MINI-REVIEW: Mammal Aging is Controlled by an Evolvability-Based Adaptive Program
Theodore C. Goldsmitha
Azinet LLC, Annapolis, USA
Received June 6, 2022; Revised June 6, 2022; Accepted June 14, 2022
As recently as in 2002 gerontologists widely thought that an aging program that purposely caused aging in mammals was impossible and therefore scientifically ridiculous because it violated widely accepted concepts regarding the nature of the evolution process. However, a number of modern evolutionary mechanics concepts such as group selection and evolvability suggest that an individually adverse trait like aging can evolve if it creates an advantage (reduced probability of extinction) for a population. Genetics discoveries suggest that aging creates multiple population advantages and, therefore, aging programs that purposely cause and regulate aging evolved in mammals. This led to various concepts regarding the nature of the program. One such concept is that aging is a completely genetically specified function of age, essentially a biological clock. However, this article presents evidence and theoretical basis for the idea that the programmed aging function is controlled by an adaptive mechanism that can sense local or temporary conditions that affect the optimum aging function and adjust it to compensate for those conditions. This issue is important for medical research because the sensing mechanisms and associated signaling provide additional points at which intervention in the aging process and associated highly age-related diseases could be attempted.
KEY WORDS: programmed aging, senescence, evolution, aging theories, evolvability, anti-aging medicine, ageingDOI: 10.1134/S000629792212001X
INTRODUCTION
It is widely agreed that aging manifestations in mammals are caused by many different types of damage to many different types of cells and tissues. Damage in this context refers to any reduction in normal youthful function, and age-related diseases and conditions resulting from the different types of damage are those in which incidence and death-rate drastically increase with age such as cancer, heart disease, and Alzheimer’s disease. Many age-related diseases sometimes occur in young people and therefore can have causes other than aging. Age-related conditions are essentially universal and include loss of strength, sensory functions, and immunity, decreased reproductive function, skin and hair changes, and “death of old age”.
Early gerontology efforts involved attempts to identify a more general cause of aging damage. Perhaps the different types of damage were ultimately caused by oxidation, or free radicals, or telomere shortening, or many other proposed biochemical processes. Although some of these damage processes are likely to be involved in aging this idea fails to explain why timing of aging characteristics is extremely specific to particular mammal species even though mammals have very similar biochemistry. Different mammal species tend to share the same age-related diseases and conditions (and therefore damage types) but on greatly different schedules.
Similarly, efforts toward explaining aging as the inevitable result of some law of physics or chemistry failed to explain the huge lifespan variation between the physically and chemically similar species.
These observations led to the idea that aging and internally determined lifespans are traits or species-specific design characteristics of an organism that have been determined by the evolution process. Mammals have very large inter-species differences in their internally determined lifespans (~200 : 1 between some whales and some mice) and, as will be described, aging is very closely related to other obviously evolved traits that are controlled by complex biological mechanisms.
It should be noted that in some non-mammals, existence of an evolved suicide mechanism that purposely limits lifespan can often be explained within traditional evolutionary mechanics theory as a trade-off between enhancing current reproduction as one option and investing in survival to create the possibility of future reproduction as another option. For example, some salmon (Oncorhynchus) are apparently programmed to die shortly after reproducing [1] in such a way that their corpses plausibly provide food for their descendants. Some non-mammals are semelparous and only reproduce once, leading to similar explanations. Other non-mammals (e.g., birds) are multiparous, tend to their young, gradually age, and appear to be similar to mammals from an evolutionary viewpoint.
THE INDIVIDUAL BENEFIT CONUNDRUM
Efforts to explain mammal aging in an evolutionary context encountered immediate difficulty that continues to be controversial. Darwin (traditional evolutionary mechanics theory) proposed [2] that evolution was very closely associated with mutations and therefore individuals and can be summarized as follows:
An inheritable mutational change occasionally occurs in a single individual. If that change causes descendant individuals possessing the change to produce more descendants than the organisms not possessing the change it propagates in a population.
This idea provided plausible explanations for the vast majority of organism design characteristics but it was immediately obvious [3] that it did not explain mammal aging considered as an evolved trait. Aging clearly decreased an individual’s opportunity for reproduction because of the reduction in fitness caused by aging. According to the traditional theory the force of evolution in mammals is therefore directed toward internal immortality or absence of any internal limitation on lifespan because that would maximize an individual’s opportunity to reproduce. Subsequently, many decades of effort failed to produce aging theories based on traditional mechanics that provided even semi-plausible explanations for mammal aging. Aging was an “unsolved problem of biology” [4].
In 1952 Medawar proposed [4] a population-oriented concept to the effect that aging, although ultimately catastrophic from an individual’s viewpoint, had little impact on the wild populations of a particular mammal species. This was apparently true since aging mammal populations had presumably existed for millions of years. Wild mammal lifetimes are limited by conditions such as predation, starvation, lack of habitat, infectious diseases, intraspecies combat, and other external causes that tended to mask the internally-caused mortality from aging. If all of the mice in a particular population were dead from external causes by age X there would be no benefit to that population from having the internal ability to live longer than X and therefore no evolutionary motivation to develop and maintain that capability.
In 1957 Williams suggested [5] that the observed fitness-adverse effects of aging such as reductions in strength or speed occurred at too early an age (e.g., ~age 25 in humans) to have zero adverse effect on a population. Studies of wild mammal populations showed that death rates increased with age [6] confirming this idea. Williams proposed that therefore aging must convey some beneficial effect for a population that explained its evolution and retention. He suggested the antagonistic pleiotropy theory as a solution [5] in which aging is permanently linked to some beneficial trait(s) thus compensating for the relatively mild adverse effect of aging suggested by Medawar.
A number of traditional evolutionary mechanics concepts are important, have wide agreement, and affect subsequent discussion.
Darwin proposed [2] that evolution was extremely incremental and occurred in “tiny steps”. The evolution process is extremely accumulative and can accumulate tiny incremental advances. Mammals are descended from and incorporate design features inherited from their earliest single-cell ancestors as well as subsequent ancestors.
Organisms are systems comprising myriad design elements that work together to produce a fitness result. The ability to produce descendants depends on the combined net effect of all inherited design characteristics (traits) of an organism. For example, more speed could benefit an antelope, and longer legs could increase speed, but substantially increasing femur length would be adverse unless combined with many complementary changes to other bones, muscles, joints, etc. The tiny steps concept leads to statistical issues described below.
Evolution does not occur during an organism’s life [2]. An organism’s inheritable design is fixed during its life.
Evolutionary selection of an organism design depends on that design’s performance in producing descendants. Latent characteristics therefore cannot participate in the selection process. Adults are therefore required for evolution of adult design characteristics.
There are now two classes of very similar evolutionary aging theories that are compatible with the Medawar–Williams modifications:
Non-programmed aging theories suggest, based on the above Medawar–Williams concepts, that the force of evolution toward living longer declines with age beyond a species-specific age. Since these theories propose that there is no evolutionary disadvantage from living too long, they depend on the existence of universal natural deteriorative processes (such as random mutations) or laws of physics (such as entropy) to explain why mammals exhibit such a large variation in lifespan. Consequently, evolutionary adaptation did not resist changes that acted to reduce lifespan and cause aging manifestations beyond that age.
Programmed aging theories, also compatible with the above Medawar–Williams concepts, suggest that beyond a species-specific age there is actually an evolutionary benefit from (and evolutionary force toward) limiting lifespan by causing aging manifestations. These theories propose that although adverse as seen from an individual’s viewpoint, aging benefits survival (non-extinction) of a population in a wide variety of different ways. Aging is a feature rather than a defect in the evolved wild mammal design. Consequently, complex biological mechanisms or programs evolved to sequence the aging manifestations in the necessary species-specific function of age.
The first programmed aging theory (essentially based on evolvability) was proposed by Weismann in 1882 [7] but widely rejected because of the conflict with the traditional theory. Williams extensively attacked Weismann’s ideas and some subsequent population-oriented concepts [8].
Both theory classes involve evolutionary concepts that are more population-oriented and less individual-oriented than traditional concepts. Both classes involve modifications to the traditional theory. Both classes provide a much better fit to mammal observations than simple damage theories or fundamental limitation theories.
However, there is a major difference in viewpoint. The non-programmed theories are based on the idea that many different cells and tissues exhibit different minor defects or reductions in function that mostly occur in older individuals and collectively cause a virtually negligible impact on prehistoric animal populations. From this viewpoint the idea that mammals possess a complex evolved mechanism that exists to cause aging is patently false. Aging is a cell-level problem.
Programmed theories are based on the idea that aging is a biological function that is essential to the evolution process. Like other functions such as digestion or reproduction the aging function requires coordination of activities between different cells and tissues to accomplish the function. Although many different cells and tissues could be involved in implementing a function, logically centralized control is essential. These ideas imply that intra-organism signaling (e.g., hormonal and/or nervous) would be required as a part of the coordination scheme. Some functions (such as sexual reproduction) include inter-organism signaling (e.g., pheromones and mating behaviors) to coordinate activities of different individuals.
For most of the more than 160 years since Darwin, the idea that mammals possess what is essentially a suicide mechanism was widely seen as scientifically ridiculous. In 2002 the gerontology community issued a position statement signed by 51 gerontologists to the effect that programmed aging was “impossible” [9] based entirely on evolutionary mechanics theory considerations (as opposed to direct evidence). However, differences between the evolutionary programmed and non-programmed theories are less than they might appear. The main issue: Does the evolutionary force toward living longer decline to essentially zero at and beyond some species-specific age or does it decline to an at least minutely negative value (i.e., the force becomes toward limiting lifespan). This sort of ephemeral theoretical issue tends to be difficult to resolve. In addition, the prevailing medical research paradigm favors the idea that each disease has a different cause and therefore different treatment and largely considers aging to be a “normal” unalterable property of life.
Meanwhile, direct evidence including ongoing genetics discoveries tends to strongly favor programmed aging.
Examples:
Some human genetic diseases such as Hutchinson–Gilford progeria [10] and Werner syndrome [11] accelerate many or most manifestations of aging suggesting a common cause. This directly conflicts with the theories to the effect that the many manifestations of aging are independent of each other.
Some non-mammals apparently do not age, i.e., do not exhibit measurable manifestations of aging, and are examples of negligible senescence [12]. Since programmed aging is presumed to be controlled by a complex common mechanism, these organisms could have lost their aging function (and therefore lost the evolutionary benefits of aging) because of a defect in the common mechanism. Negligible senescence conflicts with non-programmed theories, which tend to ignore non-mammals.
Some human hormone concentrations do not vary with age. However, some decrease with age, and some increase with age [13]. The age-variant hormones could be part of the programmed aging signaling scheme. The Conboys [14] demonstrated that exposing older mammal cells to young blood produced substantial rejuvenation, further demonstrating a signaling scenario.
In addition, substantial criticisms of various specific non-programmed theories, e.g. [15, 16] and many different proposed evolutionary benefits of programmed aging have appeared in the literature (e.g. [17-20]).
Since 2002 programmed aging has therefore attracted more interest and less opposition and is discussed along with the non-programmed theories in “main-line” gerontology venues. Currently there are multiple aging theories in each class and no wide agreement between gerontologists on any particular theory or class. Modern arguments about aging theories are actually arguments about arcane details of the evolution process.
A major practical consequence for medicine is that, as described by Williams [5], the non-programmed theories suggest that there is no treatable common cause of the many different manifestations of aging, each of which has different cell-level damage mechanisms. The programmed theories suggest existence of a potentially treatable common cause (the common program mechanism), which, in effect, regulates different cell-level damage mechanisms in order to produce the species-unique aging schedule.
In addition to Medawar–Williams, a number of other population-oriented evolutionary mechanics theories that support programmed aging have been proposed:
Group selection (1962) [21] proposes that a benefit to a group can evolve despite some level of individual disadvantage. Kin selection (1963) [22] proposes that a benefit to relatives that are not direct descendants can influence evolution. The gene-oriented theories [23] suggest that the evolution process is constrained by the nature of genes such that the evolution process is much longer than previously thought. Evolvability theories (1996+) [20, 24] suggest that benefit to a population’s ability to evolve (genetically adapt) can be selected (below).
Much more detail regarding the history and status of biological aging theory can be found elsewhere (e.g., [25]).
This article discusses a particular type of evolvability-based aging program that can adapt to local or temporary conditions that affect its optimum operation.
EVOLVABILITY SUMMARY
Evolvability [20, 24] is the most recent and least-known modern population-oriented evolutionary mechanics concept. We can define the evolvability possessed by a species-population as the rapidity and precision with which the population can genetically adapt to changes in its external world. A population that could adapt more rapidly or comprehensively to change would have an evolutionary advantage that would lead to evolution of traits that enhance evolvability. In effect, organisms can evolve their ability to evolve, an idea that greatly complicates evolutionary mechanics.
Programmed aging theories based on evolvability propose that aging increases evolvability in multiple ways, especially in more complex organisms that involve features like intelligence, immunity, social structure, and even language capability.
The traditional theory assumes that the ability to evolve is an inherent property of life and therefore essentially a constant. All organisms are subject to mutations and natural selection. Evolvability theory suggests that in mammals and other diploid organisms the ability to evolve is mainly the result of obviously evolved and very complex design characteristics of their reproduction scheme that have been revealed by genetics discoveries [26]. Many other organism characteristics can affect evolvability.
STATISTICS AND EVOLUTION
Evolution is subject to the rules of statistics. For example, whether an individual having a particular inherited design produces more descendants than an individual having a slightly different design is a matter of chance. Whether a large number of individuals having a particular design reproduce more than a large number of individuals having a slightly different design is a statistically determinable factor.
Therefore, we can consider the life of an organism to be a trial of the inherited design of that individual. Is this design more likely to produce more descendants? Consequently, both the rate and precision at which evolution would nominally take place (evolvability) would be proportional to the rate at which trials were conducted, which in turn would be proportional to the size of the population and inversely proportional to average lifetime, or, put more simply, proportional to death rate. Because the death of a juvenile does not contribute to the evolution of traits that are only fully expressed in adults, we might instead say adult death rate.
A further difficulty is that the progressively more complex organisms have progressively more interactions between traits (see Antelope above) that would tend to need even more time to resolve.
These concepts suggest that the larger more complex organisms with smaller populations and longer lifetimes would nominally evolve much more slowly than the earlier smaller and simpler organisms with shorter lifetimes and larger populations.
Evolvability theory suggests that this has not occurred because of factors that increase evolvability. For example, internally limiting organism lifespan (and thereby the lengths of trials) beyond a certain species-specific age would increase evolvability.
EVOLVABILITY AND THE MECHANICS OF BIOLOGICAL INHERITANCE
Genetics discoveries [26] have exposed major issues with traditional evolutionary mechanics especially regarding evolvability.
Haploid reproduction is, as suggested by traditional theory, extremely mutation oriented. A new mutation is required for any inheritable phenotypic change.
Mammals and other diploid, sexually reproducing species, are descended from earlier haploid species, and have evolved a very much more complex biological inheritance scheme, which dramatically increases evolvability in multiple ways [20].
EVOLVABILITY ISSUES
The evolvability concept logically leads to other issues and questions.
The need for evolvability can vary. Some organisms with very long lifespans such as some clams and trees (e.g., Ocean Quahog, lifespan ~500 years; Bristlecone pine, lifespan ~5000 years) have existed for very long periods, apparently without substantial change. Mammals have been forced to adapt rapidly by changes in other mammals. Changes in predators (faster, smarter, etc.) would force changes in prey. Changes in prey would force changes in predators.
The individual benefit requirement, based on the extremely mutation-based traditional concept may be essentially valid in the simple haploid species like bacteria. However, genetics discoveries showed [26] that the sexually-reproducing diploid species have evolved a very different reproductive scheme that is much less dependent on individual mutations and individual benefit. For example, selective breeding has created very large phenotypic differences between the dog breeds in a very short time (by evolution standards). All of these intra-species differences could have been created by diploid recombination of existing mutational differences inherited from the wolf ancestor and do not require any new mutations although creation of a new species would require new mutations. Notice also that the inherited mutational differences presumably existed in a substantial portion of the wolf population and therefore have been prescreened to eliminate those that individually cause major change and therefore likely to be adverse.
Darwin’s concepts were directed at explaining the origin of species and therefore are mutation-oriented but it is clear that the diploid mechanisms and within-species changes substantially affect the evolution process. Note that the inter-breed differences include significant differences in lifespan that were accidently caused by selective breeding. Genetics discoveries [26] eventually disclosed many other details of diploid sexual reproduction that plausibly enhance the evolution process including recessive genes, the existence of linked chromosomes, transposition, introns, the genetic distance effect, and the ability of mammals to choose mates [20].
AGING ENABLES EVOLUTION OF ACQUISITION TRAITS
We can define acquisition traits as those that depend for their evolutionary value on the acquisition of something that accumulates during an organism’s life but is not genetically passed to descendants. For example, experience can be defined as the accumulative and progressive collection and retention (memory) of information about an animal’s external world and is nominally proportional to age. Intelligence can be described as the ability to process this accumulated information in a way that alters animal behavior toward increased fitness. Wisdom is essentially the product of intelligence and age, i.e., the Intelligence Quotient (IQ) concept. Wisdom increases the animal’s ability to survive and reproduce and is the property that would be selected by natural selection. The problem here is that in a non-aging population, an older, less intelligent but more experienced individual, could be wiser than a more intelligent but less experienced individual, interfering with the evolution of intelligence. Therefore, gradual aging contributes to the evolution of intelligence (and similarly immunity and language capability).
VARIATION AND DIGITAL GENETICS
Darwin specified [2] that variation in inherited design characteristics between the members of a species population was essential to the evolution process and therefore an evolvability issue. Without variation there would be no inheritable differences for natural selection to select! Darwin suggested that variation was caused by mutations, each of which originally occurred in a single individual and that natural variation was a fundamental property of life. Notice that variation is a characteristic of a population.
However, genetics discoveries, most notably the discovery of DNA’s role [27] showed that biological inheritance involves transmission of design information in digital form between the parent and descendent of any organism. Digital information transfer has advantages and constraints common to all digital information schemes [28]. One major advantage is that it spectacularly allows incremental and accumulative evolution such that current organisms incorporate design characteristics inherited from ancestors that lived billions of years ago. One limitation: variation is not a natural property of digital information schemes [28]. To illustrate, the variation that we see between mammal siblings results from complex and obviously evolved diploid mechanisms such as sexual reproduction, diploid genomic structure, pattern-matching recombination (unequal crossover), and recessive genes. Identical twins result from the occasional malfunction of these evolved mechanisms.
The vary large obviously evolved differences in the inheritance mechanisms between haploid species and diploid, sexually reproducing species have a profound effect on evolvability.
LOCAL VARIATION
Whether the squirrels in Europe are faster, smarter, or better at climbing trees than the squirrels in North America would appear to have a much slower effect on the evolution of squirrels than the local variation we see produced by the diploid variation mechanisms in mammal siblings and other species members that could plausibly directly compete with each other. The evolution process in diploid sexually reproducing organisms is not the same as the evolution process in haploid organisms.
NATURE OF THE AGING PROGRAM
A number of options exist regarding the details of an evolved aging program.
1. One concept for an aging program is that it is a mechanism that simply sequences aging manifestations as a genetically specified function of age. This function could vary greatly between different mammal species and to a lesser extent between individual members of a particular species – matching many observations. The function might operate at a cell level with each cell determining when to execute the aging function through a cell-level clock mechanism such as telomere shortening.
2. A second concept is that the cells that exhibit aging manifestations are equipped to receive and respond to inter-cellular signaling regarding the genetically-specified aging originating from some common (logically single, organism-wide) clock mechanism. Receiving the signals would cause the cell to implement the aging function in that cell. Implementation could involve cell-level damage mechanisms like age-related diseases and conditions.
3. A third concept is that the central (common) clock mechanism can alter the genetically specified aging function in response to the detection of internal or external conditions that affect optimum values for the aging function. This would allow the aging function to rapidly respond to local or temporary conditions that alter the optimum values. As described below there is extensive evidence and theoretical support for this option.
AGING AND REPRODUCTIVE CHARACTERISTICS ARE STRONGLY
RELATED
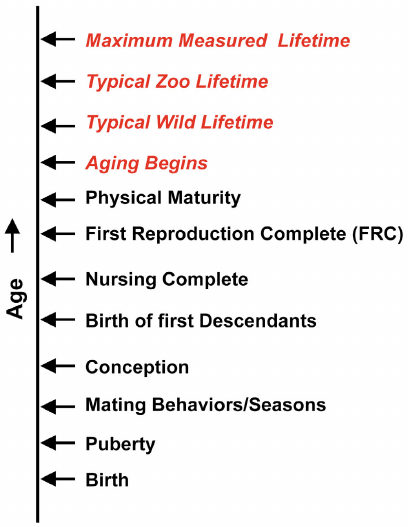
There is a very strong relationship between aging and the reproductive characteristics of a particular mammal species (figure). For example, if we consider the fitness of a female individual to be a function of age, the fitness function would be zero before the earliest age at which the animal could complete a first reproduction (FRC). This age would be determined by its age at reproductive maturity (puberty), mating rituals and processes (that delay mating), time required for pregnancy, litter size, maturity at birth, time required by the nursing stage, and other details of the particular mammal’s reproductive scheme.
Sequence of mammal life cycle events vs age (female)
Although the male fitness function could be somewhat different, depending on the details of the reproductive scheme, male reproduction depends on females.
RATIONALE FOR AN ADAPTIVE AGING PROGRAM
Reproductive functions are obviously programmed and many are controlled by adaptive programs. Example: a mating season requires the ability to sense and respond to seasonal changes. The nature of the aging program is highly dependent on the reproduction program.
Adaptive programs are common in mammals. For example, some mammals can change the genetically-specified density and color of their fur coats in response to seasonal changes [29]. This allows the animal to operate over a wider geographic range without migration. Muscle size, blood supply, and strength can be increased in response to sensing physical stress. Absence of such stress could cause a decrease in muscle mass and therefore the food supply needed by that individual, an evolutionary benefit.
The reproductive programs are obviously highly adaptive and some respond to inter-individual signaling (pheromones).
We can agree that the ability to evolve is less urgent than more immediate local or temporary threats to a population such as famine, increased predation, and overcrowding. We can therefore imagine scenarios in which modifying the aging function in response to detection of external conditions would be useful.
Mitteldorf’s programmed aging theory suggests that aging could compensate for the adverse effects of overcrowding [17]. This idea suggests that a method for detecting overcrowding and temporarily decreasing lifespan in response would be useful. Such detection could involve sensing pheromones. Apfeld and Kenyon [30] have demonstrated lifespan changes in C. elegans in response to pheromones.
Caloric restriction (semi-starvation) is observed to increase lifespan in some animals [31]. An adaptive scenario here responding to famine could involve increasing lifespan while decreasing reproduction in order to reduce the population’s need for food. Internal signals would indicate starvation. In humans, temporary fasting could signal starvation and trigger the caloric restriction response.
A logical adaptive response to local or temporary increase in predation could involve increasing reproduction and increasing lifespan. Predation of mammals is likely to be accompanied by extreme but brief increase in physical activity. Detecting predation could involve detecting internal indications of extreme activity or agitation. Exercise regimens including high-intensity interval training might simulate predation and generally delay aging.
AGING RESEARCH CONSIDERATIONS
Mice are often used in medical research because, as mammals, they share very similar biochemistry with humans. However, as described above, reproductive characteristics and aging characteristics (e.g., lifespan) differ greatly between mammals and specifically between humans and mice. Therefore, mouse data may not be directly applicable to anti-aging agent performance in humans although safety testing of the prospective anti-agents in mice is still valid.
If aging is controlled by an evolved biological function, signaling is likely to be a major factor. Interfering with signaling is an obvious research path.
Adverse effects of aging include exponential increase of death rate with age at least until approximately age 100 in humans [20]. Death is a very determinable factor not subject to interpretation and placebo effect, so trials in elderly humans may be a rapid method for determining effectiveness of anti-aging agents.
CONCLUSIONS
Aging is a feature (not a defect) of evolved mammal design that serves an evolutionary purpose.
Mammal aging is controlled by a logically central complex evolved adaptive program mechanism that involves detection of external and internal conditions that affect the optimum aging function.
The species-unique aging function is highly dependent on the reproductive characteristics of a particular mammal species as well as external circumstances surrounding a particular population.
While many different damage mechanisms cause most instances of the many different age-related diseases and conditions, a single controlling mechanism determines timing of those events. This has potentially immense consequences for our ability to treat or prevent diseases and conditions caused by aging.
Multiple characteristics of diploid reproduction increase evolvability.
Scientific arguments about the nature of aging now extending for more than a century are mainly based on disagreements regarding fine details of the evolution process.
Ethics declarations. The author declares no conflicts of interest in financial or any other sphere. This article does not contain any studies with human participants or animals performed by the author.
REFERENCES
1.Lackey, R., Lach, D., and Duncan, S. (2006)
Salmon 2100: The Future of Wild Pacific Salmon, Bethesda,
American Fisheries Society.
2.Darwin, C. (1859) On the Origin of Species by
Means of Natural Selection, London Murray, London.
3.Darwin, C. (1872) The Origin of Species 6th
Edition, London Murray, London.
4.Medawar, P. (1952) An Unsolved Problem of
Biology, Lewis, London.
5.Williams, G. (1957) Pleiotropy, Natural
Selection and the Evolution of Senescence. Evolution 11, pp.
398-411.
6.Loison, A., Festa-Bianchet, M., Gaillard, G.-M.,
Jorgenson, J., and Jullien, J.-M. (1999) Age-specific survival in five
populations of ungulates: evidence of senescence, Ecology,
80, 2539-2554, doi:
10.1890/0012-9658(1999)080[2539:ASSIFP]2.0.CO;2.
7.Weismann, A. (1882) Uber die Dauer des
Lebens [in Deutsch], Fischer, Jena.
8.Williams, G. (1966) Adaptation and Natural
Selection: A Critique of Some Current Evolutionary Thought,
Princeton UP.
9.Olshansky, S., Hayflick, L., and Carnes, B. (2002)
No truth to the fountain of youth, Sci. Am., 286,
92-95, doi: 10.1038/scientificamerican0602-92.
10.Sinha, J. K., Ghosh, S., and Raghunath, M. (2014)
Progeria: a rare genetic premature ageing disorder, Ind. J. Med.
Res., 139, 667-674.
11.Coppedè, F. (2013) The epidemiology of
premature aging and associated comorbidities, Clin. Intervent.
Aging, 8, 1023-1032, doi: 10.2147/CIA.S37213.
12.Guerin, J. (2004) Emerging area of aging
research: long-lived animals with “negligible senescence”,
Ann. NY Acad. Sci., 1019, 518-520, doi:
10.1196/annals.1297.096.
13.Aging Changes in Hormone Production, in
A.D.A.M Medical Encyclopedia, URL: https://medlineplus.gov/ency/article/004000.htm.
14.Conboy, I., Conboy, M., Wagers, A. J., Girma, E.
R., Weissman, I. L., et al. (2005) Rejuvenation of aged progenitor
cells by exposure to a young systemic environment, Nature,
433, 760-764, doi: 10.1038/nature03260.
15.Skulachev, V. (2011) Aging as a particular case
of phenoptosis, the programmed death of an organism (a response to
Kirkwood and Melov “On the programmed/non-programmed nature of
aging within the life history”), Aging (Albany NY),
3, 1120-1123, doi: 10.18632/aging.100403.
16.Goldsmith, T. (2013) Arguments against
non-programmed aging theories, Biochemistry (Moscow),
78, 971-978, doi: 10.1134/S0006297913090022.
17.Mitteldorf, J. (2006) Chaotic population dynamics
and the evolution of ageing, Evol. Ecol. Res.,
8, 561-574.
18.Skulachev, V. (1997) Aging is a specific
biological function rather than the result of a disorder in complex
living systems: biochemical evidence in support of Weismann’s
hypothesis, Biochemistry (Moscow), 62, 1191-1195.
19.Libertini, G. (1988) An adaptive theory of
increasing mortality with increasing chronological age in populations
in the wild, J. Theor. Biol., 132, 145-162, doi:
10.1016/s0022-5193(88)80153-x.
20.Goldsmith, T (2014) The Evolution of Aging 3rd
edition, Azinet.
21.Wynne-Edwards, V (1962) Animal Dispersion in
Relation to Social Behaviour, Oliver & Boyd, Edinburgh.
22.Hamilton, W. (1963) The evolution of altruistic
behavior, Am. Naturalist, 97, 354-356.
23.Dawkins, R. (1976) The Selfish Gene,
Oxford University Press.
24.Wagner, G., and Altenberg, L. (1996) Perspective:
Complex adaptations and the evolution of evolvability,
Evolution, 50, 3, doi: 10.2307/2410639.
25.Gu, D., and Dupre, M. (2020) Encyclopedia of
Gerontology and Population Aging, Springer Cham,
Biogerontology-General, doi: 10.1007/978-3-319-69892-2.
26.Krebs, J. E., Goldstein, E. S., and Kilpatrick,
S. T. (2017) Lewin’s GENES XII, 12th Edn., Jones &
Bartlett Learning, Burlington.
27.Watson, J., and Crick, F. (1953) A structure for
deoxyribose nucleic acid, Nature, 171, 737-738, doi:
10.1038/171737a0.
28.Goldsmith, T. C. (2019) Digital genetics,
variation, evolvability, and the evolution of programmed aging,
Biochemistry (Moscow), 84, 1451-1457, doi:
10.1134/S0006297919120046.
29.Hart, J. (2011) Seasonal changes in insulation of
fur, Can. J. Zool., 34, 53-57, doi: 10.1139/z56-007.
30.Apfeld, J., and Kenyon, C. (1999) Regulation of
lifespan by sensory perception in Caenorhabditis elegans,
Nature, 402, 804-809, doi: 10.1038/45544.
31.Spindler, S. (2005) Rapid and reversible
induction of the longevity, anticancer and genomic effects of caloric
restriction, Mech. Ageing Dev., 126, 960-966, doi:
10.1016/j.mad.2005.03.016.