung, D.
C. (2010) Hypoxia activates the K-Ras proto-oncogene to stimulate
angiogenesis and inhibit apoptosis in colon cancer cells, PLoS
One, 5, e10966, doi: 10.1371/journal.pone.0010966.
50.Muinao, T., Boruah, H. P. D., and Pal, M. (2018)
Diagnostic and prognostic biomarkers in ovarian cancer and the
potential roles of cancer stem cells – An updated review, Exp.
Cell. Res., 362, 1-10.
51.Nguyen, P. H., Giraud, J., Chambonnier, L.,
Dubus, P., Wittkop, L., et al. (2017) Characterization of biomarkers of
tumorigenic and chemoresistant cancer stem cells in human gastric
carcinoma, Clin. Cancer Res., 23, 1586-1597.
52.Corro, C., and Moch, H. (2018) Biomarker
discovery for renal cancer stem cells, J. Pathol. Clin. Res.,
4, 3-18.
53.Ayob, A. Z., and Ramasamy, T. S. (2018) Cancer
stem cells as key drivers of tumour progression, J. Biomed.
Sci., 25, 20, doi: 10.1186/s12929-018-0426-4.
54.Oliveira-Costa, J. P., Zanetti, J. S., Silveira,
G. G., Soave, D. F., Oliveira, L. R., et al. (2011) Differential
expression of HIF-1alpha in CD44+CD24–/low
breast ductal carcinomas, Diagn. Pathol., 6, 73, doi:
10.1186/1746-1596-6-73.
55.Kondoh, H., Lleonart, M. E., Gil, J., Wang, J.,
Degan, P., et al. (2005) Glycolytic enzymes can modulate cellular life
span, Cancer Res., 65, 177-185.
56.Mathieu, J., Zhang, Z., Zhou, W., Wang, A. J.,
Heddleston, J. M., et al. (2011) HIF induces human embryonic stem cell
markers in cancer cells, Cancer Res., 71, 4640-4652, doi:
10.1158/0008-5472.CAN-10-3320.
57.Nishi, H., Nakada, T., Kyo, S., Inoue, M., Shay,
J. W., and Isaka, K. (2004) Hypoxia-inducible factor 1 mediates
upregulation of telomerase (hTERT), Mol. Cell. Biol., 24,
6076-6083, doi: 10.1128/MCB.24.13.6076-6083.2004.
58.Mu, R., Zou, Y. K., Tu, K., Wang, D. B., Tang,
D., et al. (2021) Hypoxia promotes pancreatic cancer cell
dedifferentiation to stem-like cell phenotypes with high tumorigenic
potential by the HIF-1α/Notch signaling pathway, Pancreas,
50, 756-765, doi: 10.1097/MPA.0000000000001828.
59.Gunaratnam, L., Morley, M., Franovic, A., de
Paulsen, N., Mekhail, K., et al. (2003) Hypoxia inducible factor
activates the transforming growth factor-α/epidermal growth
factor receptor growth stimulatory pathway in
VHL–/– renal cell carcinoma cells, J. Biol.
Chem., 278, 44966-44974, doi: 10.1074/jbc.M305502200.
60.Forsythe, J. A., Jiang, B. H., Iyer, N. V.,
Agani, F., Leung, S. W., et al. (1996) Activation of vascular
endothelial growth factor gene transcription by hypoxia-inducible
factor 1, Mol. Cell. Biol., 16, 4604-4613.
61.Feldser, D., Agani, F., Iyer, N. V., Pak, B.,
Ferreira, G., Semenza, G. L. (1999) Reciprocal positive regulation of
hypoxia-inducible factor 1α and insulin-like growth factor 2,
Cancer Res., 59, 3915-3918.
62.Dallas, N. A., Fan, F., Gray, M. J., Van Buren,
G., Lim, S. J., et al. (2007) Functional significance of vascular
endothelial growth factor receptors on gastrointestinal cancer cells,
Cancer Metastasis Rev., 26, 433-441, doi:
10.1007/s10555-007-9070-2.
63.Grimshaw, M. J. (2007) Endothelins and
hypoxia-inducible factor in cancer, Endocr. Relat. Cancer,
14, 233-244.
64.Larráyoz, I. M., Martínez-Herrero,
S, García-Sanmartín, J., Ochoa-Callejero, L., and
Martínez, A. (2014) Adrenomedullin and tumour microenvironment,
J. Transl. Med., 12, 339, doi:
10.1186/s12967-014-0339-2.
65.Szenajch, J., Wcislo, G., Jeong, J. Y., Szczylik,
C., and Feldman, L. (2010) The role of erythropoietin and its receptor
in growth, survival and therapeutic response of human tumor cells. From
clinic to bench – a critical review, Biochim. Biophys.
Acta, 1806, 82-95, doi: 10.1016/j.bbcan.2010.04.002.
66.Keith, B., and Simon, M. C. (2007)
Hypoxia-inducible factors, stem cells, and cancer, Cell,
129, 465-472.
67.Warburg, O. (1925) The metabolism of carcinoma
cells, J. Cancer Res., 9, 148-163, doi:
10.1158/jcr.1925.148.
68.Dang, C. V., and Semenza, G. L. (1999) Oncogenic
alterations of metabolism, Trends Biochem. Sci., 24,
68-72.
69.Kobliakov, V. A. (2019) The mechanisms of
regulation of aerobic glycolysis (Warburg effect) by oncoproteins in
carcinogenesis, Biochemistry (Moscow), 84, 1117-1128,
doi: 10.1134/S0006297919100018.
70.Shanmugam, M., McBrayer, S. K., and Rosen, S. T.
(2009) Targeting the Warburg effect in hematological malignancies: from
PET to therapy, Curr. Opin. Oncol., 21, 531-536, doi:
10.1097/CCO.0b013e32832f57ec.
71.Semenza, G. L. (2009) Regulation of cancer cell
metabolism by hypoxia-inducible factor 1, Semin. Cancer Biol.,
19, 12-16, doi: 10.1016/j.semcancer.2008.11.009.
72.Brizel, D. M., Schroeder, T., Scher, R. L.,
Walenta, S., Clough, R. W., and Dewhirst, M. W. (2001) Elevated tumor
lactate concentrations predict for an increased risk of metastases in
head-and-neck cancer, Int. J. Radiat. Oncol. Biol. Phys.,
51, 349-353.
73.Petrova, V., Annicchiarico-Petruzzelli, M., and
Melino, G. (2018) The hypoxic tumour microenvironment,
Oncogenesis, 7, 10, doi: 10.1038/s41389-017-0011-9.
74.Ammirante, M., Shalapour, S., Kang, Y., Jamieson,
C. A., and Karin, M. (2014) Tissue injury and hypoxia promote malignant
progression of prostate cancer by inducing CXCL13 expression in tumor
myofibroblasts, Proc. Natl. Acad. Sci. USA, 111,
14776-14781.
75.Gilkes, D. M., Semenza, G. L., and Wirtz, D.
(2014) Hypoxia and the extracellular matrix: drivers of tumour
metastasis, Nat. Rev. Cancer, 14, 430-439.
76.Semenza, G. L. (2008) Tumor metabolism: cancer
cells give and take lactate, J. Clin. Invest., 118,
3835-3837.
77.Estrella, V., Chen, T., Lloyd, M., Wojtkowiak,
J., Cornnell, H. H., et al. (2013) Acidity generated by the tumor
microenvironment drives local invasion, Cancer Res., 73,
1524-1535, doi: 10.1158/0008_5472.CAN_12_2796.
78.Brown, J. M., and Wilson, W. R. (2004) Exploiting
tumour hypoxia in cancer treatment, Nat. Rev. Cancer, 4,
437-447.
79.Nordsmark, M., Hoyer, M., Keller, J., Nielsen, O.
S., Jensen, O. M., and Overgaard, J. (1996) The relationship between
tumor oxygenation and cell proliferation in human soft tissue sarcomas,
Int. J. Radiat. Oncol. Biol. Phys., 35, 701-708, doi:
10.1016/0360-3016(96)00132-0.
80.Nordsmark, M., Overgaard, M., and Overgaard, J.
(1996) Pretreatment oxygenation predicts radiation response in advanced
squamous cell carcinoma of the head and neck, Radiother. Oncol.,
41, 31-39, doi: 10.1016/S0167-8140(96)91811-3.
81.Höckel, M., and Vaupel, P. (2001) Tumor
hypoxia: definitions and current clinical, biologic, and molecular
aspects, J. Natl. Cancer Inst., 93, 266-276, doi:
10.1093/jnci/93.4.266.
82.Höckel, M., Schlenger, K., Aral, B., Mitze,
M., Schaffer, U., and Vaupel, P. (1996) Association between tumor
hypoxia and malignant progression in advanced cancer of the uterine
cervix, Cancer Res., 56, 4509-4515.
83.Vaupel, P., Hockel, M., and Mayer, A. (2007)
Detection and characterization of tumor hypoxia using pO2 histography,
Antioxid. Redox Signal., 9, 1221-1235, doi:
10.1089/ars.2007.1628.
84.Le, Q. T., Chen, E., Salim, A., Cao, H., Kong, C.
S., and Whyte, R. (2006) An evaluation of tumor oxygenation and gene
expression in patients with early stage non-small cell lung cancers,
Clin. Cancer Res., 12, 1507-1514, doi:
10.1158/1078-0432.CCR-05-2049.
85.Koong, A. C., Mehta, V. K., Le, Q. T., Fisher, G.
A., Terris, D. J., and Brown, J. M. (2000) Pancreatic tumors show high
levels of hypoxia, Int. J. Radiat. Oncol. Biol. Phys.,
48, 919-922.
86.Parker, C., Milosevic, M., Toi, A., Sweet, J.,
Panzarella, T., and Bristow, R. (2004) Polarographic electrode study of
tumor oxygenation in clinically localized prostate cancer, Int. J.
Radiat. Oncol. Biol. Phys., 58, 750-757, doi:
10.1016/S0360-3016(03)01621-3.
87.Lartigau, E., Randrianarivelo, H., Avril, M. F.,
Margulis, A., Spatz, A., et al. (1997) Intratumoral oxygen tension in
metastatic melanoma, Melanoma Res., 7, 400-406.
88.Xiu, Lv, Li, J., Zhang, C., Hu, T., Li, S., et
al. (2017) The role of hypoxia-inducible factors in tumor angiogenesis
and cell metabolism, Genes Diseases, 4, 19-24, doi:
10.1016/j.gendis.2016.11.003.
89.Carmeliet, P., and Jain, R. K. (2011) Principles
and mechanisms of vessel normalization for cancer and other angiogenic
diseases, Nat. Rev. Drug Discov., 10, 417-427, doi:
10.1038/nrd3455.
90.Lee, K., Zhang, H., Qian, D. Z., Rey, S., Liu, J.
O., and Semenza, G. L. (2009) Acriflavine inhibits HIF-1 dimerization,
tumor growth, and vascularization, Proc. Natl. Acad. Sci. USA,
106, 17910-17915, doi: 10.1073/pnas.0909353106.
91.Tang, N., Wang, L., Esko, J., Giordano, F. J.,
Huang, Y., et al. (2004) Loss of HIF-1alpha in endothelial cells
disrupts a hypoxia-driven VEGF autocrine loop necessary for
tumorigenesis, Cancer Cell, 6, 485-495.
92.Skuli, N., Majmundar, A. J., Krock, B. L.,
Mesquita, R. C., Mathew, L. K., et al. (2012) Endothelial HIF-2alpha
regulates murine pathological angiogenesis and revascularization
processes, J. Clin. Invest., 122, 1427-1443.
93.Staller, P., Sulitkova, J., Lisztwan, J., Moch,
H., Oakeley, E. J., and Krek, W. (2003) Chemokine receptor CXCR4
downregulated by von Hippel–Lindau tumour suppressor pVHL,
Nature, 425, 307-311.
94.Zavyalova, M. V., Denisov, E. V., Tashireva, L.
A., Savelieva, O. E., Kaigorodova, E. V., et al. (2019) Intravasation
as a key step in cancer metastasis, Biochemistry (Moscow),
84, 762-772, doi: 10.1134/S0320972519070078.
95.Burtscher, M., Mairer, K., Wille, M., Gatterer,
H., Ruedl, G., et al. (2012) Short-Term exposure to hypoxia for work
and leisure activities in health and disease: which level of hypoxia is
safe? Sleep Breathing, 16, 435-442, doi:
10.1007/s11325-011-0521-1.
96.Zarubina, I. V. (2011) Modern view on
pathogenesis of hypoxia and its pharmacological correction, Rev.
Clin. Pharmacol. Drug Ther., 9, 31-48.
97.Kirova, Y. I., Germanova, E. L., and Lukyanova,
L. D. (2013) Phenotypic features of the dynamics of HIF-1α levels
in rat neocortex in different hypoxia regimens, Bull. Exp. Biol.
Med., 154, 718-722, doi: 10.1007/s10517-013-2038-z.
98.Colgan, S. P., Furuta, G. T., and Taylor, C. T.
(2020) Hypoxia and innate immunity: keeping up with the HIFsters,
Annu. Rev. Immunol., 26, 341-363, doi:
10.1146/annurev-immunol-100819-121537.
99.Zhong, H., De Marzo, A. M., Laughner, E., Lim,
M., Hilton, D. A., et al. (1999) Overexpression of hypoxia-inducible
factor 1α in common human cancers and their metastases, Cancer
Res., 59, 5830-5835.
100.Talks, K. L., Turley, H., Gatter, K. C.,
Maxwell, P. H., Pugh, C. W., et al. (2000) The expression and
distribution of the hypoxia-inducible factors HIF-1α and
HIF-2α in normal human tissues, cancers, and tumor-associated
macrophages, Am. J. Pathol., 157, 411-421.
101.Flamme, I., Krieg, M., and Plate, K. H. (1998)
Up-regulation of vascular endothelial growth factor in stromal cells of
hemangioblastomas is correlated with up-regulation of the transcription
factor HRF/HIF-2alpha, Am. J. Pathol., 153, 25-29, doi:
10.1016/s0002-9440(10)65541-1.
102.Krieg, M., Haas, R., Brauch, H., Acker, T.,
Flamme, I., and Plate, K. H. (2000) Up-regulation of hypoxia-inducible
factors HIF-1α and HIF-2α under normoxic conditions in
renal carcinoma cells by von Hippel-Lindau tumor suppressor gene loss
of function, Oncogene, 19, 5435-5443, doi:
10.1038/sj.onc.1203938.
103.Giatromanolaki, A., Koukourakis, M. I., and
Sivridis, E. (2001) Relation of hypoxia inducible factor 1 alpha and 2
alpha in operable non-small cell lung cancer to angiogenic/molecular
profile of tumours and survival, Br. J. Cancer, 85,
881-890, doi: 10.1054/bjoc.2001.2018.
104.Wong, C. C., Zhang, H., Gilkes, D. M., Chen,
J., Wei, H., et al. (2012) Inhibitors of hypoxia-inducible factor 1
block breast cancer metastatic niche formation and lung metastasis,
J. Mol. Med. (Berl), 90, 803-815.
105.Chen, W., He, D., Li, Z., Zhang, X., Pan, D.,
and Chen, G. (2015) Overexpression of vascular endothelial growth
factor indicates poor outcomes of glioma: a systematic review and
meta-analysis, Int. J. Clin. Exp. Med., 15,
8709-8719.
106.Kaur, B., Khwaja, F. W., Severson, E. A.,
Matheny, S. L., Brat, D. J., and Van Meir, E. G. (2005) Hypoxia and the
hypoxia-inducible-factor pathway in glioma growth and angiogenesis,
Neuro Oncol., 7, 134-153, doi:
10.1215/S1152851704001115.
107.Shen, C., Beroukhim, R., Schumacher, S. E.,
Zhou, J., Chang, M., and Signoretti, S. (2011) Genetic and functional
studies implicate HIF1alpha as a 14q kidney cancer suppressor gene,
Cancer Discov., 1, 222-235.
108.Birner, P., Schindl, M., Obermair, A.,
Breitenecker, G., and Oberhuber, G. (2001) Expression of
hypoxia-inducible factor 1α in epithelial ovarian tumors: its
impact on prognosis and on response to chemotherapy, Clin. Cancer
Res., 7, 1661-1668.
109.Koukourakis, M. I., Giatromanolaki, A.,
Skarlatos, J., Corti, L., Blandamura, S., et al. (2001) Hypoxia
inducible factor (HIF-1α and HIF-2α) expression in early
esophageal cancer and response to photodynamic therapy and
radiotherapy, Cancer Res., 61, 1830-1832.
110.Mazumdar, J., Hickey, M. M., Pant, D. K.,
Durham, A. C., Sweet-Cordero, A., et al. (2010) HIF-2alpha deletion
promotes Kras-driven lung tumor development, Proc. Natl. Acad. Sci.
USA, 107, 14182-14187.
111.Triner, D., and Shah, Y. M. (2016)
Hypoxia-inducible factors: a central link between inflammation and
cancer, J. Clin. Invest., 126, 3689-3698, doi:
10.1172/JCI84430.
112.Wenwu, L., Xuejun, S., Hengyi, T., and Kan, L.
(2013) Hyperbaric oxygen and cancer: more complex than we expected,
Target. Oncol., 8, 79-81.
113.Mallory, M., Gogineni, E., and Jones, G. C.
(2015) Therapeutic hyperthermia: the old, the new, and the upcoming,
Crit. Rev. Oncol. Hematol., 97, 56-64, doi:
10.1016/j.critrevonc.2015.08.003.
114.Fallah, J., and Rini, B. I. (2019) HIF
inhibitors: status of current clinical development, Curr. Oncol.
Rep., 21, 6, doi: 10.1007/s11912-019-0752-z.
115.Greenberger, L. M., Horak, I. D., Filpula, D.,
Sapra, P., Westergaard, M., et al. (2008) A RNA antagonist of
hypoxia-inducible factor-1 alpha, EZN-2968, inhibits tumor cell growth,
Mol. Cancer Ther., 7, 3598-3608.
116.Lee, K., and Kim, H. M. (2011) A novel approach
to cancer therapy using PX-478 as a HIF-1α inhibitor, Arch.
Pharm. Res., 34, 1583-1585.
117.Terzuoli, E., Puppo, M., Rapisarda, A.,
Uranchimeg, B., Cao, L., et al. (2010) Aminoflavone, a ligand of the
aryl hydrocarbon receptor, inhibits HIF-1alpha expression in an
AhR-independent fashion, Cancer Res., 70, 6837-6848.
118.Coltella, N., Valsecchi, R., Ponente, M.,
Ponzoni, M., and Bernardi, R. (2015) Synergistic leukemia eradication
by combined treatment with retinoic acid and HIF inhibition by EZN-2208
(PEG-SN38) in preclinical models of PML-RARα and
PLZF-RARα-driven leukemia, Clin. Cancer Res., 21,
3685-3694.
119.Rapisarda, A., Zalek, J., Hollingshead, M.,
Braunschweig, T., Uranchimeg, B., et al. (2004) Schedule-dependent
inhibition of hypoxia-inducible factor-1alpha protein accumulation,
angiogenesis, and tumor growth by topotecan in U251-HRE glioblastoma
xenografts, Cancer Res., 64, 6845-6848.
120.Zhang, H., Qian, D. Z., Tan, Y. S., Lee, K.,
Gao, P., et al. (2008) Digoxin and other cardiac glycosides inhibit
HIF-1alpha synthesis and block tumor growth, Proc. Natl. Acad. Sci.
USA, 105, 19579-19586.
121.Ning, X., Wang, Y., Yan, W., Li, G., and Sang,
N. (2018) Chitin synthesis inhibitors promote liver cancer cell
metastasis via interfering with hypoxia-inducible factor 1α,
Chemosphere, 206, 231-237, doi:
10.1016/j.chemosphere.2018.05.014.
122.Mabjeesh, N. J., Escuin, D., LaVallee, T. M.,
Pribluda, V. S., Swartz, G. M., et al. (2003) 2ME2 inhibits tumor
growth and angiogenesis by disrupting microtubules and dysregulating
HIF, Cancer Cell, 3, 363-375.
123.Alqawi, O., Moghaddas, M., and Singh, G. (2006)
Effects of geldanamycin on HIF-1alpha mediated angiogenesis and
invasion in prostate cancer cells, Prostate Cancer Prostatic
Dis., 9, 126-135, doi: 10.1038/sj.pcan.4500852.
124.Xiang, L., Gilkes, D. M., Chaturvedi, P., Luo,
W., Hu, H., et al. (2014) Ganetespib blocks HIF-1 activity and inhibits
tumor growth, vascularization, stem cell maintenance, invasion, and
metastasis in orthotopic mouse models of triple-negative breast cancer,
J. Mol. Med., 92, 151-164.
125.Bailey, C. M., Liu, Y., Peng, G., Zhang, H.,
He, M., et al. (2020) Liposomal formulation of HIF-1α inhibitor
echinomycin eliminates established metastases of triple-negative breast
cancer, Nanomedicine, 29, 102278, doi:
10.1016/j.nano.2020.102278.
126.Novikov, V. Y., and Levchenkova, O. S. (2013)
Hypoxia-inducible factor as a pharmacological target, Rev. Clin.
Pharmacol. Drug Ther., 11, 8-16, doi:
10.17816/RCF1128-16.
127.Sen Banerjee, S., Thirunavukkarasu, M., Tipu
Rishi, M., et al. (2012) HIF-prolyl hydroxylases and cardiovascular
diseases, Toxicol. Mech. Methods, 22, 347-358.
128.Souza, E., Cho, K. H., Harris, S. T., Flindt,
N. R., Watt, R. K., and Pai, A. B. (2020) Hypoxia-inducible factor
prolyl hydroxylase inhibitors: a paradigm shift for treatment of anemia
in chronic kidney disease? Expert. Opin. Investig. Drugs,
29, 831-844, doi: 10.1080/13543784.2020.1777276.
129.Cummins, E. P., Seeballuck, F., Keely, S. J.,
Mangan, N. E., Callanan, J. J., et al. (2008) The hydroxylase inhibitor
dimethyloxalylglycine is protective in a murine model of colitis,
Gastroenterology, 134, 156-165.
130.Karkishchenko, N. N. (2017) Biomedical
(Preclinical) Study of the Antihypoxic Activity of Drugs. Methodical
Recommendations, FMBA of Russian Federation, Moscow, p. 98.
131.Fu, Z., Mowday, A. M., Smaill, J. B., Hermans,
I. F., and Patterson, A. V. (2021) Tumour hypoxia-mediated
immunosuppression: mechanisms and therapeutic approaches to improve
cancer immunotherapy, Cells, 10, 1006, doi:
10.3390/cells10051006.
132.Fracasso, P. M., and Sartorelli, A. C. (1986)
Cytotoxicity and DNA lesions produced by mitomycin C and porfiromycin
in hypoxic and aerobic EMT6 and Chinese hamster ovary cells, Cancer
Res., 46, 3939-3944, doi: 10.1016/0304-3835(86)90123-0.
133.Baran, N., and Konopleva, M. (2017) Molecular
pathways: hypoxia-activated prodrugs in cancer therapy, Clin. Cancer
Res., 23, 2382-2390.
134.Spiegelberg, L., Houben, R., Niemans, R., de
Ruysscher, D., Yaromina, A., et al. (2019) Hypoxia-activated prodrugs
and (lack of) clinical progress: the need for hypoxia-based biomarker
patient selection in phase III clinical trials, Clin. Transl.
Radiat. Oncol., 18, 62-69, doi:
10.1016/j.ctro.2019.01.005.
135.Li, Y., Zhao, L., and Li, X.-F. (2021) The
hypoxia-activated prodrug TH-302: exploiting hypoxia in cancer therapy,
Front. Pharmacol., 12, 636892, doi:
10.3389/fphar.2021.636892.
136.Milosevic, M., Fyles, A., Hedley, D., and Hill,
R. (2004) The human tumor microenvironment: invasive (needle)
measurement of oxygen and interstitial fluid pressure, Semin.
Radiat. Oncol., 14, 249-258.
137.O’Connor, J. P., Boult, J. K., Jamin, Y.,
Babur, M., Finegan, K. G., et al. (2016) Oxygen-enhanced MRI accurately
identifies, quantifies, and maps tumor hypoxia in preclinical cancer
models, Cancer Res., 76, 787-795.
138.Samanta, D., Gilkes, D. M., Chaturvedi, P.,
Xiang, L., and Semenza, G. L. (2014) Hypoxia-inducible factors are
required for chemotherapy resistance of breast cancer stem cells,
Proc. Natl. Acad. Sci. USA, 111, E5429-5438.
139.Flamant, L., Notte, A., Ninane, N., Raes, M.,
and Michiels, C. (2010) Anti-apoptotic role of HIF-1 and AP-1 in
paclitaxel exposed breast cancer cells under hypoxia, Mol.
Cancer, 9, 191.
140.Graham, K., and Unger, E. (2018) Overcoming
tumor hypoxia as a barrier to radiotherapy, chemotherapy and
immunotherapy in cancer treatment, Int. J. Nanomed., 13,
6049.
141.Fu, Z., Chen, D., Cheng, H., and Wang, F.
(2015) Hypoxia-inducible factor-1 protects cervical carcinoma cells
from apoptosis induced by radiation via modulation of vascular
endothelial growth factor and p53 under hypoxia, Med. Sci.
Monit., 21, 318-325.
142.Peng, C., Liu, G., Huang, K., Zheng, Q., Li,
Y., and Yu, C. (2018) Hypoxia-induced upregulation of HE4 is
responsible for resistance to radiation therapy of gastric cancer,
Mol. Ther. Oncolytics, 12, 49-55.
143.Hassouna, I., Sperling, S., Kim, E.,
Schulz-Schaeffer, W., Rave-Fränk, M., et al. (2008) Erythropoietin
augments survival of glioma cells after radiation and temozolomide,
Int. J. Radiat. Oncol. Biol. Phys., 72, 927-934.
144.Milne, A. N., Carneiro, F., O’Morain, C.,
and Offerhaus, G. J. A. (2009) Nature meets nurture: molecular genetics
of gastric cancer, Hum. Genet., 126, 615-628.
145.Perera, F. P. (1997) Environment and cancer:
who are susceptible? Science, 278, 1068-1073.
146.Brooks, J. T., Elvidge, G. P., Glenny, L.,
Gleadle, J. M., Liu, C., et al. (2008) Variations within
oxygen-regulated gene expression in humans, J. Appl. Physiol.,
106, 212-220, doi: 10.1152/japplphysiol.90578.2008.
147.Van Patot, M. C., and Gassmann, M. (2011)
Hypoxia: adapting to high altitude by mutating EPAS-1, the gene
encoding HIF-2a, High Alt. Med. Biol., 12, 157-167, doi:
10.1089/ham.2010.1099.
148.Li, D., Liu, J., Zhang, W., Ren, J., Yan, L.,
and Liu, H. (2013) Association between HIF1A P582S and A588T
polymorphisms and the risk of urinary cancers: a meta-analysis, PLoS
One, 8, e63445.
149.Li, Hu. N., He, T., Yong, J. Z., Fang, D., Liu,
J., et al. (2019) HIF-1α Rs11549465 C>T Polymorphism
contributes to increased cancer susceptibility: evidence from 49
studies, J. Cancer, 10, 5955-5963, doi:
10.7150/jca.35716.
150.Jain, K., Suryakumar, G., Prasad, R., and
Ganju, L. (2013) Upregulation of cytoprotective defense mechanisms and
hypoxia-responsive proteins imparts tolerance to acute hypobaric
hypoxia, High Alt. Med. Biol., 14, 65-77, doi:
10.1089/ham.2012.1064.
151.Dzhalilova, D. S., Kosyreva, A. M., Diatroptov,
M. E., Ponomarenko, E. A., Tsvetkov, I. S., et al. (2019) Dependence of
the severity of the systemic inflammatory response on resistance to
hypoxia in male Wistar rats, J. Inflamm. Res., 12, 73-86,
doi: 10.2147/JIR.S194581.
152.Belosludtsev, K. N., Dubinin, M. V., Talanov,
E. Y., Starinets, V. S., Tenkov, K. S., et al. (2020) Transport of
Ca2+ and Ca2+-dependent permeability transition
in the liver and heart mitochondria of rats with different tolerance to
acute hypoxia, Biomolecules, 10, 114, doi:
10.3390/biom10010114.
153.Dzhalilova, D., and Makarova, O. (2020)
Differences in tolerance to hypoxia: physiological, biochemical, and
molecular-biological characteristics, Biomedicines, 8,
428, doi: 10.3390/biomedicines8100428.
154.Glazachev, O. S., Geppe, N. A., Timofeev, Yu.
S., Samartseva, V. G., Dudnik, E. N., et al. (2020) Indicators of
individual hypoxia resistance - a way to optimize hypoxic training for
children, Ros. Vestn. Perinatol. Pediatr., 65, 78-84,
doi: 10.21508/1027-4065-2020-65-4-78-84.
155.Lu, H., Wang, R., Li, W., Xie, H., Wang, C., et
al. (2016) Plasma cytokine profiling to predict susceptibility to acute
mountain sickness, Eur. Cytokine Netw., 27, 90-96, doi:
10.1684/ecn.2016.0383.
156.Soree, P., Gupta, R. K., Singh, K., Desiraju,
K., Agrawal, A., et al. (2016) Raised HIF1α during normoxia in
high altitude pulmonary edema susceptible non-mountaineers, Sci.
Rep., 6, 26468, doi: 10.1038/srep26468.
157.Sinha, S., Ray, U. S., Tomar, O. S., and Singh,
S. N. (2009) Different adaptation patterns of antioxidant system in
natives and so-journers at high altitude, Respir. Physiol.
Neurobiol., 167, 255-260.
158.Dzhalilova, D. S., Diatroptov, M. E., Tsvetkov,
I. S., Makarova, O. V., and Kuznetsov, S. L. (2018) Expression of
Hif-1α, NF-kB, and Vegf genes in the liver and blood serum levels
of HIF-1α, erythropoietin, VEGF, TGF-β, 8-isoprostane, and
corticosterone in Wistar rats with high and low resistance to hypoxia,
Bull. Exp. Biol. Med., 165, 781-785, doi:
10.1007/s10517-018-4264-x.
159.Hsieh, C. H., Lee, C. H., Liang, J. A., Yu, C.
Y., and Shyu, W. C. (2010) Cycling hypoxia increases U87 glioma cell
radioresistance via ROS induced higher and long-term HIF-1 signal
transduction activity, Oncol. Rep., 24, 1629-1636.
160.Dzhalilova, D. Sh., Polyakova, M. A.,
Diatroptov, M. E., Zolotova, N. A., and Makarova, O. V. (2018)
Morphological changes in the colon and composition of peripheral blood
lymphocytes in acute colitis in mice with different resistance to
hypoxia, Mol. Med., 6, 46-50, doi:
10.29296/24999490-2018-06-08.
161.Dzhalilova, D. Sh., Zolotova, N. A., Polyakova,
M. A., Diatroptov, M. E., Dobrynina, M. T., and Makarova, O. V. (2018)
Morphological features of the inflammatory process and subpopulation
pattern of peripheral blood lymphocytes during chronic colitis in mice
exhibiting different responses to hypoxia, Clin. Exp. Morphol.,
4, 13-19, doi: 10.31088/2226-5988-2018-28-4-13-20.
162.Fridman, I. A., Ponomarenko, E. A., Makarova,
O. V., Postovalova, E. A., Zolotova, N. A., et al. (2020) Morphological
characteristic of melanoma B16 progression in C57BL/6 mice with high
and low resistance to hypoxia, Bull. Exp. Biol. Med.,
168, 390-394, doi: 10.1007/s10517-020-04716-w.
163.Giatromanolaki, A., Sivridis, E., Kouskoukis,
C., Gatter, K. C., Harris, A. L., and Koukourakis, M. I. (2003)
Hypoxia-inducible factors 1alpha and 2alpha are related to vascular
endothelial growth factor expression and a poorer prognosis in nodular
malignant melanomas of the skin, Melanoma Res., 13,
493-501, doi: 10.1097/00008390-200310000-00008.
164.Valencak, J., Kittler, H., Schmid, K.,
Schreiber, M., Raderer, M., and Gonzalez-Inchaurraga, M. (2009)
Prognostic relevance of hypoxia inducible factor-1alpha expression in
patients with melanoma, Clin. Exp. Dermatol., 34,
e962-e964, doi: 10.1111/j.1365-2230.2009.03706.x.
165.Park, T. J., Reznick, J., Peterson, B. L.,
Blass, G., Omerbašić, D., et al. (2017) Fructose-driven
glycolysis supports anoxia resistance in the naked mole-rat,
Science, 21, 307-311, doi: 10.1126/science.aab3896.
166.Delaney, M. A., Ward, J. M., Walsh, T. F.,
Chinnadurai, S. K., Kerns, K., et al. (2016) Initial case reports of
cancer in naked mole-rats (Heterocephalus glaber), Vet.
Pathol., 53, 691-696.
167.Xiao, B., Wang, S., Yang, G., Sun, X., Zhao,
S., et al. (2017) HIF-1α contributes to hypoxia adaptation of the
naked mole rat, Oncotarget, 8, 109941-109951, doi:
10.18632/oncotarget.22767.
168.Augustin, L. S., Kendall, C. W., Jenkins, D.
J., Willett, W. C., Astrup, A., et al. (2015) Glycemic index, glycemic
load and glycemic response: An International Scientific Consensus
Summit from the International Carbohydrate Quality Consortium (ICQC),
Nutr. Metab. Cardiovasc. Dis., 25, 795-815.
169.Duan, W., Shen, X., Lei, J., Xu, Q., Yu, Y., et
al. (2014) Hyperglycemia, a neglected factor during cancer progression,
Biomed Res. Int., 2014, 461917.
170.Chen, J. (2011) Multiple signal pathways in
obesity-associated cancer, Obes. Rev., 12, 1063-1070.
171.Ranhotra, H. S., and Sharma, R. (2010) Moderately high altitude
habitation modulates lipid profile and alkaline phosphatase activity in
aged Khasis of Meghalaya, IJCB, 25, 51-56.
172.Weinberg, C. R., Brown, K. G., and Hoel, D. G.
(1987) Altitude, radiation, and mortality from cancer and heart
disease, Radiat. Res., 112, 381-390.
173.Hart, J. (2013) Land elevation and cancer
mortality in US. Cities and counties using median elevations derived
from geographic information systems, Dose Response, 11,
41-48.
174.Simeonov, K. P., and Himmelstein, D. S. (2015)
Lung cancer incidence decreases with elevation: evidence for oxygen as
an inhaled carcinogen, PeerJ., 3, e705, doi:
10.7717/peerj.705.
175.Amsel, J., Waterbor, J. W., Oler, J.,
Rosenwaike, I., and Marshall, K. (1982) Relationship of site-specific
cancer mortality rates to altitude, Carcinogenesis, 3,
461-465.
176.Aceituno-Madera, P., Buendia-Eisman, A., Olmo,
F. J., Jimenez-Moleon, J. J., and Serrano-Ortega, S. (2011) Melanoma,
altitude, and UV-B radiation, Actas Dermosifiliogr., 102,
199-205.
177.Astrom, K., Cohen, J. E., Willett-Brozick, J.
E., Aston, C. E., and Baysal, B. E. (2003) Altitude is a phenotypic
modifier in hereditary paraganglioma type 1: evidence for an
oxygen-sensing defect, Hum. Genet., 113, 228-237.
178.Torres, J., Correa, P., Ferreccio, C.,
Hernandez-Suarez, G., and Herrero, R. (2013) Gastric cancer incidence
and mortality is associated with altitude in the mountainous regions of
Pacific Latin America, Cancer Causes Control, 24,
249-256.
179.Garrido, D. I., and Garrido, S. M. (2018)
Cancer risk associated with living at high altitude in Ecuadorian
population from 2005 to 2014, Clujul Med., 91,
188-196.
180.Kabuto, M., Imai, H., Yonezawa, C., Neriishi,
K., Akiba, S., et al. (1994) Prediagnostic serum selenium and zinc
levels and subsequent risk of lung and stomach cancer in Japan,
Cancer Epidemiol. Biomarkers Prev., 3, 465-469.
181.Bikle, D. D. (2016) Extraskeletal actions of
vitamin D, Ann. NY Acad. Sci., 1376, 29-52, doi:
10.1111/nyas.13219.
182.Merrill, R. M., and Frutos, A. M. (2020)
Ecological evidence for lower risk of lymphoma with greater exposure to
sunlight and higher altitude, High Alt. Med. Biol., 21,
37-44, doi: 10.1089/ham.2019.0054.